Revolutionizing Quaternary Ammonium Synthesis for Electronics and Ionic Liquids
Revolutionizing Quaternary Ammonium Synthesis for Electronics and Ionic Liquids
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the demand for ultra-high purity materials, particularly in the semiconductor and electronic sectors. A pivotal advancement in this domain is detailed in patent CN101039913A, which outlines a novel method for producing high-purity quaternary ammonium compounds. This technology addresses critical limitations associated with conventional methylation agents by utilizing dimethyl sulfite in the presence of specific Brønsted acids. For R&D directors and procurement specialists seeking reliable quaternary ammonium compound supplier partnerships, this process represents a paradigm shift towards safer, more efficient, and higher-quality production capabilities. The ability to synthesize these vital intermediates without the carcinogenic risks of dimethyl sulfate or the harsh conditions of methyl chloride positions this methodology as a cornerstone for next-generation electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quaternary ammonium compounds has relied heavily on methylating agents such as dimethyl sulfate and methyl chloride, both of which present substantial operational and safety challenges. Dimethyl sulfate, while reactive, is a known carcinogen that necessitates rigorous safety protocols, specialized containment infrastructure, and complex waste treatment procedures, thereby inflating operational expenditures. Alternatively, methyl chloride suffers from low reactivity, requiring elevated temperatures and high pressures that often trigger undesirable secondary reactions, leading to reduced yields and complicated downstream purification. Furthermore, traditional two-step processes involving anion exchange frequently result in products contaminated with significant levels of mesylate impurities, rendering them unsuitable for sensitive applications like electrolyte salts in electronic devices where ionic purity is paramount.
The Novel Approach
The innovative methodology described in the patent data overcomes these hurdles by employing dimethyl sulfite as a methylating agent under mild, acid-catalyzed conditions. This approach operates effectively at temperatures ranging from 10°C to 100°C and avoids the need for toxic reagents or extreme pressure vessels. By introducing an inorganic or organic Brønsted acid with a pKa between 1.8 and 14, the reaction achieves high conversion rates while simultaneously suppressing the rearrangement of methylsulfite anions to mesylates. This results in a direct, one-pot synthesis that eliminates the need for separate anion exchange steps, drastically simplifying the workflow. For supply chain heads, this translates to a streamlined production cycle with fewer unit operations, reducing lead time for high-purity quaternary ammonium compounds and enhancing overall process reliability.
Mechanistic Insights into Acid-Catalyzed Methylation
The core of this technological breakthrough lies in the precise control of the reaction environment through the use of specific protic acids. When sp3-hybridized tertiary amines or sp2-hybridized tertiary imines react with dimethyl sulfite, the presence of the acid catalyst facilitates the methylation process under remarkably mild thermal conditions. The mechanism ensures that the methylsulfite intermediate decomposes cleanly into volatile sulfur dioxide and methanol upon heating, leaving behind the desired quaternary ammonium cation paired with the anion derived from the added acid. This decomposition pathway is crucial because it prevents the accumulation of non-volatile sulfur-containing byproducts that typically plague conventional syntheses. The ability to tune the anion simply by selecting the corresponding acid offers unparalleled flexibility for customizing ionic liquids and specialty salts.
Furthermore, the suppression of side reactions is a critical mechanistic advantage of this system. In prior art methods, the methylsulfite anion often undergoes rearrangement to form stable mesylate impurities that are difficult to remove and detrimental to product performance. The specific acidic conditions defined in this patent effectively inhibit this rearrangement, ensuring that the final product maintains exceptional chemical integrity. This level of impurity control is essential for applications in the electronics industry, where trace contaminants can compromise device performance. The structural versatility of the reactants is also notable, accommodating a wide range of amine and imine substrates as illustrated by the general formula for imidazoles below.
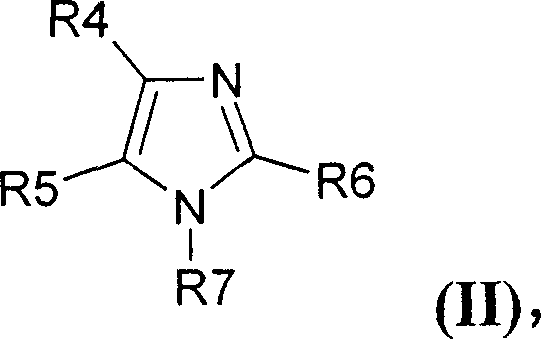
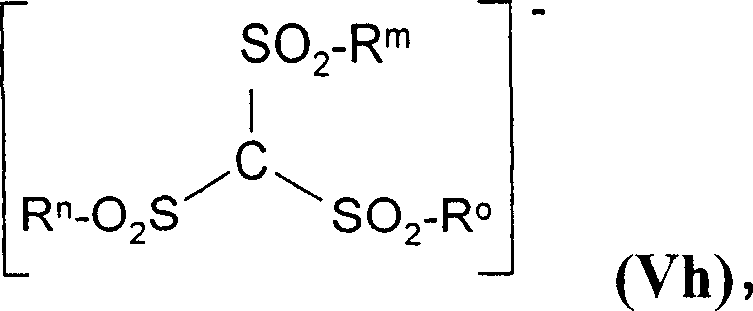
The versatility extends to the anions as well, allowing for the incorporation of complex structures such as the methide ion shown below, which is vital for advanced electrolyte formulations. The capacity to integrate such specialized anions without compromising purity underscores the robustness of this catalytic system.
How to Synthesize High-Purity Quaternary Ammonium Compounds Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process begins with the combination of the tertiary amine or imine substrate with dimethyl sulfite and the chosen protic acid in a suitable reactor. Maintaining the molar ratio of acid to amine between 0.9 and 1.5 is critical to ensure complete reaction without excess reagent carryover. The reaction mixture is then heated to the optimal temperature window, typically between 10°C and 100°C depending on the specific substrate reactivity, and held for a duration ranging from 0.5 to 24 hours. Detailed standardized synthesis steps see the guide below.
- Combine sp3-hybridized tertiary amines or sp2-hybridized tertiary imines with dimethyl sulfite and a protic acid (pKa 1.8-14) in a reaction vessel.
- Maintain the reaction mixture at a temperature between 10°C and 100°C under controlled pressure conditions to facilitate methylation.
- Remove volatile byproducts like sulfur dioxide and methanol via vacuum or heating, yielding the high-purity quaternary ammonium salt directly.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing process offers profound advantages that directly impact the bottom line and supply chain resilience. By eliminating the need for carcinogenic dimethyl sulfate, facilities can significantly reduce costs associated with safety compliance, hazardous waste disposal, and insurance liabilities. The simplified one-pot nature of the reaction removes the engineering complexity of multi-step anion exchange processes, leading to substantial cost savings in manufacturing overhead and capital equipment requirements. Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, avoiding the need for expensive high-pressure autoclaves, which further lowers the barrier to entry for production scale-up.
- Cost Reduction in Manufacturing: The elimination of toxic reagents and complex purification steps drives down the total cost of ownership for producing these specialty chemicals. By avoiding the expensive removal of heavy metal catalysts or the handling of hazardous gases like methyl chloride, manufacturers can achieve a more economical production profile. The high atom economy of the dimethyl sulfite route, coupled with the volatility of the byproducts, minimizes raw material waste and reduces the load on effluent treatment plants, contributing to long-term operational efficiency.
- Enhanced Supply Chain Reliability: Utilizing readily available and less regulated raw materials like dimethyl sulfite enhances supply chain stability compared to relying on strictly controlled substances like dimethyl sulfate. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or stringent regulatory audits. This reliability ensures consistent delivery schedules for downstream customers in the pharmaceutical and electronic sectors, fostering stronger long-term partnerships and reducing the risk of stockouts.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production without significant re-engineering. The generation of volatile byproducts like sulfur dioxide and methanol allows for easy separation and potential recycling, aligning with green chemistry principles. This environmental compatibility simplifies permitting processes and supports corporate sustainability goals, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis technology. Understanding these details is crucial for evaluating the feasibility of adopting this method for specific product lines. The answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy.
Q: How does this method improve purity compared to traditional dimethyl sulfate routes?
A: Traditional methods often leave significant mesylate impurities due to anion rearrangement. This acid-catalyzed dimethyl sulfite route suppresses that rearrangement, achieving purities greater than 97% suitable for electronics.
Q: What types of anions can be introduced using this protocol?
A: The process allows flexible introduction of various anions including tetrafluoroborate, hexafluorophosphate, trifluoromethanesulfonate, and carboxylates by selecting the appropriate protic acid.
Q: Is this process scalable for industrial production of ionic liquids?
A: Yes, the reaction operates at mild temperatures (10-100°C) and atmospheric to low pressures, making it highly suitable for batch or continuous commercial scale-up without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quaternary Ammonium Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of advanced chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of quaternary ammonium compounds meets the exacting standards required by the global electronics and pharmaceutical industries. Our commitment to quality assurance means that clients can trust our materials for their most sensitive applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your product quality while optimizing your operational costs.
