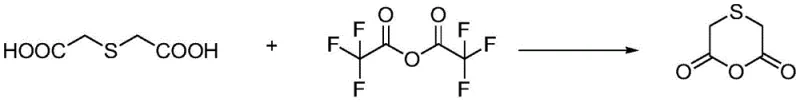
Advanced Gas-Solid Synthesis of Thiohydroxy Acetic Anhydride for High-Purity Erdosteine Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical mucolytic agents, and the preparation of thiohydroxy acetic anhydride stands as a pivotal step in the production of Erdosteine. A groundbreaking approach detailed in patent CN111995614B introduces a sophisticated suspension-state gas-solid reaction technology that fundamentally alters the manufacturing landscape for this key intermediate. Unlike traditional methods that rely heavily on liquid-phase reflux in organic solvents, this novel technique utilizes vaporized trifluoroacetic anhydride to react with solid thiodiglycolic acid in a controlled gaseous environment. This shift not only addresses long-standing issues regarding solvent residues and reaction times but also delivers a product of exceptional purity, which is paramount for meeting the stringent regulatory standards required for active pharmaceutical ingredient (API) synthesis. By leveraging this advanced gas-solid methodology, manufacturers can achieve yields exceeding 95%, representing a significant leap forward in process efficiency and product quality for the global supply chain of respiratory therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiohydroxy acetic anhydride has been plagued by inefficiencies inherent to solution-phase chemistry. Traditional protocols, such as those cited in earlier literature, typically involve refluxing thiodiglycolic acid with acetic anhydride or acetyl chloride in solvents like ethyl acetate for extended periods, often exceeding three hours. These liquid-phase reactions suffer from several critical drawbacks that impact both economic viability and product integrity. Firstly, the extensive use of organic solvents necessitates complex downstream processing, including vacuum concentration and recrystallization, which increases energy consumption and operational costs. More critically, these methods frequently result in products contaminated with residual solvents, such as ethyl acetate, which are difficult to remove completely and can interfere with subsequent coupling reactions. Furthermore, the yields associated with these conventional routes are often suboptimal, hovering around 80% to 87%, leading to significant material loss and increased waste generation. The presence of impurities and solvent residues ultimately compromises the quality of the final Erdosteine API, resulting in lower overall yields in the final drug synthesis step, thereby creating a bottleneck in the entire production value chain.
The Novel Approach
In stark contrast, the innovative method disclosed in patent CN111995614B circumvents these limitations by employing a solvent-free, suspension-state gas-solid reaction system. This approach involves vaporizing trifluoroacetic anhydride and introducing it into a reactor where solid thiodiglycolic acid is maintained in a suspended state by the upward flow of the hot gas. This unique physical arrangement ensures maximum contact area between the reactants without the dilution effect of a bulk solvent, leading to dramatically improved reaction kinetics. The process operates under mild thermal conditions, utilizing a gradient temperature profile between 45°C and 65°C, which optimizes reaction efficiency while minimizing thermal degradation. By eliminating the need for bulk solvents, the method inherently avoids solvent residue issues, producing a solid product that requires only simple washing with refrigerated petroleum ether and anhydrous ether to achieve ultra-high purity. This streamlined workflow not only reduces the reaction time to approximately two hours but also boosts yields to an impressive range of 95% to 98%, offering a robust solution for high-volume commercial manufacturing.
Mechanistic Insights into Suspension-State Gas-Solid Cyclization
The core of this technological advancement lies in the precise engineering of the gas-solid interface within the reactor. In this system, liquid trifluoroacetic anhydride is first vaporized in a dedicated heating unit equipped with electrically heated catalysts and parallel circular pipelines. This vapor is then directed into the bottom of the suspension reactor through an airflow disperser, which ensures a uniform distribution of the gaseous reagent. As the hot gas rises, it fluidizes the solid thiodiglycolic acid powder fed from the top, creating a dynamic suspension where every particle is enveloped by the reactive vapor. This state of suspension maximizes the surface area available for the dehydration and cyclization reaction, facilitating a rapid and complete conversion to the anhydride. The absence of a liquid medium prevents the solvation of intermediates that might lead to side reactions, thereby enhancing the selectivity of the transformation. The reactor is equipped with multiple thermocouples on the inner walls, allowing for real-time monitoring and precise control of the thermal gradient, which is crucial for maintaining the stability of the suspension and ensuring consistent reaction progress throughout the batch.
Furthermore, the purification mechanism is elegantly simple yet highly effective, relying on the differential solubility of impurities in cold non-polar solvents. The crude solid product, which consists primarily of the desired anhydride and minor byproducts like trifluoroacetic acid salts, is washed sequentially with refrigerated anhydrous petroleum ether and anhydrous ether. The initial wash with petroleum ether removes non-polar impurities and residual organic contaminants, while the subsequent ether wash effectively eliminates polar residues and traces of the acid reagent. This dual-washing strategy is superior to single-solvent washes, as demonstrated by comparative data showing that using either solvent alone results in lower purity levels. The volatility of the washing solvents also aids in the drying process, ensuring that the final product is free from moisture and solvent traces, which is essential for its stability and reactivity in the subsequent synthesis of Erdosteine. This meticulous control over the reaction and purification environment underscores the method's capability to deliver pharmaceutical-grade intermediates consistently.
How to Synthesize Thiohydroxy Acetic Anhydride Efficiently
Implementing this advanced synthesis route requires careful attention to the specific parameters outlined in the patent to ensure optimal performance and safety. The process begins with the preparation of the gaseous reagent stream, followed by the controlled introduction of the solid substrate into the heated reactor zone. Maintaining the correct gas flow rate is critical to keeping the solid particles in suspension without elutriating them out of the reactor. The reaction proceeds through a defined thermal profile, starting at a lower temperature to initiate the reaction gently and ramping up to ensure completion.
- Vaporize trifluoroacetic anhydride in a dedicated heating device to prepare the gaseous reactant stream.
- Introduce the vaporized reagent into a preheated suspension reactor (45-65°C) while intermittently feeding solid thiodiglycolic acid from the top to maintain a suspended state.
- React for 2 hours with gradient temperature control, then purify the solid product by washing sequentially with refrigerated anhydrous petroleum ether and anhydrous ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gas-solid synthesis technology offers profound strategic benefits that extend beyond mere technical superiority. The elimination of bulk organic solvents like ethyl acetate translates directly into substantial cost reductions, as there is no longer a need to procure, store, and recover large volumes of volatile organic compounds. This simplification of the material bill of materials (BOM) reduces the complexity of inventory management and mitigates the risks associated with solvent price volatility. Moreover, the drastic reduction in solvent usage significantly lowers the burden on waste treatment facilities, leading to decreased environmental compliance costs and a smaller carbon footprint for the manufacturing site. The high yield and purity of the process mean that less raw material is wasted, improving the overall atom economy and reducing the cost per kilogram of the final intermediate. These factors combined create a more resilient and cost-effective supply chain capable of meeting the demanding requirements of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The transition to a solvent-free gas-solid process fundamentally alters the cost structure of production by removing the expenses associated with solvent purchase, recovery, and disposal. Traditional methods require significant energy input for distillation and concentration steps to remove solvents, whereas this novel method produces a solid product that only requires simple filtration and washing. This reduction in energy intensity leads to lower utility costs and allows for faster batch turnover times. Additionally, the high yield of over 95% minimizes the loss of expensive starting materials like thiodiglycolic acid, ensuring that a greater proportion of input costs are converted into saleable product. The cumulative effect of these efficiencies is a significantly lower cost of goods sold (COGS), providing a competitive advantage in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for preventing production stoppages in the pharmaceutical sector. This new method enhances reliability by simplifying the process flow and reducing the number of unit operations that could potentially fail. The absence of complex solvent recovery systems reduces the maintenance burden on equipment and decreases the likelihood of downtime due to mechanical failures. Furthermore, the high purity of the product reduces the risk of batch rejection during quality control testing, ensuring a steady flow of compliant material to customers. The scalability of the gas-solid reactor design means that production capacity can be increased linearly to meet surges in demand without the need for disproportionate increases in facility footprint or infrastructure investment.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental emissions continue to tighten, the ability to manufacture chemicals with minimal waste is a crucial asset. This gas-solid technology aligns perfectly with green chemistry principles by eliminating solvent emissions and reducing hazardous waste generation. The process generates minimal wastewater compared to traditional aqueous workups, simplifying effluent treatment and reducing the risk of regulatory non-compliance. The reactor design, featuring airflow dispersers and thermocouples, is inherently scalable, allowing for the transition from pilot scale to multi-ton commercial production with consistent product quality. This scalability ensures that suppliers can grow alongside their customers, supporting the long-term commercialization of Erdosteine and other derivative drugs without supply bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement teams assessing supplier capabilities.
Q: What are the primary advantages of the gas-solid reaction method over traditional solvent reflux?
A: The gas-solid method eliminates the need for bulk organic solvents like ethyl acetate, significantly reducing solvent residue in the final product. It also shortens reaction time from over 3 hours to approximately 2 hours while improving yields from roughly 80% to over 95%.
Q: How does this synthesis method impact the quality of the downstream drug Erdosteine?
A: By producing thiohydroxy acetic anhydride with extremely high purity (up to 100% by HPLC in examples) and no solvent residues, the subsequent coupling reaction with homocysteine thiolactone achieves significantly higher yields, directly improving the efficiency of the final API manufacturing.
Q: Is this process scalable for industrial production?
A: Yes, the patent describes a continuous flow setup with vaporization and suspension reactors equipped with thermocouples and airflow dispersers, which are design features specifically intended to facilitate uniform heat transfer and scalability in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiohydroxy Acetic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and manufacturing of life-saving medications. Our team of expert chemists has thoroughly analyzed the potential of the gas-solid synthesis route described in CN111995614B and is fully prepared to implement this technology at an industrial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in tangible supply chain improvements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of thiohydroxy acetic anhydride meets the exacting standards required for Erdosteine synthesis. By partnering with us, you gain access to a supply chain that is not only cost-effective but also robust, compliant, and technically advanced.
We invite you to engage with our technical procurement team to discuss how we can optimize your current supply strategy. Whether you are looking to reduce costs in pharmaceutical intermediate manufacturing or seeking a more reliable source for high-purity materials, we are ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to request specific COA data and route feasibility assessments to verify our capabilities firsthand. Let us help you engineer a more efficient and sustainable future for your pharmaceutical production lines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
