Advanced Synthesis of 4-Methyl-5-Hydroxymethylthiazole for Scalable Cephalosporin Production
The pharmaceutical industry constantly seeks robust synthetic routes for critical antibiotic intermediates, and patent CN100360511C presents a transformative approach to producing 4-methyl-5-hydroxymethylthiazole. This compound serves as a pivotal building block for Cefditoren Pivoxil, a third-generation cephalosporin antibacterial agent widely used in clinical settings. The disclosed methodology replaces hazardous and waste-intensive Lewis acid systems with a benign base-catalyzed borohydride reduction, marking a significant leap forward in green chemistry for beta-lactam synthesis. By enabling reactions in standard organic solvents without stringent anhydrous requirements, this innovation addresses long-standing pain points regarding operational safety and environmental compliance. For R&D directors and process chemists, this patent offers a validated pathway to high-purity intermediates that aligns with modern regulatory expectations for impurity control and waste minimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-methyl-5-hydroxymethylthiazole relied heavily on reduction systems employing Lewis acids such as aluminum chloride coupled with sodium borohydride. While chemically feasible, this legacy approach suffers from severe industrial drawbacks that impact both cost and sustainability profiles. The requirement for strictly anhydrous solvents introduces significant expense and safety risks, as drying large volumes of solvent is energy-intensive and prone to hazards. Furthermore, the post-reaction workup is notoriously difficult, necessitating the consumption of large quantities of hydrochloric acid and sodium hydroxide to manage the reaction mixture. Most critically, this process generates substantial amounts of flocculent aluminum salt precipitates, creating a sludge disposal problem that complicates filtration and increases the environmental footprint of the manufacturing facility.
The Novel Approach
In stark contrast, the novel method described in the patent utilizes sodium borohydride or potassium borohydride in the presence of a simple base catalyst, completely bypassing the need for Lewis acids. This shift allows the reaction to proceed in common organic solvents like alcohols or ethers at moderate temperatures ranging from -20 to 100°C, with a preferred window of 60 to 80°C. The elimination of aluminum species means the reaction can be quenched simply by adding water, avoiding the complex acid-base neutralization steps required by older technologies. This streamlined workflow not only simplifies the equipment requirements but also drastically reduces the generation of hazardous waste, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
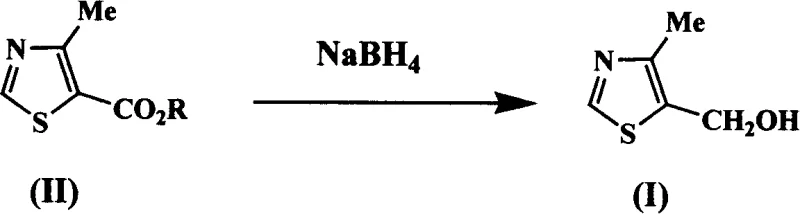
Mechanistic Insights into Base-Catalyzed Borohydride Reduction
The core chemical innovation lies in the activation of the borohydride species by the base catalyst, which facilitates the nucleophilic attack on the carbonyl carbon of the thiazole ester without the need for Lewis acid coordination. In traditional Lewis acid mechanisms, the metal center coordinates with the carbonyl oxygen to increase electrophilicity, but this creates stable complexes that are hard to break down during workup. Here, the base catalyst, which can range from inorganic hydroxides like sodium hydroxide to organic amines like pyridine or triethylamine, likely enhances the hydride transfer efficiency or stabilizes the transition state through hydrogen bonding or electrostatic interactions. This subtle mechanistic adjustment allows the reduction to proceed smoothly even in the presence of trace moisture, which would typically deactivate sensitive Lewis acid catalysts. Understanding this mechanism is crucial for process chemists aiming to optimize reaction kinetics and minimize the formation of over-reduced byproducts or ring-opened impurities.
Impurity control is another critical aspect where this mechanism offers distinct advantages over prior art. The absence of aluminum salts prevents the formation of metal-organic complexes that often trap product or create emulsions during extraction. The patent data indicates that by carefully controlling the molar ratio of the ester to the reducing agent, typically between 1:1.0 and 1:6.0, manufacturers can achieve high conversion rates while suppressing side reactions. For instance, using a slight excess of borohydride ensures complete consumption of the starting material, while the base catalyst loading is kept low, generally between 0.01 and 0.2 equivalents, to avoid promoting unwanted degradation of the thiazole ring. This precise balance results in a crude product profile that is significantly cleaner, facilitating easier downstream purification via simple distillation rather than complex chromatography.
How to Synthesize 4-Methyl-5-Hydroxymethylthiazole Efficiently
To implement this synthesis effectively, operators should follow a standardized protocol that leverages the flexibility of the solvent system and the robustness of the catalyst. The process begins by dissolving the 4-methyl-5-thiazole carboxylate starting material in a chosen organic solvent, such as ethanol, isopropanol, or tetrahydrofuran, ensuring complete solubilization before reagent addition. A base catalyst is then introduced to the mixture, followed by the controlled addition of the borohydride reducing agent while maintaining the temperature within the specified range to manage exotherms. The detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations.
- Dissolve 4-methyl-5-thiazole carboxylate in an organic solvent such as ethanol or tetrahydrofuran and add a base catalyst like pyridine or sodium hydroxide.
- Add sodium borohydride or potassium borohydride at a temperature range of -20 to 100°C, preferably maintaining 60 to 80°C for optimal kinetics.
- Quench the reaction by adding water directly, extract the product with ethyl acetate or ether, and purify via vacuum distillation to obtain the yellow oil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation strategies. The primary value driver is the drastic simplification of the post-reaction workup, which eliminates the need for expensive acid and base neutralization chemicals and the associated handling costs. By removing the generation of aluminum sludge, facilities can avoid the high disposal fees and regulatory burdens associated with hazardous solid waste, leading to substantial cost savings in waste management budgets. Additionally, the ability to use non-anhydrous solvents reduces the raw material costs and energy consumption related to solvent drying, further enhancing the overall economic viability of the production process.
- Cost Reduction in Manufacturing: The elimination of Lewis acids like aluminum chloride removes a significant cost center associated with both reagent purchase and waste disposal. Traditional methods require extensive washing and filtration to remove aluminum salts, which consumes time, labor, and utilities; this new method allows for direct water quenching and extraction, streamlining the batch cycle time. Furthermore, the reduced complexity of the purification train means less solvent is consumed overall, and equipment turnaround times are improved, allowing for higher throughput without capital investment in new hardware.
- Enhanced Supply Chain Reliability: Sourcing high-purity anhydrous solvents and managing their storage can be a logistical bottleneck, especially in regions with strict safety regulations. This process tolerates standard grade solvents, making the supply chain more resilient to market fluctuations and availability issues. The robustness of the reaction conditions also means that production is less susceptible to batch failures caused by minor variations in moisture content, ensuring a consistent and reliable supply of the critical intermediate for downstream antibiotic formulation.
- Scalability and Environmental Compliance: As regulatory pressure mounts on pharmaceutical manufacturers to reduce their environmental footprint, this technology offers a clear path to greener manufacturing. The absence of heavy metal waste simplifies environmental permitting and reduces the risk of compliance violations. The process is inherently safer due to the lack of pyrophoric reagents or highly corrosive acids, creating a better working environment for plant personnel and reducing insurance and liability costs associated with hazardous chemical handling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: How does this new method eliminate aluminum waste compared to traditional Lewis acid reductions?
A: Traditional methods utilize aluminum chloride which generates massive amounts of flocculent aluminum salt sludge requiring complex acid/base neutralization. This patented process uses simple inorganic bases or amines, allowing reaction quenching with just water, thereby fundamentally eliminating heavy metal waste streams.
Q: Does this synthesis require expensive anhydrous solvent conditions?
A: No, a major breakthrough of this technology is that it operates effectively without strictly anhydrous conditions. This removes the high cost and safety hazards associated with drying solvents, significantly simplifying the operational requirements for large-scale manufacturing.
Q: What purity levels can be achieved with this borohydride reduction route?
A: Experimental data within the patent demonstrates consistent HPLC purity exceeding 99.0%, with specific examples reaching 99.6%. The clean reaction profile minimizes side products, reducing the burden on downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-5-Hydroxymethylthiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the global antibiotic supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of multinational pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 4-methyl-5-hydroxymethylthiazole meets the highest standards for safety and efficacy. Our infrastructure is designed to support both pilot-scale development and full commercial manufacturing, providing a seamless transition from process optimization to market supply.
We invite you to collaborate with us to leverage this advanced synthetic technology for your cephalosporin production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistics constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain efficiency and product quality.
