Advanced Enzymatic Production of Icaritin for Pharmaceutical Applications
Advanced Enzymatic Production of Icaritin for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for producing high-value active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN107641621B, which details a novel glycosidase composition and an enzymatic method for preparing Icaritin from Icariin. This technology represents a paradigm shift from traditional chemical hydrolysis to a precise biocatalytic process, addressing long-standing challenges in yield, purity, and environmental compliance. By leveraging a specific dual-enzyme system, manufacturers can achieve a molar conversion rate of 98.3 percent, a figure that drastically outperforms conventional methods. For R&D directors and procurement specialists, this patent offers a tangible route to securing a reliable Icaritin supplier with a robust, scalable, and cost-effective manufacturing process that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Icaritin, a potent polyhydroxy flavonoid with significant therapeutic potential, has relied heavily on extraction from Epimedium plants or chemical hydrolysis of Icariin. The extraction route is inherently limited by the extremely low natural content of Icaritin in the raw plant material, making the separation process complex, labor-intensive, and prohibitively expensive for large-scale commercialization. Alternatively, chemical acid hydrolysis, while capable of cleaving glycosidic bonds, suffers from severe drawbacks including poor reaction control and the generation of a multitude of unwanted byproducts. These side reactions not only lower the overall yield but also create a complex impurity profile that necessitates rigorous and costly purification steps. Furthermore, the use of strong acids and organic solvents in traditional methods raises significant environmental and safety concerns, complicating waste disposal and regulatory compliance for pharmaceutical manufacturers seeking to minimize their ecological footprint.
The Novel Approach
In stark contrast to these legacy methods, the enzymatic approach disclosed in the patent utilizes a highly specific glycosidase composition to achieve precise bond cleavage without the harsh conditions associated with acid hydrolysis. This novel method employs a combination of alpha-L-rhamnosidase and a high-temperature resistant beta-glucosidase to sequentially remove sugar moieties from the Icariin backbone. The process operates under mild aqueous conditions, eliminating the need for toxic organic cosolvents and significantly reducing the formation of degradation byproducts. By optimizing the enzyme sources and reaction sequence, this technology ensures that the biological activity of the final Icaritin product is fully retained. For supply chain heads, this translates to a more predictable and stable production cycle, as the enzymatic process is less susceptible to the variability and safety hazards inherent in chemical synthesis, thereby enhancing the overall reliability of the Icaritin supply chain.
Mechanistic Insights into Dual-Enzyme Catalytic Hydrolysis
The core innovation of this technology lies in the synergistic action of two distinct enzymes: alpha-L-rhamnosidase and beta-glucosidase. The alpha-L-rhamnosidase, specifically derived from strains such as Aspergillus terreus CCF 3059, is responsible for the initial hydrolysis of the alpha-1,2-linked rhamnose residue on the Icariin molecule. This step is critical because the steric hindrance posed by the rhamnose group can inhibit the subsequent action of the beta-glucosidase. Following the removal of the rhamnose, the thermostable beta-glucosidase, often sourced from Thermotoga thermomarum, targets the beta-D-glucose bond. The patent highlights that the optimal reaction temperature for these enzymes is relatively high, with the beta-glucosidase functioning efficiently at up to 85 degrees Celsius. This thermal stability is a key mechanistic advantage, as higher temperatures improve the solubility of the flavonoid substrate in the aqueous buffer, thereby increasing the reaction kinetics and overall conversion efficiency without denaturing the biocatalyst.
Crucially, the order of enzyme addition is a deterministic factor in the success of this synthesis. Experimental data within the patent demonstrates that adding the alpha-L-rhamnosidase first, followed by the beta-glucosidase, results in a molar conversion rate of 98.3 percent. In contrast, simultaneous addition of both enzymes yields only 75 percent conversion, while reversing the order drops the efficiency to a mere 39 percent. This mechanistic insight underscores the importance of sequential catalysis in overcoming the structural constraints of the glycosidic bonds. By strictly controlling the reaction sequence and temperature profiles, manufacturers can minimize the presence of intermediate byproducts like Icariside I or II, ensuring a high-purity final product. This level of control is essential for meeting the stringent quality specifications required for pharmaceutical intermediates.
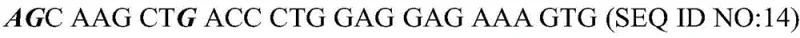
How to Synthesize Icaritin Efficiently
Implementing this enzymatic route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of a reaction system containing Icariin at a concentration of approximately 0.5 g/L in a citric acid-disodium hydrogen phosphate buffer maintained at pH 6.5. The first stage involves the addition of alpha-L-rhamnosidase at a concentration of 500 U/mL, with the reaction proceeding at 65 degrees Celsius for 8 hours. Once the rhamnose cleavage is complete, the temperature is elevated, and the beta-glucosidase is introduced at 1 U/mL to react at 85 degrees Celsius for 2 hours. This sequential thermal profile leverages the thermostability of the second enzyme to drive the reaction to completion. Following the enzymatic hydrolysis, the crude product is purified using macroporous resin chromatography, such as AB-8 type resin, to isolate the Icaritin with high purity. The detailed standardized synthesis steps are provided in the guide below.
- Prepare a reaction system with Icariin substrate in a citric acid-disodium hydrogen phosphate buffer at pH 6.5.
- Add alpha-L-rhamnosidase (derived from Aspergillus terreus) and react at 65°C for 8 hours to remove rhamnose residues.
- Subsequently add thermostable beta-glucosidase and react at 85°C for 2 hours to cleave glucose residues, followed by macroporous resin purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers substantial strategic advantages beyond mere technical feasibility. The primary benefit is the significant reduction in manufacturing costs driven by the elimination of expensive and hazardous chemical reagents. By replacing acid hydrolysis with a biocatalytic process, companies can avoid the costs associated with neutralizing large volumes of acidic waste and managing complex solvent recovery systems. The high conversion rate of 98.3 percent means that less raw material is wasted, directly improving the material yield and reducing the cost of goods sold. Furthermore, the simplified downstream processing, facilitated by the high specificity of the enzymes, reduces the burden on purification units, leading to faster batch cycles and lower operational expenditures. These factors combine to create a more cost-competitive product in the global market.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for transition metal catalysts and harsh acidic conditions, which are major cost drivers in traditional synthesis. By removing these expensive inputs and the associated waste treatment costs, the overall production expense is drastically simplified. The high yield ensures that the maximum amount of valuable Icariin feedstock is converted into the target Icaritin, minimizing raw material loss. Additionally, the ability to operate without organic cosolvents reduces solvent procurement and disposal costs, contributing to substantial cost savings in the long term.
- Enhanced Supply Chain Reliability: The use of thermostable enzymes derived from robust microbial sources ensures a consistent and reliable supply of biocatalysts. Unlike chemical reagents that may be subject to market volatility or regulatory restrictions, these enzymes can be produced via fermentation with high reproducibility. The process's tolerance to higher temperatures also reduces the risk of microbial contamination during production, leading to fewer batch failures and more consistent delivery schedules. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical partners who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system makes it inherently easier to scale from laboratory to commercial production without the safety risks associated with large-scale acid handling. The process generates significantly less hazardous waste, aligning with increasingly strict environmental regulations and corporate sustainability goals. The simplified purification process using macroporous resin is also easily scalable, allowing for the commercial scale-up of complex pharmaceutical intermediates without the need for specialized corrosion-resistant equipment. This facilitates a smoother path to regulatory approval and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic production of Icaritin. These answers are derived directly from the patent data and are intended to clarify the process advantages for potential partners. Understanding these details is key to evaluating the feasibility of integrating this technology into existing manufacturing workflows. The high specificity and yield of this method make it a superior choice for producing high-purity pharmaceutical intermediates.
Q: Why is the sequential addition of enzymes critical for Icaritin production?
A: Patent data indicates that adding alpha-L-rhamnosidase first followed by beta-glucosidase achieves a 98.3% molar conversion, whereas simultaneous addition drops efficiency to 75% due to steric hindrance and enzyme specificity conflicts.
Q: What are the advantages of using thermostable beta-glucosidase in this process?
A: The use of thermostable enzymes derived from Thermotoga thermomarum allows for reaction temperatures up to 85°C, which improves substrate solubility, reduces viscosity, and minimizes microbial contamination risks without needing organic cosolvents.
Q: How does this enzymatic method compare to traditional acid hydrolysis?
A: Unlike acid hydrolysis which generates numerous byproducts and requires harsh conditions, this enzymatic route offers high specificity, eliminates heavy metal catalysts, and simplifies downstream purification, resulting in a significantly cleaner impurity profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Icaritin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route for Icaritin and are well-positioned to support your supply chain needs. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Icaritin meets the highest industry standards. We understand the critical nature of API intermediates in the drug development timeline and are committed to delivering consistent quality and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you can access specific COA data and route feasibility assessments that will help you make informed decisions about your sourcing strategy. Contact us today to discuss how our advanced enzymatic capabilities can support your next generation of pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
