Advanced Synthesis of 2,4-Dibenzyloxybenzoic Acid Derivatives for Cardiovascular Drug Development
Advanced Synthesis of 2,4-Dibenzyloxybenzoic Acid Derivatives for Cardiovascular Drug Development
The pharmaceutical landscape for treating cardiovascular and cerebrovascular diseases is constantly evolving, driven by the urgent need for more effective therapeutics with improved pharmacokinetic profiles. Patent CN103539659A introduces a significant breakthrough in this domain by disclosing a novel series of 2,4-dibenzyloxybenzoic acid derivatives and their pharmaceutically acceptable salts. These compounds are engineered to act as potent endothelin receptor antagonists, addressing the critical limitations of earlier peptide-based therapies which often suffered from poor oral bioavailability. The structural versatility of these derivatives, characterized by variable substituents at the R1, R2, and Ar positions, allows for fine-tuning of biological activity against endothelin-1 induced vasoconstriction. As a leading entity in fine chemical manufacturing, we recognize the immense potential of this chemical space for developing next-generation medicines for heart failure, hypertension, and pulmonary arterial hypertension.
The core innovation lies in the specific arrangement of the dibenzyloxy scaffold which mimics the binding affinity of natural ligands while maintaining metabolic stability. The patent details eighteen specific embodiments ranging from acetyl and carboxy variants to cyano-substituted analogues, each demonstrating varying degrees of inhibitory activity against ET-1 induced contraction in rat thoracic aortic rings. For R&D directors evaluating new pipeline candidates, the data indicates that specific heterocyclic substitutions, such as benzoxadiazole and benzothiadiazole moieties, significantly enhance antagonistic potency. This suggests a clear structure-activity relationship that can be exploited for further medicinal chemistry optimization, making these intermediates highly valuable assets for drug discovery programs targeting the endothelin pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of endothelin antagonists was dominated by peptide-based molecules such as BQ123 and BQ160. While these peptides exhibited strong binding affinity in vitro, their clinical utility was severely hampered by low oral bioavailability and rapid metabolic clearance, necessitating parenteral administration which is inconvenient for chronic disease management. Furthermore, the synthesis of complex peptide mimetics often involves cumbersome protection and deprotection strategies, leading to lower overall yields and higher production costs. The reliance on amino acid building blocks also introduces supply chain vulnerabilities and batch-to-batch variability in stereochemical purity. Consequently, there has been a persistent industry demand for small molecule, non-peptide alternatives that can offer the same therapeutic efficacy with the convenience of oral dosing and the economic advantages of scalable chemical synthesis.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift towards robust, small-molecule architecture that circumvents the pitfalls of peptide chemistry. By utilizing a 2,4-dihydroxybenzene core coupled with diverse benzyl chloride derivatives, the inventors have created a modular synthetic platform. This approach allows for the rapid generation of a library of analogues by simply varying the aromatic substituents without altering the fundamental reaction conditions. The use of stable intermediates like gamma-phenyl-gamma-chlorobutyric acid esters ensures that the side chain installation is efficient and reproducible. Moreover, the final hydrolysis steps are performed under standard acidic or alkaline conditions, avoiding the need for exotic reagents or extreme temperatures that could degrade sensitive functional groups. This streamlined workflow not only accelerates the lead optimization process but also lays a solid foundation for cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations.
Mechanistic Insights into Etherification and Nucleophilic Substitution
The synthetic strategy relies heavily on classic Williamson ether synthesis mechanics, optimized for high regioselectivity and yield. In the initial step, the phenolic hydroxyl groups of the starting materials (such as 2,4-dihydroxyacetophenone or methyl 2,4-dihydroxybenzoate) act as nucleophiles. In the presence of a base like potassium carbonate and a phase transfer catalyst such as tetrabutylammonium bromide (TBAB), these phenols attack the electrophilic carbon of the substituted benzyl chlorides. The choice of solvent, typically acetone or DMF, is critical to solubilize both the inorganic base and the organic substrates, facilitating the SN2 displacement of the chloride ion. The patent data reveals that controlling the reaction temperature between 55°C and 65°C is essential to prevent over-alkylation or decomposition of the sensitive aldehyde or ketone functionalities present on the ring.
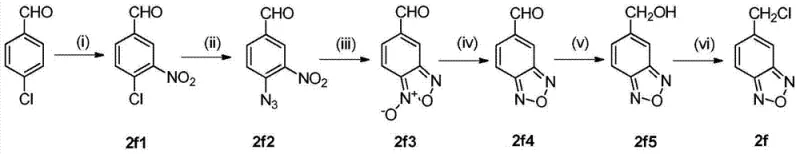
Following the formation of the ether linkage, the second critical transformation involves the coupling of this intermediate with the gamma-phenyl side chain. This step is essentially another nucleophilic substitution where the remaining phenolic oxygen attacks the gamma-carbon of the chlorobutyrate or chlorobutyronitrile derivative. The mechanistic elegance here is the tolerance of various functional groups; for instance, the synthesis of complex heterocyclic substituents like 5-chloromethyl-2,1,3-benzoxadiazole involves a multi-step sequence including nitration, azidation, and thermal cyclization. As illustrated in the reaction scheme, the conversion of p-chlorobenzaldehyde to the final benzoxadiazole chloride requires precise control of reaction conditions to manage the evolution of nitrogen gas during the cyclization step. Understanding these mechanistic nuances is vital for scaling up the process safely, as it highlights potential exotherms and gas evolution that must be managed in a commercial reactor setting to ensure operator safety and product consistency.
How to Synthesize 2,4-Dibenzyloxybenzoic Acid Derivatives Efficiently
Executing the synthesis of these high-value intermediates requires a disciplined approach to reaction monitoring and purification to meet the stringent purity specifications demanded by the pharmaceutical industry. The process generally begins with the preparation of the specialized benzyl chloride building blocks, which may involve reduction of aldehydes followed by chlorination with thionyl chloride, or more complex heterocycle formation as seen with the benzothiadiazole derivatives. Once the electrophiles are secured, the etherification with the dihydroxy core is conducted under reflux, followed by isolation via crystallization or extraction. The subsequent coupling with the gamma-phenyl side chain precursor extends the molecular framework, and the final step involves hydrolysis of the ester or nitrile groups to reveal the active carboxylic acid or maintain the cyano functionality depending on the target analogue. Detailed standardized synthetic steps see the guide below.
- Prepare substituted benzyl chlorides via reduction and chlorination of corresponding aromatic aldehydes or through nitration and cyclization of toluidine derivatives.
- Perform etherification by reacting 2,4-dihydroxy precursors (acetophenone, benzoate, or benzonitrile) with substituted benzyl chlorides using potassium carbonate and phase transfer catalysts.
- Couple the intermediate ether with gamma-phenyl-gamma-chlorobutyric acid esters or nitriles, followed by alkaline hydrolysis to yield the final carboxylic acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from peptide-based to small-molecule endothelin antagonists offers profound strategic benefits beyond mere chemical novelty. The synthetic routes described utilize commodity feedstocks such as benzene, toluene, and simple aromatic aldehydes, which are produced on a massive global scale, ensuring a stable and resilient supply chain不受 geopolitical fluctuations affecting specialized amino acids. The elimination of expensive transition metal catalysts in favor of basic inorganic reagents like potassium carbonate and sodium borohydride drastically simplifies the waste stream profile, reducing the environmental burden and the associated costs of hazardous waste disposal. This aligns perfectly with modern green chemistry initiatives and helps pharmaceutical partners meet their sustainability goals without compromising on production efficiency or output quality.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to maximize atom economy and minimize the number of isolation steps. High yields reported in the patent examples, often exceeding 80% for key transformations like the reduction of aldehydes or the final hydrolysis, indicate that raw material consumption is optimized. By avoiding the use of precious metal catalysts which require costly removal and recovery processes, the overall cost of goods sold (COGS) is significantly lowered. Furthermore, the ability to perform reactions at moderate temperatures (50°C to 90°C) reduces energy consumption compared to cryogenic or high-pressure processes, contributing to substantial cost savings in large-scale production facilities.
- Enhanced Supply Chain Reliability: The reliance on widely available aromatic starting materials mitigates the risk of supply disruptions. Unlike complex chiral pools required for peptide synthesis, the achiral nature of most intermediates in this series simplifies sourcing and quality control. The robustness of the chemistry means that multiple qualified suppliers can potentially manufacture the key intermediates, fostering a competitive sourcing environment that protects against single-source bottlenecks. This redundancy is crucial for maintaining continuous manufacturing schedules for life-saving cardiovascular medications, ensuring that patients have uninterrupted access to therapy.
- Scalability and Environmental Compliance: The processes described are inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production. The use of common solvents like ethanol, acetone, and ethyl acetate facilitates solvent recovery and recycling, further enhancing the economic and environmental viability of the process. The absence of heavy metal residues simplifies the regulatory filing process, as extensive testing for metal impurities is not required, accelerating the time to market for new drug applications. This operational simplicity translates to faster technology transfer and quicker ramp-up times for contract development and manufacturing organizations (CDMOs).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 2,4-dibenzyloxybenzoic acid derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these aspects is critical for assessing the feasibility of integrating these intermediates into your existing drug development pipelines or supply networks.
Q: What is the primary therapeutic application of these 2,4-dibenzyloxybenzoic acid derivatives?
A: These compounds function as potent non-peptide endothelin receptor antagonists, specifically designed for treating cardiovascular and cerebrovascular diseases, tumors, diabetes, nephrosis, and asthma, offering superior oral bioavailability compared to peptide alternatives.
Q: How does the synthesis route ensure high purity for pharmaceutical use?
A: The patented process utilizes robust purification techniques such as recrystallization from ethanol or methanol and flash column chromatography at critical intermediate stages, ensuring that impurities from nitration or halogenation steps are effectively removed before final coupling.
Q: Are the starting materials for these derivatives commercially scalable?
A: Yes, the synthesis relies on widely available commodity chemicals such as benzaldehyde, toluene derivatives, and succinic anhydride, which facilitates reliable supply chain continuity and cost-effective commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dibenzyloxybenzoic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for these derivatives, including precise temperature control for exothermic nitration steps and efficient solvent recovery systems for etherification processes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest standards of quality, safety, and consistency required by global regulatory bodies. Our commitment to excellence makes us the ideal partner for bringing these promising cardiovascular therapeutics to the market.
We invite you to initiate a dialogue with our technical procurement team to discuss how we can support your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. We encourage you to contact us to obtain specific COA data for our available catalog items and to request comprehensive route feasibility assessments for your custom synthesis projects. Let us collaborate to accelerate the development of life-saving medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
