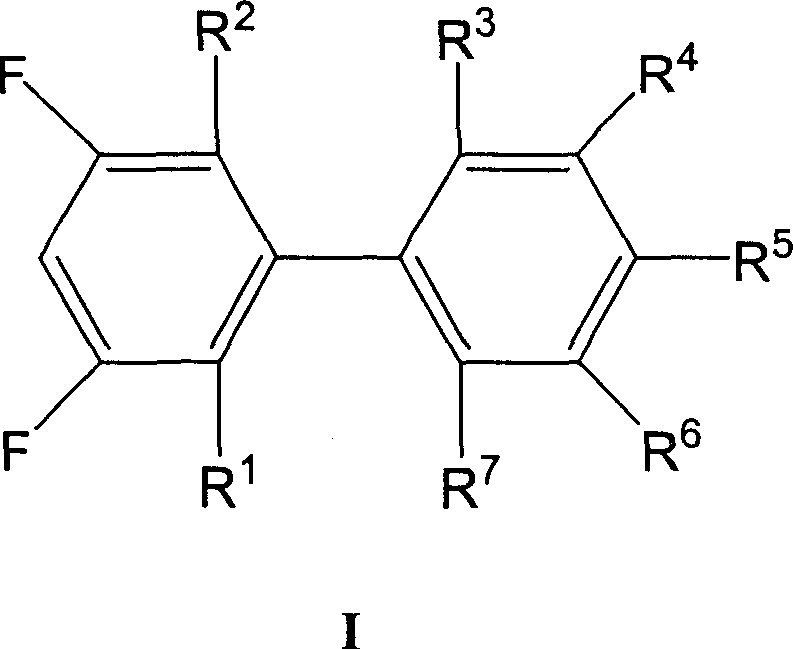
Advanced Manufacturing of 3,5-Difluorobiphenyl Derivatives: A Cost-Efficient Suzuki Coupling Strategy for Global Supply Chains
Advanced Manufacturing of 3,5-Difluorobiphenyl Derivatives: A Cost-Efficient Suzuki Coupling Strategy for Global Supply Chains
The chemical industry is constantly seeking more efficient pathways to produce high-value intermediates, particularly for the demanding electronics sector. Patent CN1903813A introduces a groundbreaking preparation method for 3,5-difluorobiphenyl derivatives, which serve as critical building blocks for advanced liquid crystal materials. These derivatives are essential for producing liquid crystals that exhibit superior viscosity coefficients, dielectric properties, and thermal stability, making them indispensable for next-generation display technologies. The core innovation lies in a refined Suzuki-Miyaura cross-coupling reaction that strategically reverses the traditional reactant roles to optimize cost and efficiency. By utilizing difluorohalogenobenzenes and phenylboronic acids under specific catalytic conditions, this method addresses the economic bottlenecks of previous synthetic routes. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable liquid crystal intermediate supplier capable of delivering high-purity materials at competitive price points.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-difluorobiphenyl derivatives has relied heavily on methods that are economically inefficient and chemically cumbersome. A prominent example found in the prior art, specifically U.S. Pat. No. 6,200,654, utilizes 3,5-difluorophenylboronic acid as a key starting material. As illustrated in the reaction scheme below, this approach necessitates the use of highly functionalized and expensive boronic acid reagents, which significantly inflates the raw material costs for large-scale production. Furthermore, conventional methods often require substantial amounts of palladium catalysts to drive the reaction to completion, leading to higher operational expenses and complicating the downstream purification processes due to heavy metal residues. The reliance on such costly precursors creates a fragile supply chain vulnerable to price volatility in the fine chemical market, posing a significant risk for manufacturers aiming for cost reduction in display material manufacturing.
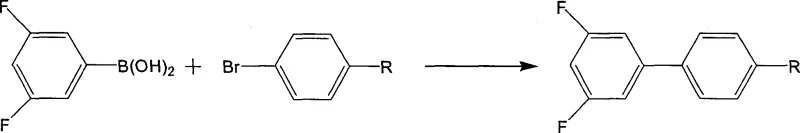
The Novel Approach
In stark contrast to the legacy techniques, the method disclosed in CN1903813A ingeniously flips the synthetic logic by employing 3,5-difluorohalobenzene as the electrophilic partner and a substituted phenylboronic acid as the nucleophile. This strategic substitution allows manufacturers to leverage 3,5-difluorohalobenzenes, which are commodity chemicals available at a fraction of the cost of their boronic acid counterparts. The novel approach not only slashes raw material expenditures but also operates with remarkably low catalyst loading, typically ranging from 0.06% to 0.1% molar ratio relative to the substrate. This drastic reduction in catalyst usage translates directly into lower production costs and simplifies the removal of metal impurities, ensuring the final product meets the rigorous purity specifications required for electronic applications. By optimizing the reaction conditions with a specific solvent system and base, this method achieves high yields while maintaining a robust and scalable process suitable for industrial implementation.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The heart of this synthesis lies in the palladium-catalyzed cross-coupling mechanism, which facilitates the formation of the carbon-carbon bond between the two aromatic rings. The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the 3,5-difluorohalobenzene, forming an organopalladium(II) intermediate. This step is crucial and is facilitated by the electron-withdrawing nature of the fluorine atoms, which activates the aryl halide towards oxidative addition. Subsequently, the phenylboronic acid, activated by the base (preferably potassium carbonate), undergoes transmetallation with the palladium complex. This transfers the aryl group from the boron atom to the palladium center, creating a diarylpalladium species. Finally, reductive elimination occurs, releasing the desired 3,5-difluorobiphenyl derivative and regenerating the active palladium(0) catalyst to continue the cycle. The efficiency of this cycle is paramount for maintaining the low catalyst loading observed in the patent, ensuring that every atom of the precious metal is utilized effectively.
Controlling impurities in such complex coupling reactions is a major concern for R&D directors focused on product quality. The choice of solvent system plays a pivotal role in managing side reactions and ensuring high selectivity. The patent specifies a mixed solvent system of toluene, water, and ethanol, which creates a biphasic environment conducive to the reaction while solubilizing both organic substrates and inorganic bases. The presence of water is essential for the hydrolysis of the boronic acid species, facilitating the transmetallation step, while the organic solvents ensure the solubility of the hydrophobic biphenyl products. Furthermore, conducting the reaction under an inert nitrogen atmosphere prevents the oxidation of the sensitive palladium catalyst and the boronic acid reagents, thereby minimizing the formation of homocoupling byproducts and phenolic impurities. This meticulous control over the reaction environment results in the reported 99.4% purity, demonstrating the method's capability to produce high-purity 3,5-difluorobiphenyl derivatives suitable for sensitive optoelectronic applications.
How to Synthesize 3,5-Difluorobiphenyl Derivatives Efficiently
The practical implementation of this synthesis route requires precise adherence to the optimized conditions detailed in the patent to maximize yield and purity. The process is designed to be operationally simple yet chemically robust, making it ideal for transfer from laboratory scale to commercial production. Operators must carefully manage the stoichiometry of the reagents, particularly the molar ratio of the base to the substrate, which is optimally maintained between 2:1 and 3:1 to ensure complete activation of the boronic acid without causing excessive hydrolysis. The following guide outlines the standardized procedure derived from the patent examples, providing a clear roadmap for technical teams looking to adopt this methodology.
- Prepare the reaction vessel under an inert nitrogen atmosphere and charge it with 3,5-difluorohalobenzene, substituted phenylboronic acid, potassium carbonate base, and a mixed solvent system of toluene, water, and ethanol.
- Add the palladium catalyst, specifically tetrakis(triphenylphosphine)palladium(0), at a molar ratio of approximately 0.06% to 0.1% relative to the halobenzene substrate to initiate the catalytic cycle.
- Heat the reaction mixture to reflux temperature for approximately 10 hours, then perform phase separation, extract the aqueous layer with toluene, and purify the organic layer via distillation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method offers transformative benefits that extend far beyond simple chemical efficiency. The primary driver for value creation is the fundamental shift in raw material sourcing, moving away from specialized, high-cost boronic acids toward widely available halobenzenes. This change inherently stabilizes the supply chain against the volatility often seen in the market for specialized organoboron compounds. Additionally, the drastic reduction in catalyst loading means that the consumption of expensive palladium resources is minimized, leading to substantial cost savings in the bill of materials. These factors combined create a manufacturing process that is not only cheaper to run but also more resilient to supply disruptions, ensuring a consistent flow of critical intermediates for downstream liquid crystal production.
- Cost Reduction in Manufacturing: The economic impact of switching to 3,5-difluorohalobenzene as the starting material cannot be overstated, as it eliminates the premium pricing associated with pre-functionalized boronic acids. By utilizing commodity halides, the direct material costs are significantly reduced, allowing for more competitive pricing strategies in the final electronic chemical market. Furthermore, the ultra-low catalyst loading of less than 0.1% drastically cuts down on the cost of precious metals, which is a major expense in traditional Suzuki couplings. This efficiency also reduces the burden on waste treatment facilities, as there is less heavy metal waste to process, contributing to overall operational expenditure reductions without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Sourcing 3,5-difluorohalobenzenes is inherently more reliable than sourcing complex boronic acids, as halobenzenes are produced on a much larger industrial scale by multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and ensures that production schedules are not held hostage by the availability of niche reagents. The robustness of the reaction conditions, which tolerate a variety of functional groups and utilize common solvents like toluene and ethanol, further enhances reliability by minimizing the risk of batch failures due to reagent sensitivity. Consequently, supply chain managers can forecast lead times with greater accuracy and maintain healthier inventory levels of critical liquid crystal intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reflux conditions and standard extraction workups that are easily adaptable to large-scale reactors ranging from pilot plants to multi-ton production units. The use of a toluene-water-ethanol solvent system allows for efficient phase separation and solvent recovery, aligning with modern green chemistry principles and environmental regulations. By minimizing the use of exotic reagents and reducing catalyst waste, the process lowers the environmental footprint of the manufacturing operation. This compliance with environmental standards is increasingly important for multinational corporations seeking to meet sustainability goals while scaling up the commercial scale-up of complex electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 3,5-difluorobiphenyl derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for qualifying new suppliers for critical projects.
Q: What is the primary cost advantage of this new synthesis method compared to prior art?
A: The primary cost advantage lies in the reversal of coupling partners; instead of using expensive 3,5-difluorophenylboronic acid as seen in US Patent 6,200,654, this method utilizes significantly cheaper 3,5-difluorohalobenzene precursors, drastically reducing raw material expenditures.
Q: How does the catalyst loading in this process compare to conventional Suzuki reactions?
A: This process employs an ultra-low catalyst loading of merely 0.06% to 0.1% molar ratio, which is substantially lower than typical industrial standards, thereby minimizing precious metal residue and reducing the cost associated with palladium catalyst consumption.
Q: What purity levels can be achieved with this manufacturing protocol?
A: Experimental data from the patent indicates that the process yields products with exceptional purity, specifically achieving 99.4% purity after fractional distillation, which meets the stringent quality requirements for high-performance liquid crystal applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Difluorobiphenyl Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the fast-evolving landscape of electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement the optimized Suzuki coupling methodology described in CN1903813A allows us to offer a reliable 3,5-difluorobiphenyl derivative supplier partnership that balances cost, quality, and delivery performance.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to our optimized manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your production of high-performance liquid crystals remains competitive and uninterrupted.
