Scalable Manufacturing of Siponimod Intermediates via Novel Palladium-Catalyzed Route for Global Pharma Supply Chains
Scalable Manufacturing of Siponimod Intermediates via Novel Palladium-Catalyzed Route for Global Pharma Supply Chains
The pharmaceutical landscape for treating secondary progressive multiple sclerosis has been significantly advanced by the development of Siponimod, a second-generation S1P receptor modulator. As detailed in patent CN113382982A, a groundbreaking preparation method has been established that fundamentally alters the synthetic approach to this critical active pharmaceutical ingredient. This novel methodology departs from traditional, hazardous pathways by utilizing 3-bromo-4-methylacetophenone as a robust starting material, proceeding through a sequence of bromination, nucleophilic substitution, vinyl coupling, and catalytic hydrogenation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent offers a compelling blueprint for high-purity production that mitigates the severe safety risks associated with diazotization reactions found in legacy processes. The strategic implementation of this route promises not only enhanced chemical safety but also a dramatic simplification of downstream processing.
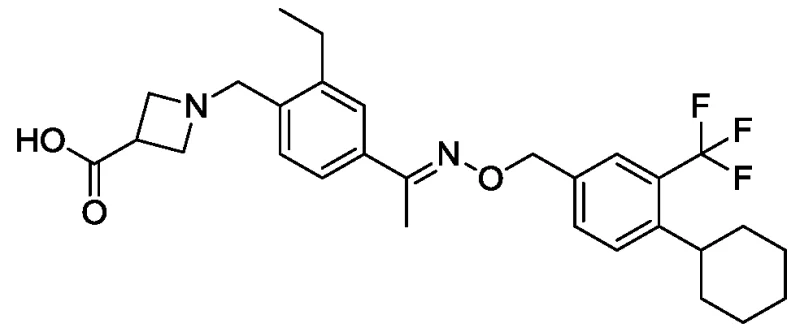
The structural complexity of Siponimod, characterized by its specific substitution pattern on the aromatic ring and the chiral azetidine moiety, demands a synthesis strategy that balances precision with scalability. The invention described herein achieves the target molecule, represented as Formula VII, through a linear and convergent strategy that avoids the fragmentation issues of earlier attempts. By focusing on the efficient assembly of the core aromatic scaffold before introducing the complex side chains, the process ensures that impurities are minimized at every stage. This approach is particularly vital for maintaining the stringent quality standards required for neurology APIs, where trace impurities can have profound biological consequences. The ability to produce this molecule with high yield and purity positions this method as a cornerstone for future commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for Siponimod have been plagued by significant operational hazards and inefficiencies that render them suboptimal for large-scale industrial application. Specifically, earlier methodologies often relied on diazotization reactions to construct key segments of the molecule, a process notorious for its potential to cause violent explosions if not meticulously controlled. Furthermore, these legacy routes frequently necessitated up to five separate column chromatography purification steps to achieve acceptable purity levels, a practice that is economically prohibitive and environmentally unsustainable in a manufacturing setting. The reliance on such labor-intensive purification techniques not only drives up the cost of goods sold but also introduces bottlenecks that severely impact reducing lead time for high-purity pharmaceutical intermediates. Additionally, the use of unstable intermediates in these old pathways increases the risk of batch failures, creating supply chain volatility that procurement managers strive to avoid.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent leverages a stable and modular synthetic sequence that begins with the readily available 3-bromo-4-methylacetophenone. This new pathway ingeniously bypasses the dangerous diazotization step entirely, replacing it with a controlled radical bromination followed by a robust nucleophilic substitution with an azetidine derivative. The subsequent installation of the vinyl group via a palladium-catalyzed cross-coupling reaction demonstrates exceptional chemoselectivity, allowing for the construction of the carbon skeleton without affecting other sensitive functional groups. This streamlined logic eliminates the need for repetitive chromatographic purification, favoring simpler isolation techniques like extraction and crystallization that are inherently more scalable. By integrating these improvements, the process delivers a substantial reduction in operational complexity while simultaneously enhancing the overall safety profile of the manufacturing facility.
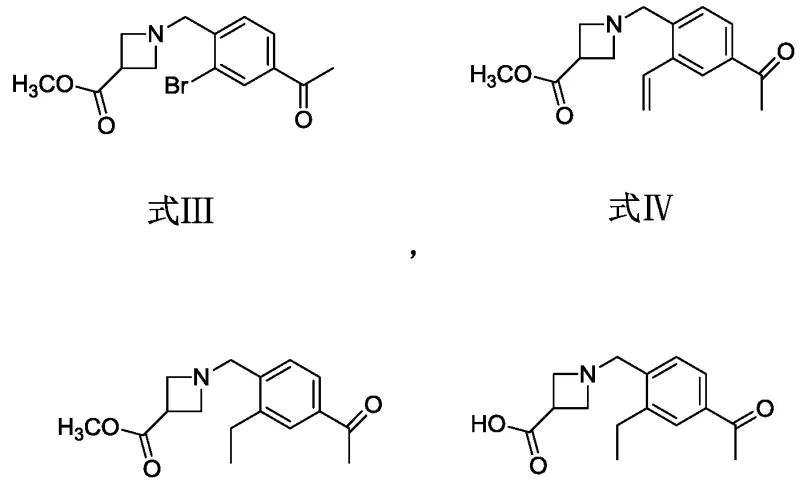
Mechanistic Insights into Palladium-Catalyzed Vinyl Coupling
The heart of this synthetic innovation lies in the efficient execution of the Suzuki-Miyaura cross-coupling reaction, which serves as the pivotal C-C bond-forming event in the sequence. In this specific transformation, the bromo-substituted azetidine intermediate (Formula III) reacts with potassium vinyltrifluoroborate in the presence of a palladium catalyst, such as [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride. The mechanism proceeds through the classic oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the organoboron reagent activated by a base like sodium carbonate. This step is critical because it installs the vinyl functionality with high fidelity, setting the stage for the subsequent hydrogenation step. The choice of a biphasic solvent system involving toluene, ethanol, and water facilitates the solubility of both organic and inorganic components, ensuring rapid reaction kinetics and high conversion rates even at moderate temperatures ranging from 60°C to 80°C.
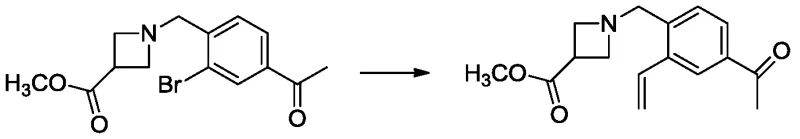
Following the coupling, the process employs a catalytic hydrogenation step to reduce the newly formed vinyl double bond to an ethyl group, transforming Formula IV into Formula V. This reduction is typically carried out using palladium on carbon (Pd/C) under a hydrogen atmosphere, a method chosen for its cleanliness and atom economy. The mechanistic simplicity of this hydrogenation ensures that no toxic metal residues remain in the final product, a crucial factor for regulatory compliance. Furthermore, the final hydrolysis of the methyl ester to the carboxylic acid (Formula VI) is achieved under mild basic conditions using aqueous sodium hydroxide, avoiding harsh acidic conditions that could degrade the sensitive azetidine ring. This careful orchestration of reaction conditions throughout the pathway exemplifies a deep understanding of impurity control, ensuring that the final condensation with the oxime ether precursor yields Siponimod with the requisite pharmaceutical grade purity.
How to Synthesize Siponimod Intermediates Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The patented procedure outlines a clear progression from simple starting materials to complex scaffolds, emphasizing the importance of stoichiometry and temperature management. For instance, the initial bromination step utilizes N-bromosuccinimide (NBS) and a radical initiator like dibenzoyl peroxide to selectively functionalize the benzylic position without affecting the aromatic ring or the ketone. Subsequent steps rely on the careful selection of bases and solvents to drive equilibria towards the desired products. While the specific experimental details are extensive, the general workflow provides a robust framework for process chemists aiming to replicate this efficiency. For a comprehensive breakdown of the specific reagents, molar ratios, and work-up procedures, please refer to the standardized guide below.
- Perform radical bromination of 3-bromo-4-methylacetophenone using NBS and BPO to generate the benzyl bromide intermediate.
- Couple the bromide with methyl azetidine-3-carboxylate followed by Suzuki-Miyaura cross-coupling with potassium vinyltrifluoroborate.
- Execute catalytic hydrogenation to reduce the vinyl group, followed by ester hydrolysis and final condensation to yield Siponimod.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers transformative benefits for procurement managers and supply chain leaders tasked with securing reliable sources of critical API intermediates. The most immediate impact is seen in the drastic simplification of the purification process; by eliminating the need for multiple columns of silica gel chromatography, manufacturers can significantly reduce solvent consumption and waste generation. This reduction in downstream processing directly translates to lower operating expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives. Moreover, the avoidance of hazardous diazotization reactions removes a major safety liability, potentially lowering insurance costs and reducing the need for specialized explosion-proof infrastructure. These factors combined create a more resilient supply chain capable of withstanding market fluctuations and regulatory scrutiny.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the replacement of expensive and inefficient purification methods with scalable crystallization and extraction techniques. By removing the dependency on column chromatography, the process drastically cuts down on the volume of organic solvents required, which are often a significant cost driver in fine chemical synthesis. Additionally, the use of stable, commercially available starting materials like 3-bromo-4-methylacetophenone ensures that raw material costs remain predictable and low. The high yields reported in the patent examples further contribute to cost efficiency by maximizing the output per batch, thereby reducing the cost per kilogram of the final intermediate. This holistic approach to cost optimization makes the technology highly attractive for cost reduction in API manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the use of exotic or unstable reagents that have long lead times or limited availability. This new route mitigates such risks by relying on commodity chemicals and robust catalytic systems that are widely accessible in the global market. The operational simplicity of the steps, which do not require cryogenic conditions or ultra-high pressures, allows for production in a broader range of manufacturing facilities, thereby diversifying the potential supplier base. This flexibility is crucial for pharmaceutical companies looking to dual-source critical materials to prevent shortages. The inherent stability of the intermediates also allows for easier storage and transportation, further smoothing out logistical challenges and ensuring a steady flow of materials to the final drug product manufacturing sites.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to the plant floor often reveals hidden bottlenecks, but this methodology is designed with scalability in mind from the outset. The reactions operate at moderate temperatures and pressures, utilizing standard stainless steel reactors that are common in the industry. The elimination of hazardous diazonium salts removes a significant barrier to scale-up, as these species are notoriously difficult to handle in large quantities. Furthermore, the reduced solvent usage and the ability to recycle aqueous streams from the hydrolysis step contribute to a greener process profile. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations aiming to meet sustainability targets while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Siponimod synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on how this method compares to existing technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new process for their own production lines. The focus remains on safety, purity, and operational efficiency, which are the primary drivers for technology transfer in the pharmaceutical sector.
Q: How does this new route improve safety compared to prior art?
A: Unlike previous methods requiring hazardous diazotization reactions which pose explosion risks, this novel pathway utilizes stable radical bromination and palladium-catalyzed coupling under mild conditions, significantly enhancing operational safety.
Q: Does this process require expensive column chromatography?
A: No, a major advantage of this invention is the elimination of multiple column chromatography purification steps found in older routes, allowing for simpler work-up procedures like extraction and crystallization which are ideal for industrial scale-up.
Q: What represents the key C-C bond forming step in this synthesis?
A: The critical carbon-carbon bond formation is achieved via a Suzuki-Miyaura cross-coupling reaction between the bromo-intermediate and potassium vinyltrifluoroborate, utilizing a palladium catalyst system that ensures high conversion and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Siponimod Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant and finally to full-scale manufacturing is seamless. We are committed to delivering Siponimod intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle the specific requirements of palladium-catalyzed reactions and hydrogenation steps safely and efficiently, guaranteeing a consistent supply of high-quality materials for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and timeline requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this superior manufacturing process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your time to market.
