Optimizing L-Phenylglycine Production: A Technical Breakthrough in Acidic Racemization and Resolution
Optimizing L-Phenylglycine Production: A Technical Breakthrough in Acidic Racemization and Resolution
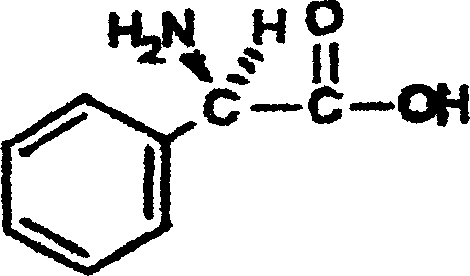
The global demand for high-purity beta-lactam antibiotic intermediates continues to drive innovation in chiral synthesis, particularly for critical building blocks like D-(-)-1-phenylglycine, also known as L-Phenylglycine. As a pivotal precursor for semi-synthetic penicillins such as ampicillin and cephalosporins like cephalexin, the efficiency of its production directly impacts the cost and availability of life-saving medications. A significant technological advancement in this domain is detailed in patent CN1293041C, which outlines a superior preparation method involving the optical disassembly of DL-phenylglycine generated through the racemization of the dextrorotation isomer. This approach fundamentally shifts the paradigm from traditional alkaline racemization to a more sustainable, acidic catalytic cycle, offering profound implications for industrial scalability and environmental compliance.
The core innovation lies in the strategic recycling of the mother liquor. In conventional resolution processes, the unwanted enantiomer often accumulates or requires harsh conditions to revert to the racemic mixture, leading to substantial material loss. The methodology described in CN1293041C introduces a cyclic operation where the acidic mother liquor, post-filtration of the target levo-isomer, is treated with specific aromatic aldehyde compounds. This treatment facilitates the conversion of the residual dextrorotation phenylglycine back into the DL-racemic form under mild acidic reflux conditions. By integrating this racemization step directly into the resolution workflow, the process achieves a closed-loop system that maximizes atom economy and minimizes the consumption of fresh starting materials, establishing a new benchmark for efficient pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of L-Phenylglycine has been plagued by inefficiencies inherent in traditional optical resolution techniques. Prior art methods, such as those disclosed in U.S. Patent 4,215,223 or Chinese Patent CN1030573A, typically rely on asymmetric fractionation using tartaric acid or camphor sulfonic acids, followed by a separate racemization step for the unwanted isomer. A critical bottleneck in these legacy processes is the racemization condition, which predominantly utilizes strong alkaline agents like sodium hydroxide or potassium hydroxide. While chemically effective at inverting chirality, the use of highly basic conditions necessitates the generation of large quantities of inorganic salt byproducts upon neutralization. This not only complicates the downstream purification process but also imposes a heavy burden on wastewater treatment facilities due to high salinity and pH fluctuations. Furthermore, the multi-step nature of isolating the unwanted isomer, neutralizing the base, and re-acidifying the solution for the next resolution cycle elongates the production timeline and increases energy consumption, rendering these methods less competitive in a cost-sensitive market.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages an acidic environment to drive the racemization equilibrium, effectively bypassing the need for caustic bases. By introducing aromatic aldehyde compounds—such as benzaldehyde, salicylaldehyde, or phenylacetaldehyde—into the acidic mother liquor, the process catalyzes the racemization of the dextrorotation phenylglycine at temperatures between 105°C and 110°C. This shift to acidic catalysis is transformative; it eliminates the formation of inorganic salts associated with base neutralization, thereby significantly reducing the "three wastes" (wastewater, waste gas, and solid waste) profile of the plant. Moreover, the process is designed for continuous circulation. The reaction liquid, after the removal of the aromatic aldehyde via azeotropic distillation, is merged with fresh DL-phenylglycine and resolving agent for immediate reuse. This seamless integration of racemization and resolution not only shortens the technological process flow but also ensures that the valuable chiral material is never discarded, leading to a drastic improvement in overall yield and resource utilization compared to batch-wise alkaline methods.
Mechanistic Insights into Acidic Aldehyde-Catalyzed Racemization
The chemical elegance of this process resides in the interaction between the amino acid substrate and the aromatic aldehyde under acidic conditions. In the presence of sulfuric acid and an aromatic aldehyde, the primary amine group of the phenylglycine likely undergoes a reversible condensation to form a Schiff base (imine) intermediate. This imine formation renders the alpha-proton adjacent to the imine nitrogen significantly more acidic than in the free amino acid, facilitating its abstraction and subsequent reprotonation. This mechanism allows for the rapid equilibration of the stereocenter, converting the pure dextrorotation isomer back into a thermodynamic 1:1 mixture of DL-phenylglycine. The use of d-camphorsulfonic acid (CSA) as the resolving agent complements this mechanism perfectly; CSA forms a highly insoluble double salt specifically with the D-(-)-enantiomer in the acidic medium. This differential solubility drives the equilibrium forward, precipitating the desired product while leaving the unwanted isomer in the solution to be recycled. The precision of this chiral recognition ensures that the optical purity of the final crystalline product remains exceptionally high, as evidenced by specific rotation values consistently exceeding -154° in the provided embodiments.
From an impurity control perspective, this acidic pathway offers superior selectivity compared to alkaline hydrolysis. Strong base conditions can sometimes promote side reactions such as peptide bond formation (dimerization) or degradation of the sensitive phenylglycine structure. By maintaining a controlled acidic pH (adjusted to approximately 0.0 ± 0.4 with sulfuric acid), the integrity of the amino acid backbone is preserved throughout the rigorous reflux conditions. Furthermore, the azeotropic removal of the aromatic aldehyde serves a dual purpose: it drives the racemization equilibrium to completion by removing water and prevents the accumulation of aldehyde-derived impurities in the final product. The ability to recycle the aldehyde distillate further underscores the robustness of the mechanism, ensuring that the catalytic promoter does not become a contaminant in the final high-purity pharmaceutical intermediate. This meticulous control over reaction parameters results in a product profile that meets the stringent specifications required for downstream antibiotic synthesis.
How to Synthesize D-(-)-1-phenylglycine Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize the efficiency of the chiral pool. The process begins with the dissolution of DL-phenylglycine and d-camphorsulfonic acid in an aqueous sulfuric acid medium, followed by a controlled heating phase to effect resolution. Once the target double salt is filtered, the critical racemization step is initiated in the mother liquor by adding 5-10 weight percent of an aromatic aldehyde. The mixture is then subjected to reflux for 3 to 5 hours, ensuring complete stereochemical inversion. Following this, the aromatic aldehyde is stripped via distillation, and the recycled liquor is fortified with fresh substrate for the next cycle. For a comprehensive breakdown of the specific temperature ramps, stirring rates, and crystallization protocols necessary for GMP-compliant production, please refer to the standardized operating procedure outlined below.
- Perform optical resolution of DL-phenylglycine using d-camphorsulfonic acid in an acidic sulfuric acid medium to isolate the D-(-)-enantiomer double salt.
- Treat the acidic mother liquor containing the unwanted dextrorotation isomer with 5-10 wt% aromatic aldehyde and reflux at 105-110°C to induce racemization.
- Remove the aromatic aldehyde via azeotropic distillation, merge the recycled mother liquor with fresh substrate, and repeat the resolution cycle to maximize yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this acidic racemization technology represents a strategic opportunity to optimize the cost structure of antibiotic manufacturing. The elimination of strong alkali reagents like sodium hydroxide removes a significant variable cost component and, more importantly, eradicates the logistical and financial burden associated with the disposal of high-salt wastewater. In many jurisdictions, the cost of treating saline effluent is a major operational expense; by switching to a process that generates minimal inorganic waste, manufacturers can achieve substantial cost savings in utilities and environmental compliance. Additionally, the recyclability of the aromatic aldehyde catalyst means that the effective consumption of this reagent is negligible over multiple batches, further driving down the raw material cost per kilogram of the final active pharmaceutical ingredient (API) intermediate.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the high atom economy achieved through the cyclic reuse of the mother liquor. Unlike linear processes where the unwanted enantiomer is discarded or requires expensive separate processing, this method converts nearly 100% of the starting material into the desired product over successive cycles. The removal of the neutralization step (acid-base reaction) saves on both reagent costs and the energy required for heating and cooling large volumes of neutralization mixtures. Consequently, the overall production cost is significantly reduced, providing a competitive pricing advantage for reliable pharmaceutical intermediate suppliers who adopt this technology.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses and the dependency on diverse raw material streams. This streamlined process simplifies the supply chain by reducing the number of distinct chemical inputs; the aromatic aldehydes used are commodity chemicals with stable global availability. Furthermore, the robustness of the acidic resolution reduces the risk of batch failures due to pH excursions or salt precipitation issues common in alkaline methods. This reliability translates to more predictable lead times and a steadier flow of materials for downstream drug manufacturers, mitigating the risk of production stoppages.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste management challenges, but this technology is inherently green by design. The absence of heavy metal catalysts and the minimization of salt waste make it easier to scale from pilot plants to multi-ton commercial reactors without hitting environmental permitting bottlenecks. The simplified work-up procedure, which primarily involves filtration and distillation, is easily adaptable to large-scale continuous processing equipment. This scalability ensures that commercial scale-up of complex chiral intermediates can be achieved rapidly to meet surging market demand for generic antibiotics, all while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this specific racemization and resolution pathway. These answers are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing infrastructure. Understanding these nuances is critical for R&D teams assessing the feasibility of technology transfer and for quality assurance teams defining critical process parameters.
Q: What resolving agent is utilized in this novel L-Phenylglycine process?
A: The process utilizes d-camphorsulfonic acid (CSA) as the chiral resolving agent within a sulfuric acid medium, which forms a separable double salt with the target D-(-)-1-phenylglycine enantiomer.
Q: How does this method reduce environmental waste compared to traditional alkaline racemization?
A: Unlike conventional methods that require strong bases like sodium hydroxide or potassium hydroxide—generating significant inorganic salt waste—this invention employs acidic racemization catalyzed by aromatic aldehydes, drastically reducing three-waste pollution.
Q: Is the aromatic aldehyde catalyst recoverable in this synthesis route?
A: Yes, the aromatic aldehyde compound (such as benzaldehyde or salicylaldehyde) is removed via water-aldehyde azeotropic distillation after the racemization step, allowing it to be separated and recycled for subsequent batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-(-)-1-phenylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN1293041C requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this acidic racemization process are fully realized in a GMP environment. Our facility is equipped with state-of-the-art reactor systems capable of handling the precise temperature control and azeotropic distillation required for this chemistry, alongside rigorous QC labs that enforce stringent purity specifications for every batch of L-Phenylglycine we produce.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-efficient technology for your antibiotic supply chain. By optimizing the synthesis of this critical intermediate, we can help you achieve significant reductions in COGS (Cost of Goods Sold) without compromising on quality or regulatory compliance. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your competitive position in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
