Revolutionizing Esterification Efficiency with Recyclable Aminopyridine Derivatives for Industrial Scale-Up
Revolutionizing Esterification Efficiency with Recyclable Aminopyridine Derivatives for Industrial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless pursuit of greener, more cost-effective, and scalable catalytic processes. A significant breakthrough in this domain is documented in patent CN109970635B, which details a novel class of aminopyridine derivatives designed specifically to overcome the longstanding limitations of traditional acylation and esterification catalysts. This technology addresses the critical pain points associated with the production of high-value esters, such as linalyl acetate, a cornerstone ingredient in the flavor and fragrance industry. By introducing a catalyst that combines the high nucleophilic activity of DMAP with the physical properties necessary for easy recovery and reuse, this innovation promises to redefine the economic and operational standards for reliable flavor & fragrance intermediates supplier networks globally.
The core of this invention lies in the structural modification of the pyridine ring, creating a robust catalytic system that operates efficiently under solvent-free or low-solvent conditions. For R&D directors and process chemists, the implications are profound: the ability to achieve high conversion rates while minimizing downstream purification burdens. The patent outlines a versatile synthetic route that allows for the tuning of the catalyst's electronic and steric properties through various substituents on the benzyl groups. This adaptability ensures that the catalyst can be optimized for a wide range of alcohol substrates, from simple primary alcohols to sterically hindered tertiary alcohols, making it a powerful tool for cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the esterification of tertiary alcohols like linalool has been fraught with challenges due to the inherent instability of the substrate under acidic conditions. Traditional acid-catalyzed methods, while effective for primary alcohols, often lead to significant degradation when applied to tertiary systems. As illustrated in the reaction scheme below, protonic acids or Lewis acids can trigger unwanted side reactions such as dehydration, isomerization, and skeletal rearrangement.
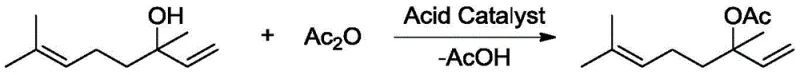
These side reactions not only reduce the overall yield of the desired ester but also generate a complex mixture of by-products like geranyl acetate and myrcene, which are notoriously difficult to separate from the target molecule. Furthermore, while basic catalysts like pyridine or DMAP offer a milder alternative, they come with their own set of economic and logistical drawbacks. The use of DMAP, for instance, typically requires high catalyst loadings and often necessitates the addition of stoichiometric amounts of auxiliary bases to neutralize the acetic acid by-product generated during the reaction.

This reliance on excess reagents increases the raw material costs and complicates the waste stream management. Moreover, DMAP is relatively expensive and, due to its lower boiling point and high solubility in organic phases, is difficult to recover and recycle from the reaction mixture. This single-use nature of conventional nucleophilic catalysts represents a significant inefficiency in large-scale commercial scale-up of complex esters, driving up the cost of goods sold and limiting the sustainability profile of the manufacturing process.
The Novel Approach
The technology disclosed in CN109970635B presents a paradigm shift by introducing aminopyridine derivatives that retain the exceptional nucleophilic catalytic activity of DMAP while possessing distinct physical properties that facilitate recovery. The key innovation is the introduction of bulky benzyl groups at the 4-position of the pyridine ring. This structural modification significantly increases the molecular weight and boiling point of the catalyst without compromising its ability to activate the acylating agent. Consequently, the catalyst remains in the reaction pot (still residue) while the lower-boiling ester product is distilled off, enabling a seamless separation process.
This approach effectively decouples the catalytic cycle from the product isolation step, allowing the catalyst to be recycled multiple times with minimal loss of activity. The patent data demonstrates that these derivatives can catalyze the esterification of linalool with acetic anhydride to achieve yields exceeding 95%, comparable to or better than DMAP, but with the added benefit of reusability. This capability directly translates to reducing lead time for high-purity flavor & fragrance intermediates by simplifying the workup procedure and eliminating the need for extensive chromatographic purification to remove catalyst residues.
Mechanistic Insights into Nucleophilic Catalysis with Bulky Aminopyridines
To understand the efficacy of this new catalyst class, one must examine the mechanistic pathway of nucleophilic catalysis in acylation reactions. The aminopyridine derivative acts as a nucleophilic catalyst, attacking the carbonyl carbon of the acetic anhydride to form a highly reactive N-acylpyridinium intermediate. This intermediate is significantly more electrophilic than the original anhydride, thereby lowering the activation energy for the subsequent attack by the alcohol substrate. The bulky benzyl substituents on the nitrogen atom play a dual role: electronically, they donate electron density to the pyridine ring, enhancing its nucleophilicity, similar to the dimethylamino group in DMAP.
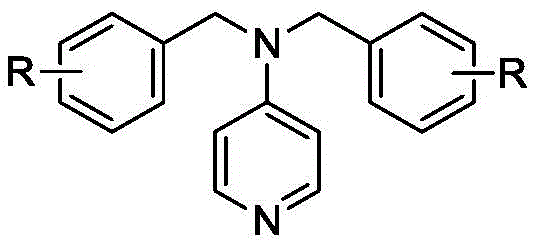
Sterically, however, these groups provide the necessary bulk to prevent the catalyst from co-distilling with the product. The mechanism proceeds through a classic acyl-transfer pathway where the alcohol attacks the N-acyl species, releasing the ester product and regenerating the free amine catalyst. Crucially, the stability of the N-acyl intermediate is balanced such that it reacts rapidly with the alcohol but does not decompose via hydrolysis or other side pathways under the reaction conditions. This precise tuning of reactivity ensures that even acid-sensitive substrates like linalool are converted cleanly without undergoing the dehydration or rearrangement typical of acid catalysis.
Furthermore, the impurity profile is tightly controlled because the reaction conditions are inherently basic and mild. Unlike acid catalysis, which promotes carbocation formation leading to terpenes rearrangement, this basic nucleophilic pathway preserves the integrity of the allylic double bonds in the substrate. The result is a product stream with high chemical purity, reducing the burden on downstream refining columns. For quality control teams, this means a more consistent impurity spectrum, facilitating easier validation and compliance with stringent stringent purity specifications required for food and fragrance applications.
How to Synthesize 4-Dibenzylaminopyridine Efficiently
The synthesis of the catalyst itself is designed for industrial practicality, utilizing readily available starting materials and straightforward reaction conditions. The process involves the N-benzylation of 4-aminopyridine using benzyl halides in the presence of a strong base. This transformation is robust and scalable, making it an ideal candidate for in-house production or procurement from a specialized chemical partner. The following section outlines the standardized protocol derived from the patent examples, ensuring reproducibility and safety during scale-up.
- Prepare the reaction vessel under nitrogen atmosphere and dissolve 4-aminopyridine in an aprotic solvent such as tetrahydrofuran.
- Add a strong base reagent like sodium hydride in batches at room temperature to deprotonate the amine, stirring for approximately one hour.
- Heat the mixture to 60-80°C and dropwise add the benzyl halide reactant, maintaining the temperature for 5-10 hours to complete the benzylation.
Following the reaction, the crude product is typically purified via recrystallization or simple washing steps, yielding the catalyst in high purity (often >98%). The simplicity of this synthesis route underscores the economic viability of the technology; there are no exotic reagents or complex multi-step sequences required. This ease of access ensures that the supply of the catalyst can be maintained consistently, supporting continuous manufacturing operations without the risk of bottlenecks associated with scarce specialty chemicals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aminopyridine derivative technology offers compelling strategic advantages that extend beyond mere technical performance. The primary value driver is the drastic reduction in operating costs associated with catalyst consumption. Since the catalyst can be recovered from the still bottoms and reused for multiple batches—potentially over ten cycles as indicated in the patent data—the effective cost per kilogram of product is significantly lowered. This eliminates the recurring expense of purchasing fresh DMAP for every run, transforming a variable cost into a manageable fixed asset investment.
- Cost Reduction in Manufacturing: The economic model shifts favorably because the high boiling point of the catalyst allows for thermal separation rather than chemical quenching or extraction. In traditional DMAP processes, removing the catalyst often requires acidic washes followed by basic neutralization, generating substantial aqueous waste streams that require treatment. By contrast, the distillation-based recovery of this novel catalyst minimizes solvent usage and waste generation. This reduction in effluent treatment costs, combined with the savings on raw catalyst materials, results in substantial overall cost optimization for the manufacturing facility.
- Enhanced Supply Chain Reliability: The raw materials for synthesizing this catalyst—4-aminopyridine and substituted benzyl chlorides or bromides—are commodity chemicals with stable global supply chains. Unlike proprietary immobilized enzymes or complex organometallic complexes that may rely on single-source suppliers, the precursors for this technology are widely available from multiple vendors. This diversification of the supply base mitigates the risk of disruption and ensures business continuity. Furthermore, the long shelf-life and stability of the solid catalyst simplify inventory management and logistics.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is superior. The ability to run the esterification under solvent-free conditions or with minimal solvent reduces the volatile organic compound (VOC) emissions associated with the process. Additionally, the avoidance of strong mineral acids eliminates the corrosion risks to reactor vessels and the hazards associated with handling corrosive waste. These factors streamline the regulatory approval process for new facilities and support corporate sustainability goals by reducing the carbon footprint of the chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aminopyridine catalyst technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for decision-makers evaluating this process for adoption.
Q: How does this aminopyridine derivative improve upon traditional DMAP catalysts?
A: Unlike DMAP, which is difficult to recover and often requires stoichiometric amounts of additional base, this novel derivative has a significantly higher molecular weight and boiling point. This physical property allows it to remain in the still residue during product distillation, enabling multiple recycling cycles without significant loss of catalytic activity.
Q: Can this catalyst be used for acid-sensitive substrates like tertiary alcohols?
A: Yes, the catalyst operates under mild basic conditions, avoiding the harsh acidic environments that typically cause dehydration, isomerization, and rearrangement in sensitive tertiary alcohols like linalool, thereby ensuring high product purity and yield.
Q: What are the primary raw materials required for synthesizing this catalyst?
A: The synthesis relies on commercially available and cost-effective starting materials, specifically 4-aminopyridine and various substituted benzyl halides, reacting in common aprotic solvents with standard inorganic bases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Dibenzylaminopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your move towards this efficient esterification method is seamless and risk-free. Our state-of-the-art facilities are equipped to handle the synthesis of complex aminopyridine derivatives with rigorous QC labs dedicated to maintaining stringent purity specifications, guaranteeing that every batch meets the exacting standards required for high-value fragrance and pharmaceutical applications.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this catalyst into your existing production lines. Whether you require custom synthesis of specific substituted derivatives or a comprehensive Customized Cost-Saving Analysis for your current esterification processes, our experts are ready to assist. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, taking the first step towards optimizing your supply chain and achieving superior economic performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
