Advanced Metal-Triggered Trifluoromethylation for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce fluorine atoms into complex molecular scaffolds, particularly the trifluoromethyl group, due to its profound impact on metabolic stability and lipophilicity. Patent CN101973829A introduces a groundbreaking approach for the trifluoromethylation of aromatic heterocyclic compounds utilizing trifluoromethyl aryl sulfonium salts under metal initiation. This technology represents a significant leap forward from conventional electrophilic or nucleophilic strategies, offering a pathway to high-purity intermediates with exceptional efficiency. By leveraging the reactivity of sulfonium salts in the presence of reducing metals such as copper, zinc, or magnesium, this method achieves nearly quantitative yields under remarkably mild conditions. For R&D directors and process chemists, this patent provides a critical solution to the long-standing challenge of functionalizing electron-deficient or sterically hindered heterocycles, which are ubiquitous in modern drug discovery pipelines.
The versatility of this chemical transformation is underscored by its compatibility with a wide array of heterocyclic cores, ranging from simple pyridines to fused systems like imidazothiazoles and benzimidazoles. The ability to tolerate diverse functional groups without extensive protection-deprotection sequences makes this route highly attractive for the synthesis of complex API intermediates. Furthermore, the reaction proceeds in common polar organic solvents like DMF, DMSO, or THF, facilitating easy integration into existing manufacturing workflows. As we delve deeper into the technical specifics, it becomes evident that this metal-triggered mechanism not only solves synthetic bottlenecks but also aligns perfectly with the industry's demand for greener, more cost-effective production methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into aromatic heterocycles has been fraught with significant technical hurdles that limit scalability and economic viability. Traditional electrophilic trifluoromethylation relies heavily on electron-rich systems, rendering it ineffective for the vast majority of nitrogen-containing heterocycles which are inherently electron-deficient. Conversely, nucleophilic approaches using reagents like TMSCF3 often require stringent anhydrous conditions and strong bases, leading to compatibility issues with sensitive functional groups commonly found in pharmaceutical candidates. Perhaps most problematic is the reliance on free-radical initiated methods, which frequently suffer from poor regioselectivity, necessitating difficult purification steps that drive up waste and cost. Additionally, many legacy processes demand high temperatures or intense UV irradiation, posing safety risks and energy inefficiencies that are unacceptable in modern GMP manufacturing environments. These limitations collectively create a bottleneck in the supply chain for fluorinated building blocks, forcing procurement teams to rely on expensive custom synthesis or multi-step routes that erode profit margins.
The Novel Approach
The methodology disclosed in CN101973829A circumvents these historical constraints by employing a unique metal-reduction strategy that activates trifluoromethyl aryl sulfonium salts to generate reactive trifluoromethyl-metal species in situ. This novel pathway allows for the direct substitution of halogen atoms (iodine or bromine) on the heterocyclic ring with a trifluoromethyl group, effectively bypassing the electronic limitations of electrophilic attacks. The reaction conditions are exceptionally mild, typically operating between 0°C and 120°C, and surprisingly, do not always mandate rigorous exclusion of air or moisture, which drastically simplifies reactor setup and operation. By utilizing inexpensive and abundant metals like copper powder as the trigger, the process eliminates the need for precious metal catalysts often associated with cross-coupling reactions, thereby delivering substantial cost reduction in pharmaceutical intermediate manufacturing. The result is a streamlined, high-yielding process that transforms difficult-to-access fluorinated heterocycles into commodities accessible for large-scale production.
Mechanistic Insights into Metal-Initiated Trifluoromethylation
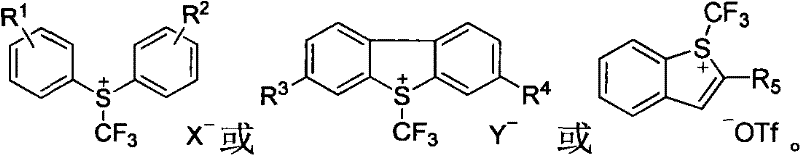
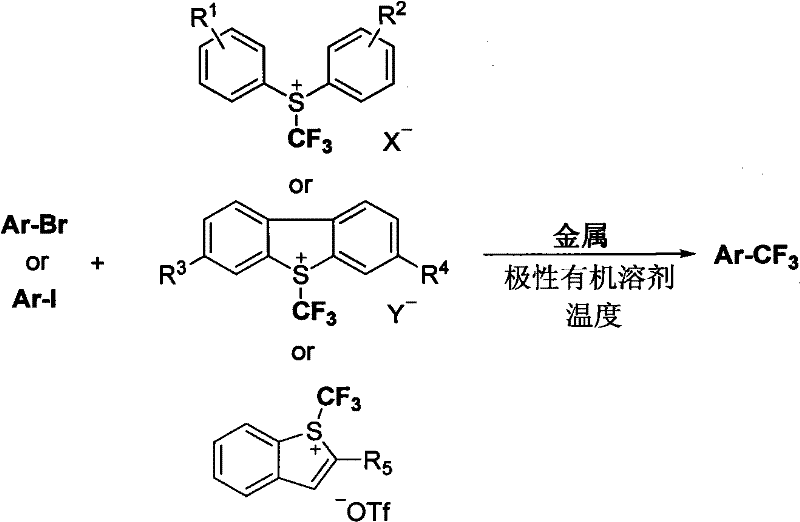
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The core of this chemistry lies in the single-electron transfer (SET) from the reducing metal to the sulfonium salt, which cleaves the sulfur-carbon bond to release a trifluoromethyl radical or generate a trifluoromethyl-metal complex. This reactive intermediate then engages with the halogenated heterocycle, likely through a radical abstraction or oxidative addition-reductive elimination cycle depending on the specific metal used. The beauty of this mechanism is its tolerance; because the reactive species is generated in a controlled manner by the metal surface, side reactions such as homocoupling or decomposition of the heterocyclic core are minimized. This controlled reactivity ensures that even complex molecules with multiple potential reaction sites can be trifluoromethylated with high fidelity, preserving the integrity of the molecular scaffold.
Furthermore, the choice of counterions in the sulfonium salt, such as SbF6 or OTf, plays a pivotal role in stabilizing the cationic precursor and influencing the kinetics of the reduction step. The patent elucidates that the nature of the substituents on the sulfonium salt's aromatic rings can also be tuned to modulate the reduction potential, offering a handle for fine-tuning the reaction rate. For quality control professionals, this mechanistic clarity translates to a predictable impurity profile, as the primary byproducts are well-defined sulfur species and metal salts that are easily removed during aqueous workup. This level of control is essential for meeting the stringent purity specifications required for clinical-grade materials, ensuring that the final API is free from genotoxic impurities or heavy metal residues that could derail regulatory approval.
How to Synthesize Trifluoromethylated Heterocycles Efficiently
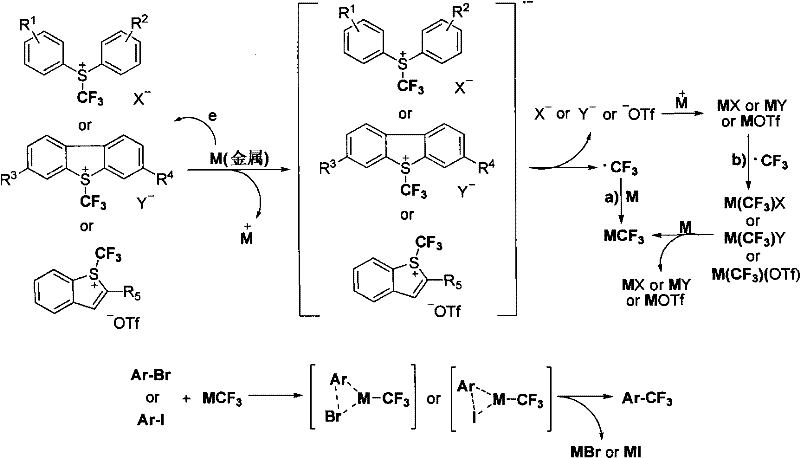
The practical implementation of this technology involves a straightforward protocol that balances reaction efficiency with operational simplicity. Typically, the halogenated heterocyclic substrate and the trifluoromethyl aryl sulfonium salt are dissolved in a polar aprotic solvent, followed by the addition of the metal powder. The mixture is then heated to the desired temperature, allowing the reaction to proceed to completion over a period of 2 to 30 hours. Workup generally involves dilution with an organic solvent like ether or ethyl acetate, followed by aqueous washing to remove inorganic salts and sulfur byproducts. The crude product is often pure enough for direct use or requires only simple column chromatography for final polishing. For detailed standard operating procedures and specific parameter optimization for your target molecule, please refer to the comprehensive guide below.
- Prepare the reaction mixture by dissolving the halogen-substituted heterocyclic compound (Ar-X) and the trifluoromethyl aryl sulfonium salt in a polar organic solvent such as DMF or DMSO.
- Add the reducing metal powder (e.g., Copper, Zinc, or Magnesium) to the solution under standard atmospheric conditions or inert atmosphere depending on specific substrate sensitivity.
- Heat the reaction mixture to a temperature between 0°C and 120°C for 2 to 30 hours, then isolate the trifluoromethylated product via standard workup and purification techniques.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-triggered trifluoromethylation technology offers transformative benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies. The shift away from exotic reagents and harsh conditions towards commodity chemicals like copper powder and common solvents fundamentally alters the cost structure of producing fluorinated intermediates. This process simplification reduces the dependency on specialized equipment capable of handling high pressures or cryogenic temperatures, thereby lowering capital expenditure requirements for manufacturing partners. Moreover, the high yields reported in the patent examples minimize raw material waste, directly contributing to a more sustainable and economically efficient supply chain. For companies scaling up production, these factors combine to create a resilient sourcing model that is less susceptible to market volatility in reagent pricing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric amounts of cheap metals like copper or zinc significantly lowers the bill of materials. Additionally, the ability to run reactions without strict anhydrous or anaerobic setups reduces energy consumption and infrastructure costs, leading to substantial overall savings in production expenses. This economic efficiency allows for more competitive pricing of the final API, enhancing market positioning.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, specifically trifluoromethyl aryl sulfonium salts and halogenated heterocycles, are becoming increasingly available from global chemical suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production can be transferred between different manufacturing sites with minimal re-validation, ensuring continuity of supply even in the face of regional disruptions. This flexibility is critical for maintaining just-in-time inventory levels for high-demand pharmaceutical products.
- Scalability and Environmental Compliance: The mild thermal profile and the generation of manageable inorganic byproducts make this process highly amenable to scale-up from gram to ton quantities without significant engineering challenges. The reduced need for hazardous reagents and the potential for solvent recycling align with modern green chemistry principles, simplifying environmental permitting and waste disposal compliance. This ensures that manufacturing operations remain sustainable and compliant with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
To further assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope and limitations of this trifluoromethylation method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a realistic view of what can be achieved in a production setting. Understanding these nuances is key to successful project planning and risk mitigation.
Q: What are the primary advantages of using metal-triggered sulfonium salts for trifluoromethylation compared to traditional radical methods?
A: Unlike traditional radical-induced trifluoromethylation which often suffers from poor selectivity and requires harsh UV initiation or high temperatures, the metal-triggered method described in CN101973829A operates under mild thermal conditions (0-120°C) with excellent chemoselectivity and nearly quantitative yields.
Q: Which types of heterocyclic substrates are compatible with this trifluoromethylation protocol?
A: This method demonstrates broad substrate scope, successfully trifluoromethylating various five-to-twelve-membered heterocycles containing nitrogen, oxygen, or sulfur, including pyridines, pyrazoles, indoles, benzoxazoles, and imidazothiazoles, provided they possess a suitable leaving group like iodine or bromine.
Q: Does this process require strictly anhydrous and anaerobic conditions for industrial application?
A: While inert conditions can optimize sensitive reactions, the patent indicates that the trifluoromethylation can be conducted in polar organic solvents with or without strict water and oxygen removal, significantly simplifying the operational complexity and reducing equipment costs for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorination technologies in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the metal-triggered trifluoromethylation route described in CN101973829A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors and rigorous QC labs capable of handling the specific requirements of sulfonium salt chemistry, ensuring that every batch meets stringent purity specifications required for clinical and commercial supply. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry, delivering high-quality intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your pipeline projects. Whether you require custom synthesis of novel trifluoromethylated building blocks or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall cost of goods sold.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
