Advanced C-Glycoside Surfactants: Scalable Green Synthesis for Commercial Applications
The chemical industry is currently witnessing a paradigm shift towards sustainable, bio-based amphiphilic compositions, driven by the urgent need for environmentally benign surfactants that do not compromise on performance. Patent CN102812032A introduces a groundbreaking class of amphiphilic C-glycoside derivatives, specifically novel linear and cyclic enone-glycolipids, which represent a significant technological leap over traditional surfactant chemistries. These compounds are characterized by a robust carbon-carbon linkage between the carbohydrate head group and the hydrophobic tail, providing exceptional stability against hydrolysis compared to conventional O-glycosides. The invention details a versatile synthetic methodology that leverages renewable biomass fractions, such as glucose, to create high-performance surface-active agents suitable for diverse applications ranging from personal care formulations to pharmaceutical drug delivery systems. This report analyzes the technical merits and commercial viability of this technology, highlighting its potential to redefine supply chain standards for high-purity specialty chemicals.
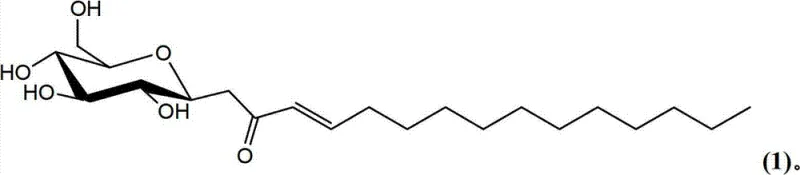
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional surfactant manufacturing often relies heavily on petrochemical feedstocks and involves complex multi-step syntheses that require hazardous organic solvents and harsh reaction conditions. Conventional O-glycosidic surfactants, while widely used, suffer from inherent chemical instability due to the acetal linkage, which is susceptible to acid-catalyzed hydrolysis, limiting their utility in formulations with low pH or high thermal stress. Furthermore, the purification processes for these traditional molecules frequently involve energy-intensive distillation or chromatography steps that generate significant volumes of toxic waste, posing challenges for environmental compliance and increasing the overall cost of goods sold. The reliance on non-renewable resources also exposes manufacturers to volatile raw material pricing and supply chain disruptions, making long-term strategic planning difficult for procurement teams seeking stability and predictability in their sourcing portfolios.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a green chemistry framework that prioritizes atom economy and the use of safer solvents, specifically water. By forming a C-glycosidic bond through an aldol-type condensation, the resulting enone-glycolipids exhibit enhanced thermal and chemical stability, ensuring consistent performance throughout the product lifecycle. The synthesis avoids the use of heavy metal catalysts or protected sugar intermediates, streamlining the production workflow and reducing the environmental footprint associated with waste disposal. This method allows for the direct functionalization of the enone moiety, providing a reactive handle for further derivatization or polymerization, which opens up new avenues for creating customized amphiphilic polymers. The ability to tune the hydrophobic-lipophilic balance (HLB) by simply varying the aldehyde chain length offers formulators unprecedented flexibility without necessitating changes to the core synthetic infrastructure.
Mechanistic Insights into C-Glycoside Aldol Condensation
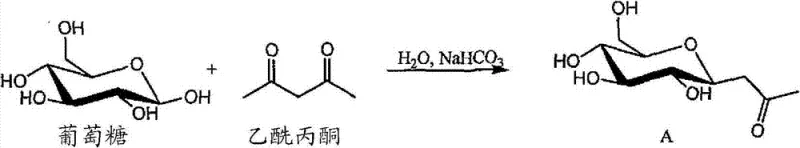
The core of this technology lies in a sophisticated yet operationally simple two-step mechanistic pathway that ensures high regioselectivity and yield. The first step involves the reaction of a carbohydrate, such as glucose, with a 1,3-diketone like acetylacetone in a mild alkaline aqueous solution. This Novenger-type condensation facilitates the formation of a C-glycosyl ketone intermediate, establishing the critical carbon-carbon bond that defines the stability of the final product. The use of water as a solvent not only aligns with green chemistry principles but also simplifies the workup procedure, as inorganic salts can be easily removed via ion exchange or simple filtration. The reaction conditions are mild enough to prevent the degradation of the sensitive carbohydrate moiety, preserving the stereochemical integrity required for specific biological interactions in pharmaceutical applications.
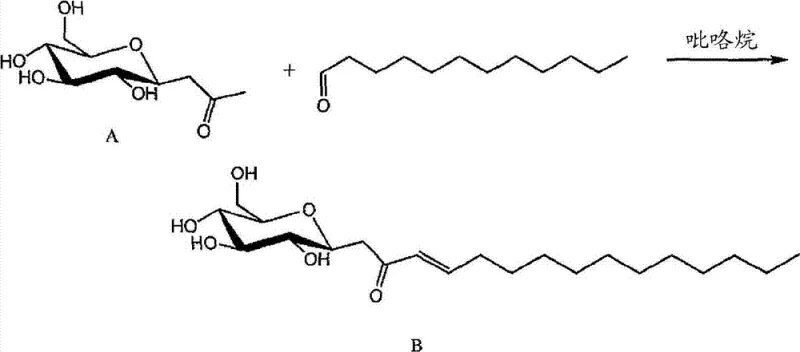
In the second step, the C-glycosyl ketone intermediate undergoes an aldol condensation with a selected aldehyde in the presence of an organocatalyst such as pyrrolidine. This step introduces the hydrophobic lipid tail, completing the amphiphilic architecture of the molecule. The enone functionality generated during this process is conjugated, providing rigidity to the molecular center which influences the self-assembly behavior of the surfactant in solution, leading to unique micellar properties and critical micelle concentration (CMC) profiles. The mechanism allows for the incorporation of various lipid chains, including saturated and unsaturated fatty acids, enabling precise tuning of the surfactant's physical properties. This modularity is crucial for R&D directors who need to optimize formulation viscosity, foaming characteristics, and emulsification efficiency without compromising on the biodegradability of the final product.
How to Synthesize Linear Enone-Glycoside Surfactants Efficiently
Implementing this synthesis route requires careful control of reaction parameters to maximize yield and purity, particularly when scaling from laboratory benchtop to pilot plant operations. The process begins with the preparation of the C-glycoside intermediate in an aqueous medium, followed by the addition of the lipid precursor and catalyst under controlled temperature conditions. Detailed standardized operating procedures are essential to ensure batch-to-batch consistency, especially regarding the stoichiometry of the aldehyde and the duration of the catalytic cycle. The following guide outlines the critical operational milestones necessary for successful production, ensuring that the final material meets stringent quality specifications required for high-end commercial applications.
- React carbohydrate with 1,3-diketone in mild alkaline aqueous solution to form C-glycoside intermediate.
- React the C-glycoside intermediate with an aldehyde in the presence of a catalyst like pyrrolidine.
- Purify the resulting enone glycoside via extraction and ion exchange to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial strategic advantages that directly address the pain points of modern supply chain management and cost optimization initiatives. The reliance on water as a primary solvent eliminates the need for expensive solvent recovery systems and reduces the regulatory burden associated with volatile organic compound emissions. This simplification of the manufacturing process translates into lower capital expenditure for production facilities and reduced operational costs over time. Furthermore, the use of biomass-derived starting materials insulates the supply chain from the volatility of the petrochemical market, providing a more stable and predictable cost structure for long-term contracts. The robustness of the C-glycosidic bond also reduces the risk of product degradation during storage and transport, minimizing losses due to spoilage and ensuring reliable delivery of high-quality materials to downstream customers.
- Cost Reduction in Manufacturing: The elimination of hazardous organic solvents and heavy metal catalysts significantly lowers the cost of raw materials and waste treatment. By utilizing a streamlined two-step process that operates under mild conditions, energy consumption is drastically reduced compared to traditional high-temperature surfactant syntheses. The ability to regenerate and reuse the organocatalyst further enhances the economic efficiency of the process, driving down the variable cost per kilogram of produced surfactant. These cumulative efficiencies allow for a more competitive pricing strategy while maintaining healthy profit margins, making this technology an attractive option for cost-sensitive markets without sacrificing performance quality.
- Enhanced Supply Chain Reliability: Sourcing renewable carbohydrate feedstocks provides a decentralized and resilient supply base that is less susceptible to geopolitical disruptions affecting oil-dependent industries. The simplicity of the synthesis route reduces the number of critical processing units required, thereby lowering the risk of equipment failure and production downtime. This operational robustness ensures consistent lead times and reliable inventory levels, which is critical for procurement managers managing just-in-time manufacturing schedules. Additionally, the high stability of the final product extends its shelf life, allowing for larger safety stock holdings without the risk of quality degradation, further securing the supply chain against unexpected demand surges.
- Scalability and Environmental Compliance: The aqueous nature of the reaction mixture facilitates easy scale-up using standard stainless steel reactors without the need for specialized corrosion-resistant linings required for acidic or halogenated processes. This compatibility with existing infrastructure accelerates the timeline for commercial deployment and reduces the barrier to entry for manufacturing partners. Moreover, the biodegradable nature of the C-glycoside surfactants aligns with increasingly stringent global environmental regulations, reducing the liability associated with effluent discharge and end-of-life product disposal. This proactive compliance posture enhances brand reputation and opens up markets in regions with strict ecological standards, future-proofing the investment against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these novel amphiphilic compounds. These insights are derived directly from the patent specifications and experimental data, providing clarity on the practical aspects of adopting this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating C-glycoside surfactants into their existing product portfolios or development pipelines.
Q: What distinguishes C-glycoside surfactants from traditional O-glycosides?
A: C-glycosides feature a carbon-carbon bond linking the sugar and lipid moieties, offering superior hydrolytic stability compared to the ether linkage in O-glycosides, ensuring longer shelf life and performance in varying pH conditions.
Q: Is the synthesis process environmentally sustainable?
A: Yes, the patented method utilizes water as the primary solvent and renewable biomass-derived carbohydrates, significantly reducing volatile organic compound (VOC) emissions and hazardous waste generation.
Q: Can these intermediates be scaled for industrial production?
A: The process is designed for scalability, employing simple batch reactions with readily available catalysts and avoiding extreme pressure or temperature conditions, facilitating seamless transition from lab to commercial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-Glycoside Surfactants Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise and infrastructure required to bring complex synthetic pathways like the C-glycoside enone technology to commercial fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and composition of every batch. Our commitment to quality assurance guarantees that the surfactants you receive will perform reliably in your formulations, meeting the high standards expected by global end-users in the personal care and pharmaceutical sectors.
We invite you to collaborate with us to explore the full potential of this sustainable surfactant technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and formulation needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can drive value and innovation in your supply chain. Together, we can accelerate the adoption of greener chemistry solutions that deliver both economic and environmental benefits.
