Revolutionizing Electronic Chemical Intermediates: Scalable One-Step Production of p-Hydroxystyrene
The semiconductor and flexible electronics industries are currently witnessing a paradigm shift in the sourcing of critical monomers, driven by the urgent need for cost-effective and scalable synthetic routes. A pivotal development in this sector is documented in patent CN109553511B, which discloses a groundbreaking method for preparing p-hydroxystyrene through a one-step coupling of hydrogenation and dehydration reactions. This technology addresses the longstanding bottlenecks in producing poly-p-hydroxystyrene, the backbone material for chemically amplified photoresists used in integrated circuit manufacturing. By transforming p-hydroxyacetophenone directly into the target vinyl monomer under alkaline conditions, this innovation eliminates the need for complex multi-step sequences and expensive transition metal catalysts. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-performance electronic chemicals, ensuring both purity and economic viability in a competitive market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-hydroxystyrene has been plagued by inefficient methodologies that hinder large-scale commercial adoption. Traditional routes often rely on the Wittig reaction involving p-hydroxybenzaldehyde, which necessitates the use of costly phosphorus ylide reagents and generates substantial amounts of phosphine oxide waste, creating severe environmental and disposal challenges. Alternative pathways, such as the deamination of 4-(1-aminoethyl)phenol or the dehydrohalogenation of halogenated benzene derivatives, suffer from similar drawbacks including high raw material costs and the generation of hazardous halogenated byproducts. Furthermore, existing methods frequently require harsh conditions, such as high-temperature dehydrogenation with phenol additives, which increase energy consumption and complicate reactor safety protocols. These legacy processes result in fragmented supply chains where yield losses accumulate at each isolation step, ultimately driving up the cost per kilogram for the final photoresist monomer.
The Novel Approach
In stark contrast, the methodology outlined in CN109553511B introduces a streamlined, one-pot strategy that fundamentally reimagines the synthetic logic. By utilizing p-hydroxyacetophenone—a readily available and inexpensive commodity chemical—as the sole substrate, the process achieves a direct transformation via a base-catalyzed mechanism. This approach couples the reduction of the ketone group with the subsequent dehydration of the resulting alcohol intermediate in a single reaction vessel, effectively bypassing the need for isolated intermediate steps. The use of common industrial alkalis such as potassium hydroxide or sodium oxide replaces precious metal catalysts, significantly lowering the barrier to entry for manufacturing. This consolidation of reaction steps not only simplifies the operational workflow but also enhances the overall atom economy, providing a robust foundation for cost reduction in electronic chemical manufacturing without compromising on the structural integrity of the final product.
Mechanistic Insights into Base-Catalyzed Coupling Hydrogenation and Dehydration
The core scientific breakthrough of this patent lies in the elucidation of a unique reaction pathway where a base catalyst facilitates both reduction and elimination sequentially. As illustrated in the reaction scheme below, the process initiates with the nucleophilic attack or hydride transfer mediated by the alcohol solvent and base, converting the carbonyl group of p-hydroxyacetophenone into a secondary alcohol intermediate. This intermediate then undergoes base-promoted elimination of water to form a quinone methide species, which subsequently tautomerizes or rearranges to yield the stable vinyl group of p-hydroxystyrene. This mechanistic understanding is crucial for R&D teams aiming to optimize reaction parameters, as it highlights the delicate balance required between the basicity of the catalyst and the temperature to prevent polymerization of the reactive styrene monomer during synthesis.
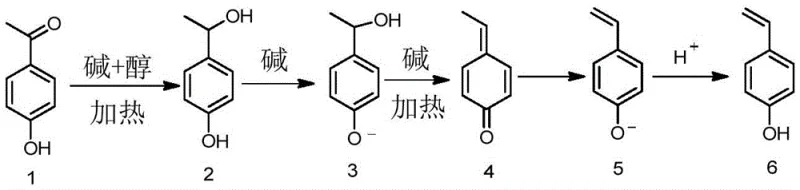
Furthermore, the control of impurity profiles is intrinsically linked to this specific mechanistic pathway. In traditional acid-catalyzed dehydrations, the presence of strong protons can initiate cationic polymerization of the styrene double bond, leading to oligomeric impurities that are difficult to remove and detrimental to photoresist performance. The alkaline environment of this novel method suppresses cationic initiation, thereby preserving the monomeric state of the product. Additionally, the choice of anhydrous alcohol solvents plays a critical role in stabilizing the transition states and preventing side reactions such as ether formation. By maintaining strict control over the water content and base concentration, manufacturers can achieve conversion rates exceeding 95%, ensuring that the crude product requires minimal downstream purification to meet the stringent specifications demanded by the semiconductor industry.
How to Synthesize p-Hydroxystyrene Efficiently
To implement this technology effectively, process engineers must adhere to precise operational parameters regarding solvent ratios and thermal profiles. The patent specifies a mass-to-volume ratio of substrate to alcohol between 0.01 and 0.2 kg/L, ensuring optimal solubility and heat transfer throughout the reaction mass. Detailed standardized synthesis steps, including specific workup procedures and purification protocols, are essential for replicating the high yields reported in the experimental data.
- Dissolve p-hydroxyacetophenone in anhydrous alcohol solvent and add industrial alkali catalyst such as potassium hydroxide.
- Heat the reaction mixture to 150-200°C under closed conditions for 0.2 to 12 hours to facilitate coupling hydrogenation and dehydration.
- Filter solids, neutralize filtrate with dilute acid, and purify the crude product via chromatography to obtain high-purity p-hydroxystyrene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond simple yield improvements. The shift from specialized, high-cost reagents to commodity chemicals fundamentally alters the cost structure of the final product. By eliminating the dependency on imported phosphorus reagents or rare metal catalysts, manufacturers can insulate their production costs from volatile global markets. This stability is critical for long-term contracting with semiconductor fabs, where price consistency is often as valued as technical performance. Moreover, the simplification of the process flow reduces the capital expenditure required for plant infrastructure, as fewer reactors and separation units are needed to achieve the same output volume.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of expensive transition metal catalysts and phosphorus-based reagents from the bill of materials. Instead, the process relies on industrial-grade alkalis like sodium hydroxide or potassium hydroxide, which are available globally at a fraction of the cost. This substitution drastically reduces the raw material expense per batch. Additionally, the one-step nature of the reaction minimizes solvent usage and energy consumption associated with heating and cooling cycles between multiple stages. The cumulative effect is a substantial decrease in the cost of goods sold (COGS), allowing for more competitive pricing strategies in the high-margin electronic chemicals sector.
- Enhanced Supply Chain Reliability: Sourcing risks are significantly mitigated by the use of universally available starting materials. p-Hydroxyacetophenone is a bulk chemical produced by numerous suppliers worldwide, ensuring that production is never halted due to the shortage of a niche precursor. Unlike complex chiral catalysts or custom-synthesized ylides that may have long lead times, the reagents for this process can be procured on short notice. This resilience ensures continuous supply continuity for downstream photoresist manufacturers, reducing the risk of production stoppages that could ripple through the entire semiconductor supply chain.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method offers a cleaner profile that simplifies regulatory compliance. The absence of heavy metals means there is no need for expensive and complex metal scavenging steps or the disposal of hazardous metal-contaminated waste streams. The waste generated is primarily saline and organic solvent, which can be treated using standard industrial wastewater protocols. This environmental friendliness facilitates faster permitting for new production lines and aligns with the increasing sustainability mandates of major technology corporations, making the supply chain more robust against future regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for feasibility assessments.
Q: What are the primary advantages of this one-step method over traditional Wittig reactions?
A: Unlike traditional Wittig reactions which require expensive phosphorus ylide reagents and generate significant stoichiometric waste, this novel method utilizes common p-hydroxyacetophenone and industrial alkali. It achieves conversion rates exceeding 95% in a single vessel, drastically reducing raw material costs and waste treatment burdens associated with phosphine oxide byproducts.
Q: Can this process be scaled for industrial photoresist monomer production?
A: Yes, the process is highly amenable to scale-up as it avoids sensitive transition metal catalysts and uses robust industrial bases like NaOH or KOH. The reaction operates in standard alcohol solvents at elevated temperatures, utilizing equipment commonly available in fine chemical manufacturing facilities without requiring specialized high-pressure hydrogenation reactors.
Q: How does the impurity profile compare to acid-catalyzed dehydration routes?
A: This base-catalyzed coupling mechanism minimizes polymerization side reactions often seen in strong acid dehydration environments. By controlling the pH and utilizing specific alcohol solvents, the formation of oligomeric impurities is suppressed, resulting in a crude product yield of up to 85% and facilitating easier downstream purification to electronic-grade standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxystyrene Supplier
As the demand for advanced photoresists continues to surge, partnering with a manufacturer capable of executing complex synthetic routes is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of p-hydroxystyrene meets the exacting standards required for semiconductor applications. We understand the critical nature of electronic chemical intermediates and are committed to delivering products that enable the next generation of chip manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative one-step synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless and scientifically validated.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
