Scalable Synthesis of Thioalkylamine Derivatives via Oleum Esterification for Commercial Production
Scalable Synthesis of Thioalkylamine Derivatives via Oleum Esterification for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing sulfur-nitrogen containing scaffolds, which are ubiquitous in bioactive molecules. Patent CN1671656A introduces a transformative approach to the preparation of thioalkylamine derivatives, addressing critical bottlenecks in traditional synthesis routes. This novel process leverages a two-step sequence involving the reaction of amino alcohols with oleum to generate sulfate esters, followed by immediate conversion with mercaptans or their salts. By eliminating the need for isolating unstable intermediates and avoiding hazardous reagents like hydrocyanic acid or toxic aziridines, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
From a strategic procurement perspective, the significance of this patent lies in its operational simplicity and enhanced safety profile. Conventional methods often suffer from low overall yields due to multi-step sequences involving hydrolytic cleavage of complex heterocycles or the handling of volatile, dangerous precursors. In contrast, the disclosed method operates under atmospheric pressure with readily available starting materials, ensuring a consistent supply of critical building blocks. For R&D directors evaluating process feasibility, the ability to conduct the reaction in a continuous manner without intermediate evaporation represents a significant reduction in processing time and energy consumption, directly translating to improved cost efficiency in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioalkylamine derivatives has been plagued by inefficient and hazardous methodologies that hinder commercial scalability. One prevalent route involves the hydrolytic cleavage of thiazoline or thiazolidinone derivatives; however, since these precursors themselves require multiple reaction steps to prepare, the cumulative yield of the entire process becomes unacceptably low for industrial application. Another common approach utilizes the reaction of amino alcohol sulfate esters with ammonium sulfide, but this necessitates long reaction times in sealed vessels, drastically reducing throughput and increasing capital expenditure on specialized high-pressure equipment. Furthermore, methods relying on aziridine intermediates impose severe safety constraints due to the high toxicity and potential instability of aziridines, requiring rigorous containment protocols that inflate operational costs and complicate regulatory compliance for fine chemical manufacturers.
The Novel Approach
The innovative process described in CN1671656A circumvents these historical challenges through a streamlined, two-step protocol that maximizes space-time yield while minimizing risk. By reacting amino alcohols directly with oleum (sulfur trioxide in sulfuric acid), the method generates reactive sulfate esters in situ without the need for isolation or evaporation to dryness, a step that often poses thermal hazards and efficiency losses in prior art. These esters are subsequently treated with mercaptans or their alkali metal salts in the presence of a base, facilitating a clean nucleophilic substitution. This approach not only simplifies the workflow by allowing the reaction mixture to be stirred and processed continuously but also expands the substrate scope to include a wide variety of alkyl and aryl groups, providing chemists with unprecedented flexibility in designing complex thioalkylamine structures for drug discovery programs.
Mechanistic Insights into Oleum-Mediated Sulfation and Substitution
The core of this technological advancement lies in the precise control of the sulfation mechanism using oleum as both a dehydrating agent and a sulfonating source. In the first step, the hydroxyl group of the amino alcohol undergoes esterification with sulfur trioxide dissolved in sulfuric acid, typically maintaining a temperature between 80°C and 90°C to prevent carbonization while ensuring complete conversion. The use of oleum, rather than concentrated sulfuric acid alone, drives the equilibrium towards the formation of the sulfate ester by minimizing water production, which could otherwise hydrolyze the sensitive intermediate. This mechanistic nuance is critical for R&D teams, as it allows for the use of amino alcohols containing up to 15% water without compromising reaction efficiency, thereby relaxing the purity specifications for raw material procurement and further reducing input costs.
Following sulfation, the reaction mixture is diluted and neutralized to a pH of 11-12 using bases such as sodium hydroxide or potassium carbonate, creating the optimal environment for the subsequent nucleophilic attack. The thiol or thiolate anion, acting as a potent nucleophile, displaces the sulfate group in an SN2-type mechanism, forming the desired carbon-sulfur bond with high fidelity. This step is typically conducted at moderate temperatures ranging from 60°C to 80°C, which balances reaction kinetics with the stability of the amine functionality. The robustness of this mechanism is evidenced by its tolerance to diverse substituents, including halogens and alkoxy groups, ensuring that the final thioalkylamine products maintain high chemical purity with minimal formation of elimination byproducts or disulfide impurities.
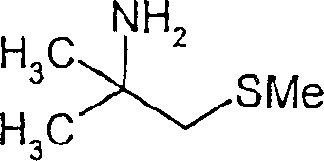
To illustrate the versatility of this synthetic route, consider the structural diversity achievable through variations in the thiol component. The process accommodates aliphatic thiols, aromatic thiols, and even heterocyclic mercaptans, enabling the synthesis of a broad library of derivatives. For instance, the reaction can successfully couple short-chain alkyl thiols to produce volatile amines or longer-chain hydroxy-functionalized thiols to generate amphiphilic surfactants. This adaptability is crucial for supply chain managers who require a single, flexible platform technology to produce multiple SKUs without retooling reactors or changing fundamental operating parameters, thus enhancing the overall agility of the manufacturing facility in responding to market demands for specialized chemical intermediates.
![Structure of 6-[(2-amino-2-methylpropyl)thio]-1-hexanol demonstrating long-chain substrate compatibility](/insights/img/thioalkylamine-synthesis-oleum-pharma-supplier-20260308194634-04.webp)
How to Synthesize Thioalkylamine Derivatives Efficiently
Implementing this synthesis route requires careful attention to thermal management and pH control to ensure optimal yields and safety. The process begins with the controlled addition of the amino alcohol to cooled oleum, followed by a heating phase to drive sulfation, and concludes with a neutralization and substitution phase where the thiol is introduced. Detailed standard operating procedures regarding stoichiometry, specifically the molar ratios of SO3 to amino alcohol and thiol to sulfate ester, are critical for reproducibility. For a comprehensive guide on the exact experimental conditions, reagent grades, and workup protocols validated by the patent examples, please refer to the standardized synthesis steps outlined below.
- React amino alcohols with oleum (containing 15-60% SO3) at 80-90°C to form sulfate esters without isolation.
- Dilute the reaction mixture with water, neutralize with base (e.g., NaOH) to pH 11-12, and add mercaptans or their salts.
- Stir at 60-80°C for several hours, then extract the product using organic solvents like methyl tert-butyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oleum-based process offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of the evaporation-to-dryness step, which is mandatory in many competing technologies, results in a drastic reduction in energy consumption and processing time per batch. This efficiency gain allows manufacturing facilities to increase their throughput significantly without expanding their physical footprint or investing in additional drying equipment, effectively lowering the unit cost of production. Furthermore, the ability to utilize amino alcohols with modest water content relaxes the quality control burden on incoming raw materials, allowing procurement teams to source from a broader supplier base and negotiate more favorable pricing terms without risking batch failure.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for energy-intensive distillation or evaporation of intermediate sulfate esters, which traditionally accounts for a significant portion of utility expenses in batch processing. Additionally, the use of inexpensive and commodity-grade reagents such as oleum and sodium hydroxide, instead of exotic catalysts or hazardous gases like hydrogen cyanide, minimizes raw material expenditures. The simplified workup procedure, which relies on standard liquid-liquid extraction rather than complex chromatographic purification, further reduces solvent usage and waste disposal costs, contributing to a leaner and more economically viable production model for high-volume thioalkylamine derivatives.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials like amino alcohols and mercaptans, this method mitigates the risk of supply disruptions often associated with specialized or regulated precursors such as aziridines. The robustness of the reaction conditions, which tolerate slight variations in temperature and moisture, ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is paramount for maintaining continuous supply to downstream pharmaceutical clients, enabling manufacturers to commit to tighter delivery schedules and build long-term partnerships based on trust and dependability in the global fine chemicals market.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed to operate safely at atmospheric pressure with mechanical stirring, which simplifies the engineering requirements for scale-up from pilot plant to commercial tonnage. The avoidance of toxic aziridines and the minimization of hazardous waste streams align with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturing sites. Moreover, the high atom economy of the substitution reaction and the potential for solvent recycling in the extraction phase support sustainability goals, making this technology an attractive option for companies aiming to reduce their carbon footprint while maintaining competitive production capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this thioalkylamine synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this process within their existing manufacturing infrastructure and quality assurance frameworks.
Q: What are the safety advantages of this oleum-based process compared to aziridine routes?
A: Unlike conventional methods requiring the preparation and isolation of highly toxic and unstable aziridines, this process utilizes stable amino alcohols and oleum under controlled temperatures, significantly reducing industrial safety risks.
Q: Does this method require energy-intensive evaporation steps?
A: No, a key advantage of this invention is that the intermediate sulfate esters do not need to be evaporated to dryness. The reaction mixture can proceed directly to the second step after dilution and neutralization, saving substantial energy.
Q: How is product purity maintained during the nucleophilic substitution step?
A: Purity is maintained by precise pH control (pH 11-12) using bases like sodium hydroxide during the thiol addition, which facilitates clean nucleophilic substitution while minimizing side reactions, followed by standard solvent extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioalkylamine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the successful commercialization of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and secure. We are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thioalkylamine derivatives we supply meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to navigate complex chemical landscapes, delivering high-quality intermediates that empower our clients' drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced oleum-mediated process can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the reliable supply of complex fine chemical intermediates.
