Advanced Fe/Cu Catalytic Route for Commercial Levofloxacin Hydrochloride Manufacturing
Introduction to Patent CN102850377B Technology
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for producing critical antibiotics like Levofloxacin Hydrochloride, a potent fluoroquinolone antibacterial agent. Patent CN102850377B introduces a groundbreaking preparation method that fundamentally shifts the catalytic paradigm from expensive noble metals to a highly efficient Iron and Copper (Fe/Cu) dual catalytic system. This innovation addresses the longstanding challenges of high production costs and environmental toxicity associated with traditional Palladium or Platinum-catalyzed coupling reactions. By leveraging the synergistic effects of CuI and Fe(AcAc)3, the disclosed methodology achieves exceptional reaction selectivity and yield across multiple critical bond-forming steps, including C-C, C-O, and C-N couplings. This technical breakthrough not only streamlines the synthetic route but also aligns perfectly with modern green chemistry principles, offering a sustainable solution for the commercial manufacturing of high-purity pharmaceutical intermediates.
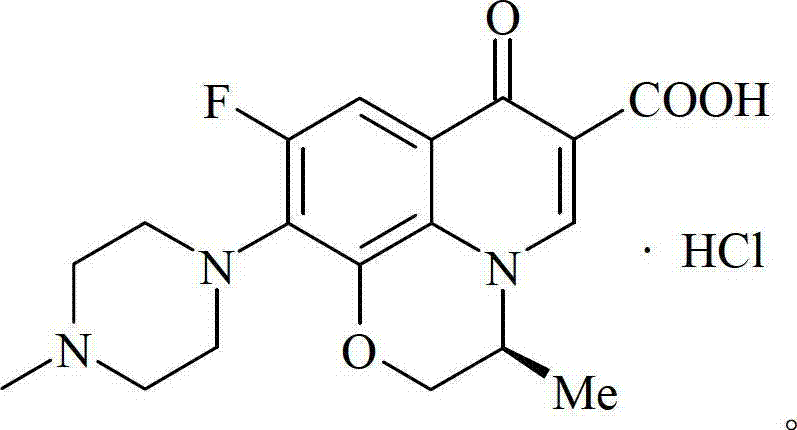
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex fluoroquinolone antibiotics has relied heavily on transition metal catalysts such as Palladium, Platinum, Ruthenium, and Rhodium to facilitate crucial carbon-carbon and carbon-nitrogen bond formations. While effective, these noble metal catalysts present significant logistical and economic hurdles for large-scale industrial applications, primarily due to their exorbitant market prices and susceptibility to deactivation by impurities. Furthermore, the removal of trace heavy metal residues from the final Active Pharmaceutical Ingredient (API) requires sophisticated and costly purification processes to meet strict regulatory safety limits. The disposal of spent noble metal catalysts also poses environmental challenges, complicating waste management protocols and increasing the overall ecological footprint of the manufacturing process. Consequently, reliance on these traditional catalytic systems often results in inflated production costs and extended lead times, creating bottlenecks for supply chain managers aiming to optimize procurement strategies for essential antibiotics.
The Novel Approach
In stark contrast to conventional methodologies, the novel approach detailed in Patent CN102850377B utilizes an earth-abundant Fe/Cu catalytic system that delivers superior performance without the financial burden of precious metals. This innovative route employs Copper Iodide (CuI) and Iron(III) acetylacetonate (Fe(AcAc)3) to drive key coupling reactions with remarkable efficiency, demonstrating broad catalytic scope and excellent functional group tolerance. The substitution of noble metals with this inexpensive dual-metal system drastically simplifies the downstream processing requirements, as the catalysts can be easily recovered and recycled through simple filtration techniques. This shift not only reduces the direct material costs but also minimizes the generation of hazardous heavy metal waste, thereby enhancing the overall sustainability profile of the synthesis. By integrating this greener catalytic technology, manufacturers can achieve a more resilient and cost-effective production workflow that is better suited for the demands of the global generic pharmaceutical market.
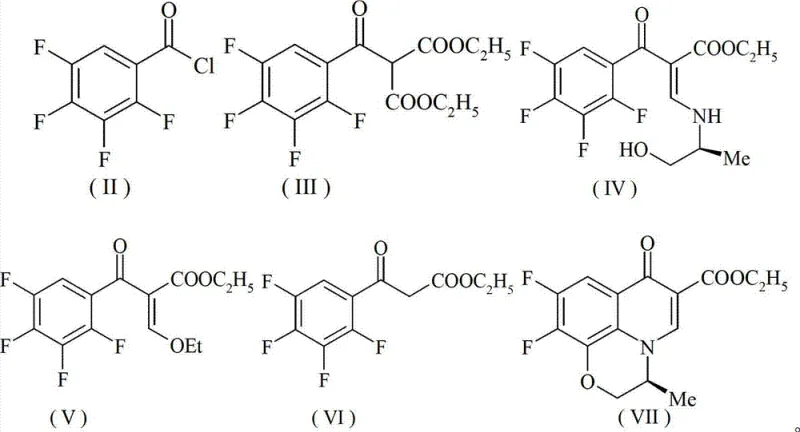
Mechanistic Insights into Fe/Cu-Catalyzed Cyclization and Coupling
The core mechanistic advantage of this synthesis lies in the synergistic interaction between the copper and iron species, which facilitates complex bond constructions under relatively mild conditions. In the critical cyclization steps, the CuI likely acts as a soft Lewis acid to activate halide leaving groups, while the Fe(AcAc)3 serves as a redox-active center that promotes radical or organometallic intermediates necessary for ring closure. This dual activation mechanism allows for the efficient formation of the quinolone backbone and the subsequent introduction of the chiral piperazine moiety with high stereochemical fidelity. The patent data suggests that this catalytic manifold is particularly effective for C-N bond formation, a notoriously difficult transformation that often requires harsh conditions or specialized ligands when using single-metal systems. By optimizing the molar ratios and reaction temperatures, the process achieves selectivity rates approaching 99%, effectively suppressing the formation of regioisomers and other structural impurities that typically plague fluoroquinolone synthesis. This high level of control is essential for ensuring the biological potency and safety profile of the final Levofloxacin product.
Furthermore, the robustness of the Fe/Cu system extends to its tolerance of various functional groups present in the polyfluorinated aromatic precursors, which are prone to side reactions under aggressive catalytic conditions. The mechanism likely involves a coordinated cycle where the iron species stabilizes the enolate or carbanion intermediates generated during the malonate coupling, preventing premature decomposition or polymerization. This stability is crucial during the high-temperature cyclization steps performed in polar aprotic solvents like DMF or DMSO, where traditional catalysts might degrade or precipitate. The ability to maintain catalytic activity over extended reaction times without significant loss of turnover number indicates a durable catalytic cycle that supports continuous or semi-continuous processing modes. For R&D directors, understanding this mechanistic resilience provides confidence in scaling the process from laboratory benchtop to multi-ton commercial reactors without encountering unpredictable kinetic barriers or yield drops.
How to Synthesize Levofloxacin Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a comprehensive, step-by-step guide for transforming readily available 2,3,4,5-tetrafluorobenzoic acid into the final Levofloxacin Hydrochloride salt through a nine-step sequence. The process begins with the activation of the benzoic acid derivative followed by sequential coupling, cyclization, and functionalization reactions, each optimized for maximum yield and purity using the proprietary Fe/Cu catalyst system. Detailed operational parameters, including solvent volumes, temperature gradients, and reagent stoichiometry, are meticulously defined to ensure reproducibility across different manufacturing scales. The following section outlines the standardized synthesis steps derived directly from the patent examples, serving as a foundational reference for process engineers and chemists looking to implement this technology.
- Acylation of 2,3,4,5-tetrafluorobenzoic acid followed by Fe/Cu catalyzed coupling with diethyl malonate.
- Hydrolysis, decarboxylation, and condensation to form the enamine intermediate.
- Chiral amine introduction and dual Fe/Cu catalyzed cyclization to form the quinolone core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Fe/Cu catalytic technology represents a strategic opportunity to significantly reduce the total cost of ownership for Levofloxacin production. By eliminating the dependency on volatile noble metal markets, manufacturers can stabilize their raw material costs and mitigate the financial risks associated with price fluctuations of Palladium and Platinum. The simplified purification workflow, driven by the ease of catalyst removal and recycling, translates directly into reduced processing time and lower utility consumption per kilogram of product. Additionally, the use of less corrosive reagents and the generation of less hazardous waste streamline compliance with environmental regulations, potentially lowering waste disposal fees and insurance premiums. These cumulative efficiencies create a more competitive cost structure, allowing suppliers to offer high-quality API intermediates at more attractive price points while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with abundant Iron and Copper salts results in a drastic reduction in direct material expenses, which is a primary driver of overall manufacturing costs. Since the catalysts can be recovered and reused multiple times without significant loss of activity, the effective cost per batch is further diminished over the lifecycle of the production campaign. This economic advantage is compounded by the high reaction yields reported in the patent, which minimize the loss of valuable starting materials and reduce the need for extensive reprocessing of off-spec batches. Consequently, the overall cost reduction in pharmaceutical intermediates manufacturing is substantial, providing a clear financial incentive for switching to this greener synthetic route.
- Enhanced Supply Chain Reliability: Sourcing Iron and Copper catalysts is far more reliable and geographically diverse compared to the concentrated supply chains of rare earth and noble metals, which are often subject to geopolitical tensions and export restrictions. This diversification of the supplier base ensures a steady flow of critical reagents, reducing the risk of production stoppages due to material shortages. Furthermore, the robustness of the reaction conditions allows for greater flexibility in scheduling and inventory management, as the process is less sensitive to minor variations in reagent quality or environmental conditions. This enhanced reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical clients who depend on uninterrupted supplies of essential antibiotics.
- Scalability and Environmental Compliance: The nine-step synthesis described is designed with scalability in mind, utilizing common industrial solvents and equipment that are readily available in standard chemical manufacturing facilities. The reduced corrosivity of the reagents extends the lifespan of reactor vessels and piping, lowering capital expenditure on maintenance and replacement of infrastructure. From an environmental perspective, the non-toxic nature of the catalysts and the minimized waste generation align with increasingly stringent global environmental standards, facilitating easier permitting and regulatory approval for new production lines. This alignment with green chemistry principles not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the healthcare value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Fe/Cu catalytic route for Levofloxacin synthesis. These answers are derived from the specific experimental data and beneficial effects described in Patent CN102850377B, providing clarity on the practical implications of adopting this technology. Understanding these details is vital for stakeholders evaluating the feasibility and advantages of integrating this method into their existing production portfolios.
Q: What is the primary advantage of the Fe/Cu catalytic system over traditional methods?
A: The Fe/Cu system replaces expensive noble metals like Palladium and Platinum, significantly reducing raw material costs while maintaining high selectivity for C-C and C-N bond formation.
Q: Is the catalyst recyclable in this Levofloxacin synthesis process?
A: Yes, the patent specifies that CuI and Fe(AcAc)3 can be recovered and recycled after simple filtration, enhancing the environmental profile and long-term cost efficiency.
Q: What is the expected purity of the final Levofloxacin Hydrochloride product?
A: Experimental data in the patent indicates HPLC purity analysis consistently exceeding 99% across multiple scale-up examples, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levofloxacin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Fe/Cu catalytic technology disclosed in Patent CN102850377B for the efficient production of Levofloxacin Hydrochloride. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes are translated into robust, GMP-compliant manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of API intermediate delivered meets the highest international quality standards. We are committed to leveraging our technical expertise to help partners navigate the transition to this cost-effective and environmentally friendly synthesis method.
We invite pharmaceutical companies and procurement leaders to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the confidence of a proven and scalable technological solution.
