Advanced Manufacturing of Halomethyloxazole Intermediates for Pharmaceutical Applications
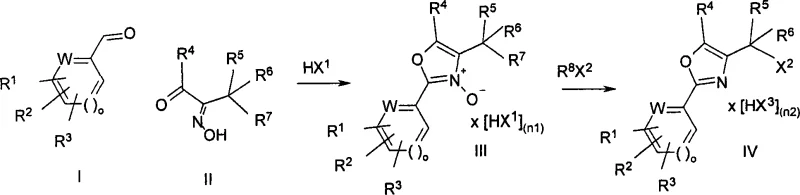
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic scaffolds, particularly oxazoles, which serve as pivotal building blocks for bioactive molecules such as PPAR agonists. Patent CN101080398A discloses a groundbreaking process for the preparation of oxazoles of formula IV by condensing aromatic aldehydes with alpha-ketoximes to form N-oxides, followed by a subsequent reaction with activated acid derivatives. This technical disclosure represents a significant leap forward in fine chemical manufacturing, addressing long-standing challenges regarding yield, purity, and operational safety. The methodology enables the conversion of aromatic aldehydes via N-oxide intermediates into halomethyloxazoles, which are essential precursors for drugs influencing lipid and glucose metabolism. For R&D directors and procurement specialists, understanding this technology is crucial for optimizing supply chains and reducing the cost of goods for complex API intermediates.
The limitations of conventional methods for synthesizing oxazoles have historically hindered efficient commercial production. Traditional literature describes the use of reagents such as phosphorus(III) chloride (PCl3), phosphorus oxychloride (POCl3), and acetic anhydride for the conversion of N-oxides to oxazoles. However, these reagents are often not widely applicable and frequently result in no product formation or highly contaminated mixtures. Obtaining sufficient purity from these conventional reactions typically necessitates costly and inconvenient purification methods, such as column chromatography, which are impractical for large-scale industrial operations. Furthermore, the reaction conditions described in older methodologies often require the isolation of the N-oxide intermediate. For N-oxides that possess the potential for exothermic decomposition, this isolation step constitutes a considerable safety risk factor, effectively preventing the implementation of these processes on an industrial scale due to hazard concerns.
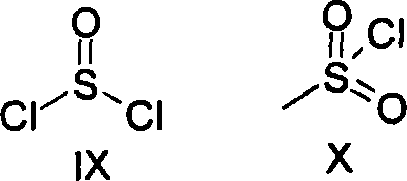
In stark contrast, the novel approach detailed in the patent utilizes inorganic thionyl halides or organic sulfonyl halides to achieve the conversion of N-oxides to halomethyloxazoles. Surprisingly, it has been found that this transformation proceeds smoothly with high yields and high purity when using these specific activating agents. A key advantage is that halomethyloxazoles can, in some cases, precipitate directly from the reaction mixture as pure free bases or salts, thereby bypassing the need for complex downstream purification. Moreover, this method allows for the safe preparation of N-oxides in dilute solution and their further direct reaction without isolation. This telescoped approach eliminates the safety hazards associated with handling unstable intermediates and streamlines the manufacturing workflow, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Thionyl and Sulfonyl Halide Activation
The mechanistic pathway involves a two-stage sequence that maximizes atomic efficiency and minimizes waste generation. Initially, an aromatic aldehyde of formula I undergoes condensation with an alpha-ketoxime of formula II in the presence of an acid HX1, such as hydrogen chloride, sulfuric acid, or trifluoroacetic acid. This step generates the N-oxide of formula III, either as a salt or a free base. The choice of acid and solvent system, such as glacial acetic acid or mixtures with toluene, is critical for controlling the reaction kinetics and ensuring complete conversion. The reaction temperature can vary broadly, but优选 temperatures between 0°C and 60°C are selected to balance reaction rate with thermal safety. This initial condensation establishes the oxazole ring framework with the N-oxide functionality poised for activation.
The second stage involves the reaction of the N-oxide intermediate with an activated acid derivative, represented by reagent R8X2. Preferred reagents include thionyl chloride (SOCl2) and methanesulfonyl chloride (MsCl). These reagents act as powerful electrophiles that facilitate the transformation of the N-oxide into the final halomethyloxazole structure. The mechanism likely involves the activation of the N-oxide oxygen, rendering it a better leaving group or facilitating a rearrangement that introduces the halomethyl group at the appropriate position on the oxazole ring. The use of solvents like dichloromethane or toluene, or even running the reaction in excess reagent, ensures homogeneous mixing and heat dissipation. This precise control over the activation step is what allows for the high purity observed, as side reactions common with phosphorus reagents are effectively suppressed.
How to Synthesize Halomethyloxazole Efficiently
The synthesis of halomethyloxazoles via this patented route offers a streamlined protocol suitable for both laboratory optimization and plant-scale production. The process begins with the careful selection of starting materials, specifically the aromatic aldehyde and the alpha-ketoxime, which determine the substitution pattern of the final oxazole. The condensation step is performed under controlled acidic conditions to form the N-oxide, which is then treated directly with the activating halide. Detailed standardized synthetic steps for implementing this chemistry are provided in the guide below, ensuring reproducibility and safety compliance.
- Condense an aromatic aldehyde with an alpha-ketoxime in the presence of an acid catalyst (such as HCl) to form the oxazole N-oxide intermediate.
- React the resulting N-oxide intermediate directly in solution with an activated acid derivative like thionyl chloride or methanesulfonyl chloride.
- Isolate the final halomethyloxazole product either as a salt or free base through filtration or extraction, avoiding chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process. By enabling the direct precipitation of products or the use of simple aqueous workups, the method eliminates the need for expensive chromatographic resin and the associated solvent consumption. This reduction in material usage translates directly into lower variable costs per kilogram of product. Additionally, the avoidance of phosphorus-based reagents, which generate significant amounts of phosphorous waste requiring specialized disposal, aligns with modern environmental compliance standards and reduces waste treatment overheads.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps represents a significant operational cost saving. Conventional methods often require silica gel columns and large volumes of eluents, which are capital and labor-intensive. By achieving high purity through crystallization or precipitation, this process reduces the demand for consumables and shortens the production cycle time. Furthermore, the reagents used, such as thionyl chloride and methanesulfonyl chloride, are commodity chemicals with stable supply chains and competitive pricing compared to specialized phosphorus reagents. This stability in raw material costs contributes to more predictable budgeting and lower overall cost of goods sold for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical process enhances supply chain continuity. Because the reaction tolerates a wide range of substrates and conditions, it is less prone to batch failures caused by minor variations in raw material quality. The ability to telescope the reaction without isolating the potentially unstable N-oxide intermediate reduces the number of unit operations and hold times. Fewer processing steps mean fewer opportunities for delays or quality deviations, ensuring a more reliable delivery schedule for downstream customers. This reliability is critical for maintaining the production schedules of pharmaceutical clients who depend on just-in-time delivery of key intermediates.
- Scalability and Environmental Compliance: From a scale-up perspective, the safety profile of this method is superior. Avoiding the isolation of energetic N-oxide intermediates removes a major bottleneck in scaling reactions from grams to tons. The process can be safely executed in standard glass-lined or stainless steel reactors without requiring exotic containment systems. Environmentally, the shift away from phosphorus waste streams simplifies effluent treatment. The byproducts generated, such as sulfonic acids or sulfur dioxide, are easier to neutralize and manage than phosphorous sludge. This facilitates regulatory approval for new manufacturing sites and supports the company's sustainability goals by reducing the overall environmental footprint of the chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to provide clarity on the feasibility, safety, and economic viability of adopting this route for commercial production of pharmaceutical intermediates.
Q: What are the safety advantages of this new oxazole synthesis method?
A: The process allows for the preparation and subsequent reaction of N-oxides in dilute solution without isolation. This mitigates the risk of exothermic decomposition associated with isolating unstable N-oxide intermediates, significantly enhancing industrial safety.
Q: How does this method improve product purity compared to conventional routes?
A: Unlike traditional methods using phosphorus chlorides which often yield highly contaminated products requiring chromatography, this novel approach using thionyl or sulfonyl halides frequently results in the direct precipitation of pure products, eliminating the need for expensive purification steps.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is specifically designed for scalability. By avoiding the isolation of hazardous intermediates and utilizing readily available reagents like thionyl chloride, the process supports robust commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halomethyloxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has extensively evaluated the synthetic route described in CN101080398A and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profiles of complex oxazole derivatives, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for oxazole-based intermediates. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our expertise in this advanced synthesis technology, you can secure a stable, cost-effective, and high-quality source of critical intermediates, accelerating your drug development timelines and enhancing your market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
