Advanced Continuous Flow Manufacturing of High-Purity Piperazine Pyridine Intermediates
Advanced Continuous Flow Manufacturing of High-Purity Piperazine Pyridine Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift from traditional batch processing to continuous flow manufacturing, particularly for complex heterocyclic intermediates used in oncology therapies. A pivotal development in this domain is documented in patent CN113121424A, which outlines a novel method for preparing piperazine pyridine compounds via a continuous reaction system. This technology specifically targets the synthesis of key precursors for CDK4/6 inhibitors like Palbociclib, addressing critical bottlenecks in heat transfer and reaction control that have long plagued batch methodologies. By leveraging micro-channel reactor technology, this process transforms a multi-hour batch operation into a matter of seconds, drastically improving throughput while maintaining stringent quality standards. For R&D directors and procurement managers alike, understanding this transition is vital, as it represents not just a chemical optimization, but a fundamental restructuring of supply chain reliability and cost efficiency in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
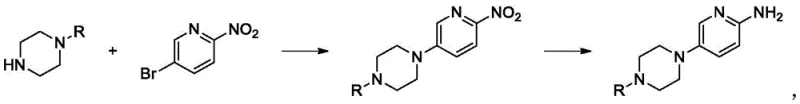
Historically, the synthesis of 4-(6-aminopyridin-3-yl)piperazine derivatives has relied heavily on traditional kettle-type batch reactors, a method fraught with inherent inefficiencies and safety hazards. In a standard batch setup, the condensation of 5-bromo-2-nitropyridine with protected piperazine often suffers from poor mass transfer efficiency, leading to localized hotspots where the concentration of reactants becomes excessively high. These thermal gradients can trigger uncontrolled exothermic events, posing significant risks of flash fires or explosions, especially during the subsequent reduction steps involving energetic nitro groups. Furthermore, the slow mixing dynamics in large vessels promote the formation of stubborn dimer impurities, which are notoriously difficult to purge during downstream purification, thereby dragging down overall yield and increasing solvent consumption. The reliance on batch processing also means that reaction times extend over several hours, during which the concentration of substrates gradually depletes, causing the reaction rate to plummet and making complete conversion increasingly difficult to guarantee without excessive reagent loading.
The Novel Approach
In stark contrast, the continuous flow methodology described in the patent introduces a highly controlled environment where reactants are mixed instantaneously within a micro-channel system. This approach allows for the precise regulation of reaction parameters, such as maintaining the condensation temperature strictly between 20°C and 60°C with a residence time of merely 80 seconds. The superior heat exchange capacity of the micro-reactor ensures that the exothermic heat generated during the nucleophilic substitution is immediately dissipated, eliminating the risk of thermal runaway. Following condensation, the stream flows directly into a hydrogenation unit—either a fixed-bed or a continuous micro-reactor—where the nitro group is reduced to an amine under controlled pressure and temperature conditions. This seamless integration of steps not only accelerates the production timeline from hours to mere minutes but also ensures a consistent product profile with significantly reduced impurity levels, offering a robust solution for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Micro-Channel Continuous Flow Synthesis
The core of this technological advancement lies in the precise manipulation of fluid dynamics and catalytic interactions within a confined spatial volume. The process initiates with a nucleophilic aromatic substitution where the piperazine nitrogen attacks the electron-deficient pyridine ring, displacing the bromine leaving group. In a micro-reactor, the laminar flow regime ensures that the diffusion distance for molecules is minimized, allowing the base (such as triethylamine) to effectively scavenge protons and drive the equilibrium forward without the concentration gradients seen in batch tanks. This rapid mixing prevents the accumulation of the intermediate species that typically leads to dimerization, a common side reaction in batch processes. The subsequent hydrogenation step utilizes a heterogeneous catalyst, such as palladium on carbon or Raney nickel, packed within a fixed bed or suspended in a three-phase micro-mixer. Here, hydrogen gas is introduced via a mass flow controller, ensuring a stoichiometric excess (1.05 to 2.0 equivalents) is maintained constantly at the catalyst surface, which maximizes the reduction efficiency while minimizing the exposure of the sensitive amine product to over-reduction or degradation.
From an impurity control perspective, the continuous flow architecture acts as a kinetic filter against byproduct formation. In traditional batch reactors, the prolonged exposure of reactants to elevated temperatures and the presence of unreacted starting materials towards the end of the cycle create a fertile ground for secondary reactions. However, the plug-flow nature of the micro-reactor ensures that every molecule of the reaction mixture experiences the exact same residence time and thermal history. This uniformity means that once the desired conversion is achieved at the outlet of the reactor, the product is immediately quenched or moved to the next stage, preventing further degradation. Additionally, the ability to perform inline liquid-liquid separation allows for the immediate removal of inorganic salts and aqueous byproducts before the hydrogenation step, protecting the expensive hydrogenation catalyst from poisoning. This level of process analytical technology integration results in a crude product purity that often exceeds 90%, significantly reducing the burden on final crystallization and chromatography steps.
How to Synthesize Piperazine Pyridine Derivatives Efficiently
Implementing this continuous flow protocol requires a systematic approach to reagent preparation and equipment configuration to ensure optimal performance. The process begins with the preparation of two distinct feed streams: Solution A, containing the piperazine compound and an organic base like triethylamine in an ether solvent, and Solution B, containing the nitropyridine substrate. These streams are pumped simultaneously into a static mixer preceding the pipeline reactor, where the temperature is rigorously controlled to facilitate the initial condensation. Following the reaction zone, a quenching stream is introduced to neutralize the mixture before it enters a liquid-liquid separator, effectively isolating the organic phase for the subsequent reduction. The detailed standardized synthesis steps, including specific pump rates, back-pressure regulator settings, and catalyst loading procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Prepare raw material streams by mixing protected piperazine with base in an ether solvent, and separately dissolving the nitropyridine substrate.
- Pump both streams into a static mixer and pipeline reactor maintained at 20-60°C for rapid condensation with a residence time of approximately 80 seconds.
- Direct the organic phase into a fixed-bed or micro-reactor for continuous hydrogenation at 40-80°C under 0.3-1.8 MPa pressure to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous flow technology translates into tangible strategic benefits that go beyond simple chemical yield improvements. The transition from batch to flow fundamentally alters the cost structure of manufacturing by decoupling production volume from reactor size, allowing for significant capital expenditure savings on large-scale infrastructure. Moreover, the drastic reduction in reaction time and the elimination of extensive purification steps required to remove dimer impurities lead to a substantial decrease in solvent consumption and waste generation. This efficiency gain directly impacts the bottom line, offering a pathway for cost reduction in pharmaceutical intermediate manufacturing that is both sustainable and economically robust. By minimizing the reliance on hazardous batch operations, companies can also reduce insurance premiums and safety compliance costs, further enhancing the overall value proposition of the supply chain.
- Cost Reduction in Manufacturing: The continuous flow process eliminates the need for large, expensive batch reactors and the associated energy costs for heating and cooling massive volumes of solvent over long periods. By achieving high conversion rates in seconds rather than hours, the throughput per unit of equipment is exponentially higher, allowing manufacturers to produce the same quantity of API intermediate with a significantly smaller physical footprint. Furthermore, the high selectivity of the micro-reactor system reduces the formation of difficult-to-remove impurities, which means less solvent is required for recrystallization and less product is lost during purification. This qualitative improvement in process efficiency drives down the cost of goods sold (COGS) without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: One of the most critical advantages for supply chain stability is the inherent consistency of continuous manufacturing. Unlike batch processes, which can suffer from variability between different batches due to slight differences in mixing or temperature ramping, flow chemistry offers a steady-state operation where every drop of product is identical. This consistency reduces the risk of batch failures and out-of-specification results, ensuring a uninterrupted supply of critical oncology intermediates to downstream drug manufacturers. Additionally, the modular nature of the equipment allows for rapid scale-up through numbering-up strategies, meaning that production capacity can be increased quickly to meet surges in demand without the long lead times associated with constructing new large-scale batch facilities.
- Scalability and Environmental Compliance: The environmental footprint of this continuous process is markedly lower than traditional methods, aligning with the increasing regulatory pressure for greener chemistry in the pharmaceutical sector. The precise control over reaction conditions minimizes the generation of hazardous waste and reduces the overall E-factor (mass of waste per mass of product). The ability to safely handle energetic intermediates and exothermic reactions in a micro-scale environment also reduces the risk of catastrophic accidents, making the facility safer for workers and the surrounding community. This improved safety profile and reduced waste generation simplify the permitting process for new manufacturing sites and ensure long-term compliance with evolving environmental regulations, securing the license to operate for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of continuous flow technology for piperazine pyridine synthesis. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of transitioning from batch to flow platforms for their specific product portfolios.
Q: How does continuous flow improve the safety of nitro-group reduction compared to batch processing?
A: Continuous flow micro-reactors possess a high surface-area-to-volume ratio, enabling exceptional heat dissipation. This prevents the thermal runaway often associated with exothermic hydrogenation of nitro compounds in large batch kettles, significantly mitigating explosion risks.
Q: What represents the primary impurity challenge in traditional batch synthesis of this intermediate?
A: Traditional batch methods often suffer from poor mass transfer and localized concentration spikes, leading to the formation of difficult-to-remove dimer byproducts. The precise mixing in flow chemistry suppresses these side reactions, enhancing crude purity.
Q: Can this micro-reactor technology be scaled for industrial tonnage production?
A: Yes, the technology utilizes 'numbering up' rather than scaling up vessel size. By operating multiple micro-reactor units in parallel, manufacturers can achieve industrial capacities while maintaining the exact reaction parameters optimized in the laboratory.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Pyridine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to continuous flow manufacturing represents the future of high-quality API intermediate production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of micro-reactor technology are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of piperazine pyridine compound meets the exacting standards required for oncology drug development. Our commitment to technological innovation allows us to offer partners a supply chain that is not only cost-effective but also resilient and adaptable to the changing needs of the global pharmaceutical market.
We invite you to collaborate with us to optimize your supply chain for critical cancer therapy intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our continuous flow capabilities can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
