Scalable Manufacturing of Chiral Alpha-Amino Suberate Intermediates via Optimized Oxidation and Wittig Olefination
Scalable Manufacturing of Chiral Alpha-Amino Suberate Intermediates via Optimized Oxidation and Wittig Olefination
The pharmaceutical industry continuously demands efficient, scalable, and cost-effective routes for non-natural amino acids, particularly those serving as critical building blocks for complex peptide therapeutics and metabolic precursors. Patent CN101168514B introduces a robust and practical synthesis method for optical activity alpha-amino suberate and alpha-amino suberic acid monoester, specifically addressing the longstanding challenges of low yield and poor enantioselectivity found in prior art. This technology leverages a concise five-step sequence starting from readily available chiral pool materials, utilizing a strategic combination of ruthenium-catalyzed oxidation and Wittig olefination to construct the eight-carbon backbone with high fidelity. By shifting away from enzymatic resolution and high-pressure asymmetric hydrogenation, this methodology offers a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in terms of process mass intensity and overall throughput. The following analysis details the technical merits and commercial viability of this advanced synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (S)-2-amino suberic acid derivatives has been plagued by significant inefficiencies that hinder large-scale adoption. Traditional approaches often rely on enzymatic kinetic resolution, which inherently caps the theoretical yield at 50% because the unwanted enantiomer is discarded or requires complex recycling processes, leading to substantial raw material waste and inflated costs. Alternatively, methods involving asymmetric catalytic hydrogenation of glutamic acid derivative aldehydes post-Wittig reaction suffer from moderate yields, typically hovering around 60%, and generate difficult-to-separate cis-trans isomers. Furthermore, these hydrogenation protocols frequently necessitate expensive chiral catalysts and operate under high temperature and high pressure conditions, posing safety risks and requiring specialized reactor infrastructure that increases capital expenditure for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
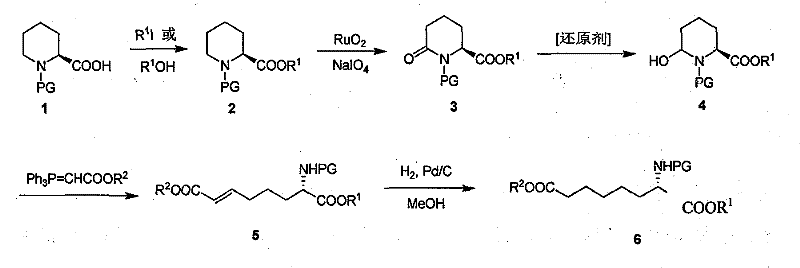
In stark contrast, the novel approach detailed in the patent utilizes (S)-N-Boc-piperidine-2-carboxylic acid as a stable, optically pure starting material, effectively bypassing the need for dynamic kinetic resolution or expensive chiral catalysts in the later stages. The route is characterized by its succinctness and logical progression: esterification followed by selective oxidation to a ketolactam, reduction to a hemiacetal, chain extension via Wittig reaction, and final hydrogenolysis. This strategy ensures that the chiral center established in the starting material is preserved throughout the synthesis, resulting in products with superior optical purity compared to the 70-96% ee range typical of older hydrogenation methods. The process conditions are notably milder, with key steps like oxidation occurring at room temperature, thereby facilitating easier commercial scale-up of complex pharmaceutical intermediates without the need for extreme operating parameters.
Mechanistic Insights into RuO4-Catalyzed Oxidation and Wittig Olefination
The cornerstone of this synthetic strategy is the highly selective oxidation of the piperidine ring alpha to the nitrogen atom, achieved using a catalytic amount of Ruthenium Oxide (RuO2) coupled with Sodium Periodate (NaIO4) as a co-oxidant. This system generates Ruthenium Tetroxide (RuO4) in situ, which acts as a potent electrophilic oxidant capable of abstracting hydride from the position adjacent to the nitrogen, ultimately converting the secondary amine functionality within the ring into a lactam carbonyl group. This transformation is critical as it activates the ring for subsequent nucleophilic attack and ring-opening strategies. The reaction proceeds efficiently in a biphasic or mixed solvent system of water and acetonitrile at ambient temperature, minimizing thermal degradation of the sensitive Boc-protected intermediate and ensuring high conversion rates within a short reaction window of 3 to 6 hours.
Following oxidation, the stereoselective reduction of the resulting ketolactam to a hemiacetal is pivotal for setting up the Wittig reaction. As illustrated in the specific embodiment using Lithium Triethylborohydride, the choice of reducing agent is crucial for chemoselectivity. 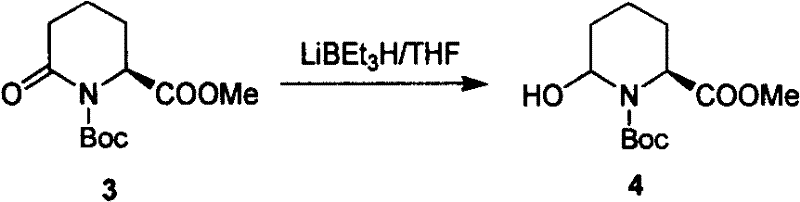 The use of bulky hydride sources like LiBEt3H or DIBAL-H at cryogenic temperatures (-78°C to -40°C) ensures that the ester moiety remains intact while the more reactive lactam carbonyl is reduced to the hemiacetal state. This hemiacetal then serves as the electrophile in the subsequent Wittig olefination, where it reacts with phosphonium ylides (such as Ph3P=CHCOOR) to extend the carbon chain by four atoms, forming the requisite suberic acid skeleton with a trans-double bond geometry that is easily saturated in the final step.
The use of bulky hydride sources like LiBEt3H or DIBAL-H at cryogenic temperatures (-78°C to -40°C) ensures that the ester moiety remains intact while the more reactive lactam carbonyl is reduced to the hemiacetal state. This hemiacetal then serves as the electrophile in the subsequent Wittig olefination, where it reacts with phosphonium ylides (such as Ph3P=CHCOOR) to extend the carbon chain by four atoms, forming the requisite suberic acid skeleton with a trans-double bond geometry that is easily saturated in the final step.
How to Synthesize (S)-2-Amino Suberic Acid Esters Efficiently
The synthesis of these valuable chiral building blocks follows a streamlined protocol designed for reproducibility and high yield at scale. The process begins with the protection and activation of the starting amino acid, followed by the critical ring oxidation and expansion steps described previously. Operators must pay close attention to temperature control during the reduction phase to prevent over-reduction or ester hydrolysis, and ensure rigorous exclusion of moisture during the Wittig step to maintain ylide stability. The final hydrogenation step is straightforward, utilizing standard palladium catalysts to saturate the alkene and remove benzyl protecting groups if applicable, yielding the target monoester or diester with high purity. For a comprehensive breakdown of the specific reagent quantities, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized operational guide below.
- Esterify (S)-N-Boc-piperidine-2-carboxylic acid using methyl iodide or DCC/DMAP coupling to form the corresponding ester.
- Oxidize the alpha-position of the piperidine ring using Ruthenium Oxide (RuO2) and Sodium Periodate (NaIO4) to generate the ketolactam intermediate.
- Reduce the ketolactam to a hemiacetal using selective reducing agents like DIBAL-H or LiBEt3H at low temperatures (-78°C).
- Perform a Wittig reaction on the hemiacetal using a phosphonium ylide reagent in refluxing acetonitrile to extend the carbon chain.
- Conduct catalytic hydrogenation using Pd/C or Pd(OH)2/C in methanol or ethanol to saturate the double bond and obtain the final amino suberate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative benefits by fundamentally altering the cost structure and risk profile of producing alpha-amino suberates. The elimination of enzymatic resolution steps immediately doubles the theoretical yield relative to the starting chiral material, drastically reducing the raw material burden per kilogram of finished product. Furthermore, the avoidance of high-pressure asymmetric hydrogenation removes the dependency on scarce and costly noble metal chiral catalysts, replacing them with more abundant and recoverable ruthenium and palladium systems that are standard in fine chemical facilities. This shift not only lowers direct material costs but also simplifies the regulatory dossier by removing complex chiral separation validation steps, thereby accelerating time-to-market for downstream drug candidates.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing inexpensive, commodity-grade oxidants like sodium periodate and avoiding the need for specialized high-pressure reactors required for asymmetric hydrogenation. The high yields reported in the experimental examples, often exceeding 90% for individual steps such as esterification and reduction, indicate a process with minimal waste generation and high atom economy. By consolidating the synthesis into fewer steps with robust intermediates, the overall manufacturing cost is substantially decreased, allowing for more competitive pricing models in the global market for amino acid derivatives.
- Enhanced Supply Chain Reliability: The reliance on (S)-N-Boc-piperidine-2-carboxylic acid as a starting material leverages a stable supply chain, as this compound is derived from common chiral pool resources rather than bespoke synthetic precursors. The reagents employed, including methyl iodide, ruthenium oxide, and standard phosphonium salts, are widely available from multiple global vendors, mitigating the risk of single-source bottlenecks. Additionally, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or equipment limitations, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, utilizing solvents like acetonitrile, methanol, and ethyl acetate which are easily recovered and recycled in standard distillation trains. The absence of toxic heavy metals in stoichiometric quantities (using only catalytic ruthenium) simplifies wastewater treatment and aligns with increasingly stringent environmental regulations regarding heavy metal discharge. The process design facilitates a smooth transition from laboratory gram-scale to multi-ton production, supporting the growing demand for these intermediates in the synthesis of methionine precursors and other bioactive molecules without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational advantages over legacy methods. Understanding these nuances is essential for process chemists and technical buyers evaluating this technology for integration into their existing manufacturing portfolios.
Q: What are the limitations of traditional enzymatic splitting for amino suberates?
A: Traditional enzymatic kinetic resolution methods suffer from a theoretical maximum yield of only 50% for the desired enantiomer, making them economically inefficient for large-scale industrial production compared to the total synthesis route described in patent CN101168514B.
Q: How does this new method ensure high optical purity?
A: The process utilizes optically pure (S)-N-Boc-piperidine-2-carboxylic acid as the starting material from the chiral pool. Since the chirality is established at the beginning and the subsequent reactions (oxidation, reduction, Wittig) proceed with high stereoretention, the final product maintains excellent enantiomeric excess without requiring difficult chiral separations.
Q: Why is the RuO4/NaIO4 oxidation system preferred over other oxidants?
A: The ruthenium tetroxide system generated in situ allows for the selective oxidation of the carbon adjacent to the nitrogen atom under mild conditions (room temperature). This avoids the harsh conditions and potential racemization associated with other strong oxidants, ensuring the integrity of the chiral center while efficiently forming the key ketolactam intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-Amino Suberic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and compliant. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of (S)-2-amino suberic acid ester meets the exacting standards required for GMP pharmaceutical synthesis, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability, and let us support your supply chain with reliable, high-performance chemical solutions.
