Advanced Manufacturing of Base-Modified Nucleotides for Next-Generation DNA Sequencing Applications
The rapid evolution of DNA sequencing technologies, particularly the transition towards third-generation single-molecule sequencing, has created an urgent demand for high-purity, chemically modified nucleotides. Patent CN103588838A addresses a critical bottleneck in the biotechnology supply chain by disclosing a robust synthesis method for base-modified nucleotides, specifically focusing on the efficient generation of 5'-nucleoside triphosphates. These compounds serve as essential reversible terminators in modern sequencing platforms, yet their historical production has been plagued by complex synthetic routes, poor reaction selectivity, and prohibitive costs associated with purification. The disclosed technology offers a strategic pathway to overcome these limitations through the use of specialized phosphorylation reagents, including in situ generated cyclic agents and synergistic phosphorus oxychloride systems, thereby enabling more accessible and scalable manufacturing of these vital biochemical reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing modified nucleotide triphosphates have long suffered from inherent chemical inefficiencies that drive up costs and limit supply reliability. A primary challenge lies in the lack of regioselectivity during the phosphorylation step, where reagents often react indiscriminately with both the 3'- and 5'-hydroxyl groups of the nucleoside substrate. This non-selective reactivity results in a complex mixture of products, necessitating rigorous and expensive purification protocols such as repeated FPLC or preparative HPLC to isolate the desired 5'-triphosphate. Furthermore, conventional methods frequently require harsh reaction conditions that can degrade sensitive base modifications or lead to significant decomposition of the sugar moiety. The cumulative effect of these inefficiencies is a low overall yield and a final product with a high price point, which acts as a barrier to the widespread adoption of advanced sequencing technologies in clinical and research settings.
The Novel Approach
In contrast, the methodology outlined in the patent introduces a paradigm shift by employing milder reaction conditions and highly selective phosphorylation strategies. By utilizing specific cyclic phosphorylating agents or a协同 system of phosphorus oxychloride and tributylammonium pyrophosphate, the process achieves superior selectivity for the 5'-hydroxyl position. A key innovation involves strategically controlling the reaction conversion; by stopping the reaction when the raw material transformation reaches approximately 70%, the formation of unwanted 3'-phosphorylated byproducts is minimized. Since the starting nucleosides are significantly less expensive than the final nucleotides, this approach optimizes the cost-benefit ratio by simplifying the downstream purification burden. This novel approach not only enhances the chemical purity of the final product but also streamlines the entire manufacturing workflow, making it highly suitable for industrial scale-up.
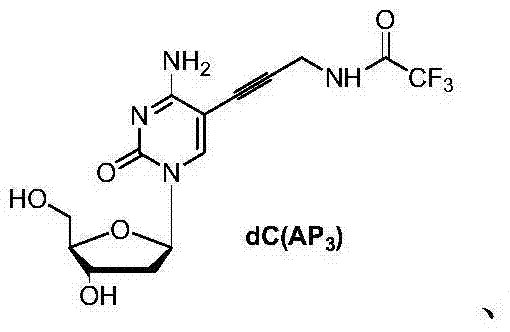
Mechanistic Insights into Selective Phosphorylation and Coupling
The core of this synthetic advancement relies on a two-stage mechanistic strategy involving transition metal catalysis followed by controlled phosphorylation. Initially, the synthesis employs a Sonogashira coupling reaction, typically catalyzed by palladium and copper species, to attach functional handles such as propargylamine to iodinated nucleoside precursors. This step is crucial for introducing the chemical moieties required for reversible termination in sequencing applications. Following the modification of the nucleobase, the phosphorylation mechanism takes center stage. The use of cyclic phosphorylating agents allows for a more defined transition state that favors attack at the primary 5'-hydroxyl group due to steric and electronic factors, effectively suppressing reaction at the secondary 3'-hydroxyl group. This mechanistic precision is further supported by the use of tributylammonium pyrophosphate, which acts as a nucleophilic catalyst to facilitate the formation of the triphosphate chain without inducing excessive side reactions.
Impurity control is intrinsically built into the reaction kinetics described in the patent. By acknowledging that the cost of the nucleoside starting material is much lower than that of the nucleotide product, the process deliberately avoids pushing the reaction to 100% completion. This kinetic control prevents the accumulation of bis-phosphorylated species (phosphorylation at both 3' and 5' positions), which are notoriously difficult to separate from the target molecule. Consequently, the crude reaction mixture contains a higher proportion of the desired product relative to traditional methods, reducing the load on purification columns and minimizing product loss during isolation. This thoughtful integration of economic considerations into the chemical mechanism ensures that the process remains viable for large-scale commercial production while maintaining the stringent purity specifications required for enzymatic applications.
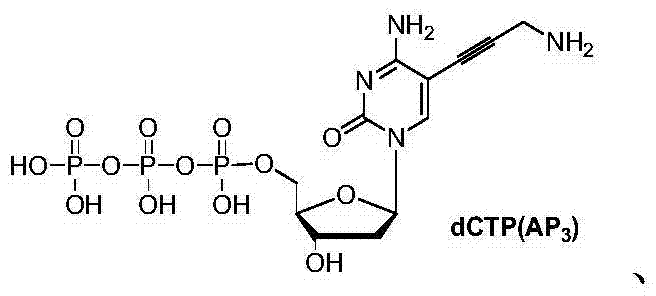
How to Synthesize Base-Modified Nucleotides Efficiently
The synthesis of these critical reagents follows a logical progression from halogenated nucleosides to functionalized triphosphates. The process begins with the preparation of iodinated precursors, such as 5-iodo-2'-deoxycytidine or 5-iodo-2'-deoxyuridine, which serve as the substrates for carbon-carbon bond formation. Subsequent steps involve the coupling of amine-functionalized alkynes to install the necessary reporter groups, followed by the critical phosphorylation sequence using the patented reagent systems. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below.
- Perform Sonogashira coupling on iodinated nucleosides (e.g., 5-iodo-2'-deoxycytidine) with propargylamine derivatives using Pd/Cu catalysts to introduce the functional handle.
- React the resulting modified nucleoside with a cyclic phosphorylating agent or a POCl3/TBAP system to selectively target the 5'-hydroxyl group.
- Control the reaction conversion to approximately 70% to minimize 3'-phosphorylation side products, followed by oxidation and hydrolysis to yield the final triphosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates directly into enhanced operational efficiency and risk mitigation. The primary value driver is the significant reduction in manufacturing complexity, which eliminates the need for multiple rounds of expensive chromatographic purification. By simplifying the isolation process, manufacturers can reduce the consumption of costly stationary phases and solvents, leading to substantial cost savings in the final product pricing. Additionally, the reliance on conventional chemical reagents and mild reaction conditions ensures that the supply chain is not dependent on exotic or hard-to-source catalysts, thereby improving the continuity of supply and reducing the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The strategic decision to halt the phosphorylation reaction at partial conversion leverages the lower cost of nucleoside starting materials to avoid the high expense of separating difficult impurities. This approach drastically reduces the burden on downstream processing units, eliminating the need for repetitive preparative HPLC runs that typically consume significant time and resources. Consequently, the overall cost of goods sold (COGS) is lowered, allowing for more competitive pricing of the final sequencing reagents without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The synthesis route utilizes widely available chemical building blocks and standard catalytic systems, such as palladium and copper salts, which are readily accessible in the global chemical market. This accessibility ensures that production schedules are not vulnerable to the supply constraints often associated with specialized biochemical reagents. Furthermore, the robustness of the reaction conditions means that the process can be reliably transferred between different manufacturing sites or scaled up without requiring bespoke equipment, ensuring a stable and continuous supply of critical sequencing components.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions, avoiding extreme temperatures or pressures, facilitates easier scale-up from laboratory to industrial production volumes. This scalability is complemented by a reduction in waste generation, as the simplified purification process requires fewer solvents and disposable chromatography media. From an environmental compliance perspective, the ability to minimize hazardous waste streams and solvent usage aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process and simplifying regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on how this method resolves traditional pain points in nucleotide manufacturing.
Q: How does this synthesis method improve purity compared to traditional phosphorylation?
A: The method utilizes specific cyclic phosphorylating agents or optimized POCl3 conditions that exhibit high selectivity for the 5'-hydroxyl group over the 3'-hydroxyl group, significantly reducing the formation of difficult-to-separate 3'-phosphorylated impurities.
Q: What are the key advantages for large-scale production of sequencing reagents?
A: The process employs conventional chemical reactions under mild conditions, avoiding extreme temperatures or pressures. Furthermore, by controlling reaction conversion and leveraging the lower cost of starting nucleosides relative to nucleotides, the need for extensive preparative HPLC purification is reduced.
Q: Can this method be applied to different nucleobases?
A: Yes, the patent demonstrates the versatility of this approach across cytidine, uridine, adenosine, and guanosine derivatives, allowing for the synthesis of a complete set of reversible terminator nucleotides required for comprehensive DNA sequencing workflows.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Base-Modified Nucleotides Supplier
As the demand for high-throughput DNA sequencing continues to surge, the ability to produce high-purity modified nucleotides at scale becomes a decisive competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your R&D and commercial needs by leveraging advanced synthetic methodologies like those described in CN103588838A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of the global biotech market. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of nucleotides meets the exacting standards required for sensitive enzymatic reactions in next-generation sequencing platforms.
We invite you to collaborate with us to optimize your supply chain for sequencing reagents. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our optimized synthesis capabilities can enhance your product quality while reducing overall procurement costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
