Industrial Scale Resolution of L-(+)-Ephedrine Using L-(-)-Dibenzoyltartaric Acid Derivatives
Industrial Scale Resolution of L-(+)-Ephedrine Using L-(-)-Dibenzoyltartaric Acid Derivatives
The global demand for optically pure pharmaceutical intermediates continues to drive innovation in chiral separation technologies, particularly for high-volume compounds like ephedrine. Patent CN100391932C introduces a robust and industrially viable method for resolving DL-ephedrine using L-(-)-tartaric acid derivatives, specifically L-(-)-dibenzoyltartaric acid. This technology addresses critical environmental and economic challenges associated with the traditional extraction of ephedrine from natural sources, which has led to severe ecological degradation in producing regions. By shifting towards a synthetic resolution pathway, manufacturers can secure a stable supply chain while mitigating the environmental impact of over-harvesting Ephedra plants. The core innovation lies in the selection of a resolving agent that balances high stereoselectivity with economic feasibility, enabling the production of (1S, 2R)-L-(+)-ephedrine with exceptional purity.
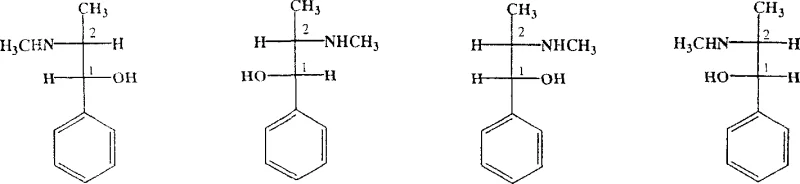
Ephedrine possesses two chiral carbon atoms, resulting in four distinct stereoisomers: D-(-)-pseudoephedrine, L-(+)-pseudoephedrine, D-(-)-ephedrine, and L-(+)-ephedrine. The ability to selectively isolate the L-(+)-ephedrine isomer is paramount for pharmaceutical applications where specific biological activity is required. The patent outlines a comprehensive strategy that leverages the formation of diastereomeric salts to exploit solubility differences, a fundamental principle in classical resolution that has been optimized here for modern manufacturing standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of DL-ephedrine has been attempted using various chiral acids, each presenting significant drawbacks that hinder large-scale industrial adoption. For instance, the use of L-(+)-mandelic acid and D-(-)-mandelic acid, while offering high resolution efficiency and optical purity, is severely limited by the high cost and scarcity of these resolving agents. Similarly, attempts to utilize natural L-(+)-tartaric acid have met with mixed success, with literature indicating that this method often fails to effectively resolve DL-ephedrine derivatives, rendering it unreliable for consistent production. Other agents like D-arabinic acid provide good resolution ratios but are prohibitively expensive and difficult to procure in the quantities necessary for ton-scale manufacturing. These limitations create a bottleneck for pharmaceutical companies seeking cost-effective and scalable routes to optically pure ephedrine.
The Novel Approach
The novel approach detailed in the patent utilizes L-(-)-dibenzoyltartaric acid or its derivatives as the resolving agent, marking a significant departure from previous methodologies. This specific chiral acid demonstrates excellent resolving power, achieving a splitting yield of over 80%, which is superior to many conventional alternatives. The process is designed to be simple and practical, utilizing common solvents such as methanol and water, which simplifies the operational complexity and reduces safety hazards associated with exotic organic solvents. Furthermore, the method incorporates a built-in recovery mechanism for the resolving agent, allowing it to be recycled with high efficiency. This closed-loop system not only lowers the variable cost per kilogram of product but also aligns with green chemistry principles by minimizing chemical waste.
Mechanistic Insights into Diastereomeric Salt Formation
The resolution mechanism relies on the interaction between the racemic DL-ephedrine and the chiral L-(-)-dibenzoyltartaric acid to form diastereomeric salts. When these components are mixed in a specific molar ratio, typically ranging from 1:0.1 to 1:1.2, they form two distinct salts: one involving the L-(+)-ephedrine enantiomer and the other involving the D-(-)-ephedrine enantiomer. Due to the three-dimensional arrangement of atoms, these two salts possess different physical properties, most notably their solubility in the chosen solvent system. The L-(+)-ephedrine L-(-)-dibenzoyl tartrate salt exhibits significantly lower solubility in the methanol-water mixture at reduced temperatures, causing it to precipitate out of the solution as crystals while the unwanted D-(-)-isomer remains dissolved in the mother liquor.
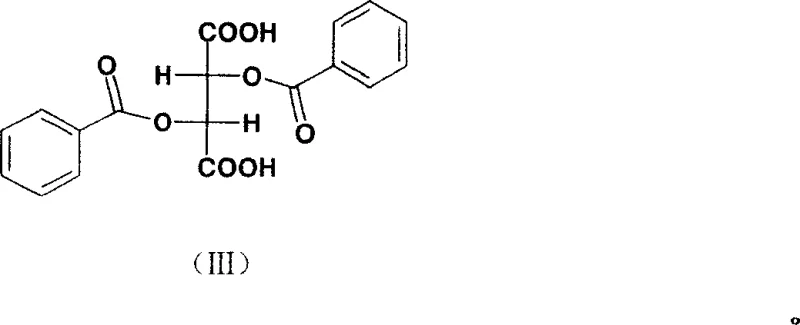
The structural integrity of the resolving agent is crucial for this selectivity. The L-(-)-dibenzoyltartaric acid molecule, characterized by its benzoyl groups attached to the tartaric acid backbone, provides the necessary steric bulk and electronic environment to differentiate between the ephedrine enantiomers. The presence of substituents such as H, CH3, Cl, F, or NO2 on the benzoyl rings can further tune the resolving power, although the unsubstituted parent compound is particularly preferred for its balance of performance and cost. The crystallization process is carefully controlled by temperature, with dissolution occurring at 50-70°C followed by slow cooling to room temperature to ensure the formation of high-quality crystals with minimal occlusion of the unwanted isomer.
How to Synthesize L-(+)-Ephedrine Efficiently
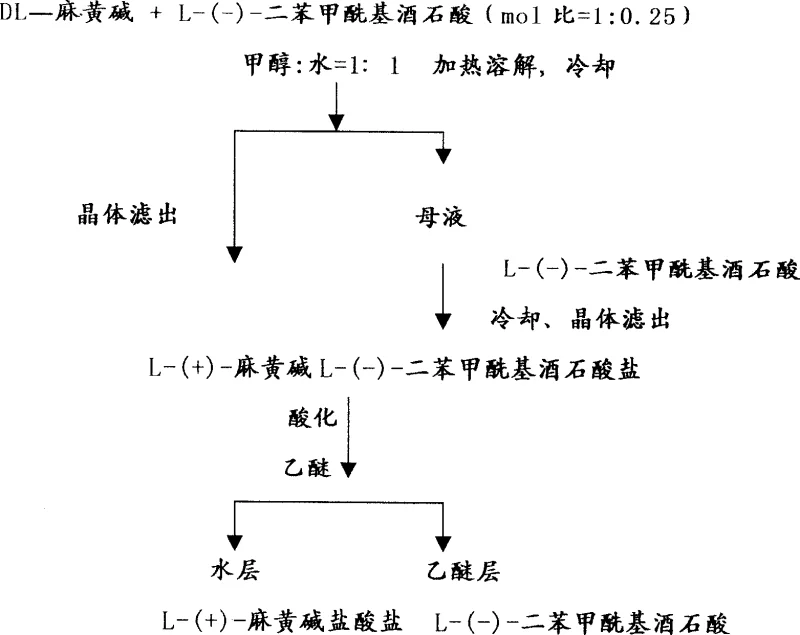
The synthesis protocol described in the patent offers a streamlined pathway for converting racemic ephedrine into its optically active form. The process begins with the preparation of the free base from DL-ephedrine hydrochloride, followed by the critical resolution step where the chiral acid is introduced. The operational parameters, including solvent ratios, temperature gradients, and stirring times, are optimized to maximize yield and purity. This section outlines the general workflow, emphasizing the importance of precise stoichiometry and thermal control to achieve the reported >80% yield. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation below.
- Mix DL-ephedrine and L-(-)-dibenzoyltartaric acid in a molar ratio of 1: 0.25 in methanol, add water at 60-70°C, and cool to crystallize the diastereomeric salt.
- Filter the crystals, wash with acetone, and treat the mother liquor with additional resolving agent to recover more product.
- Acidify the combined salts with dilute hydrochloric acid, extract the resolving agent with ether, and recrystallize the aqueous layer to obtain L-(+)-ephedrine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the significant reduction of raw material costs through the recyclability of the resolving agent. In traditional resolution processes where the chiral acid is discarded or difficult to recover, the cost of the auxiliary reagent can constitute a major portion of the total production cost. By implementing a recovery step that distills the organic layer to reclaim the L-(-)-dibenzoyltartaric acid, manufacturers can drastically reduce the net consumption of this valuable chiral pool material. This efficiency translates directly into improved margins and a more competitive pricing structure for the final API intermediate.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of expensive, single-use resolving agents like D-arabinic acid or mandelic acid. The ability to recover the resolving agent with a reported efficiency of over 96% means that the initial investment in the chiral acid is amortized over multiple production batches. Furthermore, the use of commodity solvents such as methanol, ethanol, and diethyl ether avoids the premium pricing associated with specialized chlorinated or aromatic solvents, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral acids can often be a bottleneck in the supply chain, especially for niche resolving agents. L-(-)-dibenzoyltartaric acid is a well-established chemical with a robust global supply network, ensuring consistent availability and reducing the risk of production stoppages due to raw material shortages. The simplicity of the process, which relies on standard unit operations like crystallization, filtration, and distillation, also means that it can be easily transferred between different manufacturing sites or scaled up without requiring specialized equipment, thereby enhancing supply chain flexibility and resilience.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-ton industrial production. The reliance on crystallization as the primary separation technique is advantageous for scale-up, as it is a well-understood and controllable unit operation in the chemical industry. Additionally, the recovery of the resolving agent minimizes the generation of hazardous chemical waste, simplifying wastewater treatment and helping facilities meet stringent environmental regulations. This alignment with green chemistry principles reduces the regulatory burden and potential liability associated with waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the resolution of ephedrine using L-(-)-dibenzoyltartaric acid. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What is the primary advantage of using L-(-)-dibenzoyltartaric acid over mandelic acid for ephedrine resolution?
A: Unlike mandelic acid, which is expensive and difficult to source in large quantities, L-(-)-dibenzoyltartaric acid offers a cost-effective solution with high resolution efficiency. Furthermore, the dibenzoyltartaric acid can be recovered and recycled from the organic layer after acidification, significantly reducing raw material costs compared to non-recyclable agents.
Q: Can the resolving agent be reused in this process?
A: Yes, the process includes a specific recovery step where the organic layer containing the resolving agent is distilled to remove solvents. The residue is the recovered L-(-)-dibenzoyltartaric acid, which maintains its optical purity and can be reused in subsequent batches, enhancing the overall sustainability of the manufacturing process.
Q: What is the expected yield for this resolution method?
A: According to the patent data, the resolution yield for obtaining L-(+)-ephedrine hydrochloride can reach over 80%. Additionally, the recovery rate of the resolving agent itself is reported to be as high as 96.3%, ensuring minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-(+)-Ephedrine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral resolution in the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced resolution technologies like the one described in CN100391932C, we can offer our partners a reliable source of optically pure ephedrine derivatives that comply with global regulatory standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral chemistry can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
