Advanced Synthesis of Tert-Butoxystyrene for Commercial Scale-Up and High Purity
The chemical industry continuously seeks robust methodologies for producing functionalized styrene derivatives, which serve as critical building blocks in the fabrication of advanced polymers and pharmaceutical intermediates. Patent CN108285409B introduces a groundbreaking approach for synthesizing tert-butoxy substituted styrene and its derivatives, addressing long-standing inefficiencies in traditional manufacturing pathways. This innovation is particularly relevant for organizations seeking a reliable pharmaceutical intermediate supplier capable of delivering complex structures with consistent quality. The core of this technology lies in a strategic two-step sequence that begins with the etherification of halogenated phenols, specifically utilizing m-chlorophenol and isobutene as primary feedstocks. 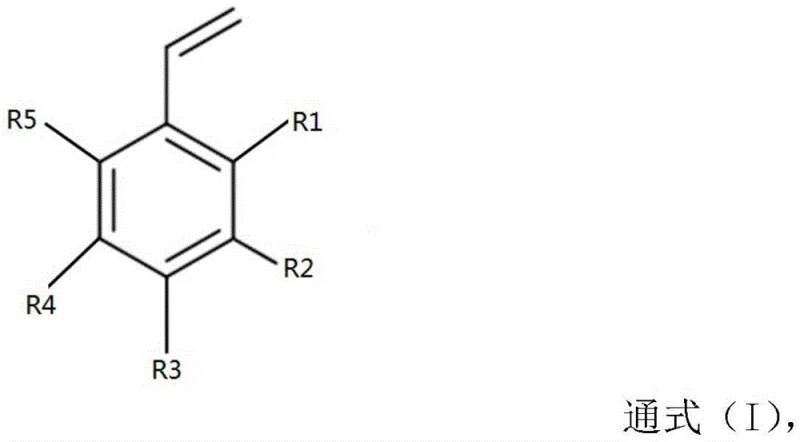 . By shifting the synthetic focus to these abundant starting materials, the process circumvents the reliance on expensive pre-functionalized aromatic halides, thereby establishing a new benchmark for cost reduction in fine chemical manufacturing. The resulting compounds, characterized by the general formula (I), exhibit exceptional stability and reactivity, making them indispensable for downstream applications in medicinal chemistry and material science where precise molecular architecture is paramount.
. By shifting the synthetic focus to these abundant starting materials, the process circumvents the reliance on expensive pre-functionalized aromatic halides, thereby establishing a new benchmark for cost reduction in fine chemical manufacturing. The resulting compounds, characterized by the general formula (I), exhibit exceptional stability and reactivity, making them indispensable for downstream applications in medicinal chemistry and material science where precise molecular architecture is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 3-tert-butoxystyrene has been hindered by significant economic and technical barriers associated with legacy synthetic routes. Conventional methodologies, such as those documented in earlier US and Japanese patents, typically rely on the direct utilization of 3-tert-butylbromo or chlorobenzene as the starting aromatic substrate. These precursors are not only costly due to their specialized nature but also introduce substantial supply chain vulnerabilities, as they are not produced in the same volumes as commodity phenols. Furthermore, traditional catalytic systems often employ iron-based catalysts which, while functional, frequently suffer from lower selectivity and require more rigorous reaction conditions to achieve acceptable conversion rates. This often leads to the formation of complex impurity profiles that necessitate extensive and expensive purification steps, ultimately inflating the cost reduction in electronic chemical manufacturing or pharmaceutical contexts. The reliance on such expensive and less efficient pathways has created a bottleneck for the commercial scale-up of complex intermediates, limiting the ability of manufacturers to respond agilely to market demand fluctuations without compromising on margin or quality standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data leverages a highly efficient etherification strategy followed by a sophisticated cross-coupling reaction. By selecting m-chlorophenol and isobutene as the initial reaction raw materials, the process taps into a supply chain of cheap and easily obtainable chemicals, fundamentally altering the cost structure of the final product. The first step involves a catalytic etherification using methanesulfonic acid, which proceeds under mild thermal conditions to yield 3-tert-butoxy chlorobenzene with high fidelity. This intermediate is then seamlessly transitioned into a Kumada coupling reaction, utilizing a divalent nickel ligand catalyst system, specifically NiCl2(dppp). This transition metal catalysis offers superior control over the reaction trajectory, ensuring that the vinyl group is introduced with high regioselectivity. The shift from expensive halides to abundant phenols, combined with the high catalytic efficiency of the nickel system, represents a paradigm shift that enables reducing lead time for high-purity intermediates while simultaneously enhancing the overall economic viability of the production line for global procurement teams.
Mechanistic Insights into NiCl2(dppp)-Catalyzed Kumada Coupling
The heart of this synthetic innovation lies in the precise orchestration of the catalytic cycle during the Kumada coupling phase, which dictates the purity and yield of the final styrene derivative. The mechanism initiates with the formation of a Grignard reagent from the 3-tert-butoxy chlorobenzene intermediate, a step that requires careful control of magnesium activation and solvent conditions, typically utilizing tetrahydrofuran to stabilize the organometallic species. Once the Grignard reagent is generated, it enters the catalytic cycle mediated by the NiCl2(dppp) complex. The nickel catalyst undergoes oxidative addition with the aryl halide moiety, followed by transmetallation with the Grignard reagent, and finally reductive elimination to forge the carbon-carbon bond with the vinyl chloride. 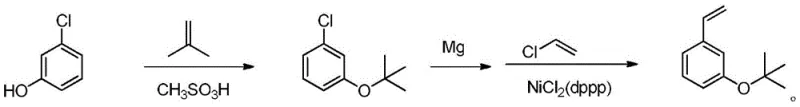 . This specific ligand environment provided by dppp (1,3-bis(diphenylphosphino)propane) is crucial as it stabilizes the nickel center against decomposition and prevents the formation of homocoupling byproducts, which are common pitfalls in nickel-catalyzed cross-couplings. The ability to maintain this catalytic activity at a controlled temperature of 23±3°C demonstrates the robustness of the system, allowing for high-purity styrene derivatives to be produced without the thermal degradation often seen in more aggressive coupling protocols.
. This specific ligand environment provided by dppp (1,3-bis(diphenylphosphino)propane) is crucial as it stabilizes the nickel center against decomposition and prevents the formation of homocoupling byproducts, which are common pitfalls in nickel-catalyzed cross-couplings. The ability to maintain this catalytic activity at a controlled temperature of 23±3°C demonstrates the robustness of the system, allowing for high-purity styrene derivatives to be produced without the thermal degradation often seen in more aggressive coupling protocols.
Impurity control is another critical aspect where this mechanistic approach excels, particularly concerning the stability of the tert-butoxy group and the vinyl functionality. The mild conditions employed in the etherification step (10-20°C) prevent the acid-catalyzed cleavage of the tert-butyl group, a common side reaction that can plague similar syntheses performed at elevated temperatures. Furthermore, the use of specific polymerization inhibitors such as phenothiazine and p-tert-butylcatechol during the workup phase is integral to maintaining the integrity of the vinyl double bond, which is prone to spontaneous polymerization. The rigorous washing protocols with alkaline solutions ensure the complete removal of acidic catalysts and magnesium salts, resulting in a crude product that is already of significant purity before final distillation. This attention to mechanistic detail ensures that the final product meets the stringent purity specifications required by R&D Directors, minimizing the risk of downstream failures in polymerization or medicinal synthesis where trace impurities can act as catalyst poisons or affect biological activity profiles.
How to Synthesize 3-Tert-Butoxystyrene Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters to fully realize the benefits of the patent technology. The procedure begins with the careful introduction of isobutene gas into a solution of m-chlorophenol and methanesulfonic acid in toluene, where temperature control is vital to manage the exothermic nature of the etherification. Following the isolation of the 3-tert-butoxy chlorobenzene intermediate, the process moves to the Grignard formation, which demands an inert atmosphere and precise initiation techniques to ensure complete consumption of the magnesium. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding addition rates and quenching procedures. . Adhering to these protocols ensures that the transition between the etherification and coupling stages is seamless, maximizing the overall yield and minimizing the generation of waste streams that would otherwise burden the environmental compliance team.
- Perform etherification of m-chlorophenol with isobutene using methanesulfonic acid catalyst at 10-20°C to form 3-tert-butoxy chlorobenzene.
- Generate the Grignard reagent from the resulting 3-tert-butoxy chlorobenzene using magnesium turnings in tetrahydrofuran.
- Execute Kumada coupling with vinyl chloride using NiCl2(dppp) catalyst at 23±3°C to yield high-purity 3-tert-butoxystyrene.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis route offers compelling advantages that directly address the pain points of cost volatility and supply continuity in the fine chemical sector. The substitution of expensive specialty halides with commodity phenols and isobutene creates a buffer against raw material price fluctuations, ensuring more predictable budgeting for long-term projects. Additionally, the high catalytic efficiency of the nickel system reduces the loading of expensive transition metals required per kilogram of product, further driving down the variable costs associated with manufacturing. This economic efficiency is complemented by the operational simplicity of the process, which utilizes standard solvents like toluene and THF that are readily available in most chemical manufacturing hubs, thereby reducing the logistical complexity of sourcing specialized reagents.
- Cost Reduction in Manufacturing: The fundamental shift in raw material strategy from high-cost 3-tert-butyl halides to low-cost m-chlorophenol results in a drastic simplification of the bill of materials. By eliminating the need for expensive precursors and reducing the catalyst loading through the use of highly efficient NiCl2(dppp), the overall production cost is significantly optimized. This allows for a more competitive pricing structure without sacrificing margin, providing a tangible financial advantage for procurement managers looking to optimize their spend on high-purity intermediates while maintaining robust quality standards throughout the supply chain.
- Enhanced Supply Chain Reliability: The reliance on m-chlorophenol and isobutene, which are produced on a massive industrial scale for other applications, ensures a stable and continuous supply of starting materials. Unlike niche intermediates that may suffer from production bottlenecks or single-source dependencies, these commodity chemicals are available from multiple global suppliers. This diversification of the supply base significantly mitigates the risk of production stoppages due to raw material shortages, ensuring that the commercial scale-up of complex intermediates can proceed without interruption and meeting the rigorous delivery schedules demanded by downstream pharmaceutical and polymer clients.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily between 10°C and 30°C, reduce the energy consumption associated with heating and cooling large-scale reactors, contributing to a lower carbon footprint for the manufacturing process. Furthermore, the high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying the waste treatment process and ensuring compliance with increasingly stringent environmental regulations. The use of standard distillation techniques for purification, rather than complex chromatographic methods, facilitates easy scale-up from pilot plant to multi-ton production, making this route highly attractive for sustainable and scalable industrial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are designed to clarify the operational feasibility and strategic value of the method for potential partners. Understanding these details is crucial for stakeholders evaluating the integration of this route into their existing manufacturing portfolios or supply networks. . By addressing these key areas, we aim to provide transparency and confidence in the technical robustness and commercial viability of the tert-butoxystyrene production method.
Q: What are the primary cost advantages of this synthesis route?
A: The method utilizes m-chlorophenol and isobutene, which are commodity chemicals significantly cheaper than the 3-tert-butylbromo/chlorobenzene required in conventional methods, drastically reducing raw material expenditure.
Q: How does the catalyst system improve reaction efficiency?
A: The use of NiCl2(dppp) in the Kumada coupling step provides high catalytic efficiency and selectivity under mild conditions (23±3°C), minimizing side reactions and simplifying downstream purification compared to iron-catalyzed alternatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at mild temperatures (10-20°C for etherification, 23±3°C for coupling) and uses readily available solvents like toluene and THF, making it highly scalable and safe for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Tert-Butoxystyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial volume is managed with precision and care. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to verify that every batch of 3-tert-butoxystyrene meets the exacting standards required for pharmaceutical and advanced material applications. Our infrastructure is designed to support the complex needs of modern chemical synthesis, providing a secure and efficient platform for your supply chain requirements.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and application requirements. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this advanced synthesis method can enhance your product portfolio. By partnering with us, you gain access to not just a chemical supplier, but a strategic ally dedicated to driving efficiency and innovation in your manufacturing processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
