Revolutionizing Dysideanone B Production: A Technical Breakdown for Global Pharmaceutical Supply Chains
Introduction to Patent CN115197058A and Strategic Significance
The pharmaceutical industry is constantly seeking robust supply chains for complex natural products with potent biological activities, and the recent disclosure in patent CN115197058A represents a pivotal advancement in the availability of Dysideanone B. This specific intellectual property details the first total synthesis of Dysideanone B, a sesquiterpene quinone marine natural product isolated from Dysidea avara, which has demonstrated remarkable antitumor activity against human cervical cancer HeLa and liver cancer HepG2 cell lines. The strategic value of this patent lies not merely in the academic achievement of total synthesis, but in the development of a scalable, efficient pathway that overcomes the historical inability of research groups to access this molecule due to its structural complexity. For R&D directors and procurement specialists, this technology offers a reliable entry point into a class of compounds previously restricted to minute isolation quantities, thereby unlocking potential for drug discovery programs focused on protein tyrosine phosphatase 1B (PTP1B) inhibition and broader oncology applications.
Furthermore, the patent elucidates a series of analogues defined by formulas (II) and (III), expanding the chemical space available for structure-activity relationship (SAR) studies beyond the natural product itself. The ability to synthesize these analogues efficiently means that pharmaceutical companies can now explore derivative libraries without being bottlenecked by natural extraction limitations. This technological breakthrough transforms Dysideanone B from a rare marine curiosity into a viable candidate for preclinical development, provided that the manufacturing process can be translated from laboratory scale to industrial production. The following analysis dissects the technical merits of this synthesis, highlighting why it serves as a cornerstone for establishing a reliable Dysideanone B supplier network capable of meeting the rigorous demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
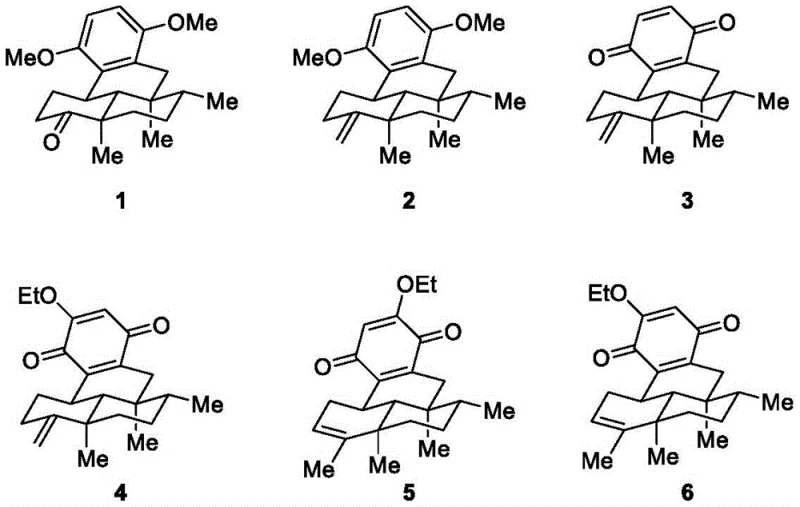
Prior to the innovations described in this patent, the chemical community faced a significant void in the availability of Dysideanone B, primarily due to the extreme difficulty associated with its molecular architecture. Conventional methods for accessing similar sesquiterpene quinones often relied on semi-synthesis from abundant natural precursors or lengthy linear sequences that suffered from poor overall yields and stereochemical control issues. The specific challenge with Dysideanone B is its unprecedented 6/6/6/6 fused tetracyclic all-carbon skeleton, which creates a highly crowded molecular environment containing five consecutive chiral centers. Traditional approaches to building such congested quaternary carbon centers often require harsh conditions, expensive chiral auxiliaries, or resolution steps that drastically reduce atom economy and increase waste generation. Moreover, the lack of any prior completed total synthesis indicated that existing methodologies were fundamentally insufficient to navigate the steric hindrance and electronic constraints inherent to this target, leaving potential therapeutic applications unexplored due to material scarcity.
The Novel Approach
The novel approach presented in patent CN115197058A circumvents these historical barriers by employing a convergent strategy rooted in the chemistry of Wieland-Miescher ketone derivatives. This methodology leverages a concise eight-step sequence that efficiently constructs the complex tetracyclic core through a combination of alkylation, methylenation, and a critical radical cyclization step. Unlike traditional routes that might struggle with the formation of the fourth ring, this process utilizes a tin-mediated radical cascade to close the ring system with high fidelity, establishing the necessary quaternary centers in a single operation. Furthermore, the route demonstrates exceptional versatility by allowing for the synthesis of multiple analogues through simple modifications of the starting materials or reaction conditions. The identification that the ethoxy group can be installed directly from the ethanol solvent during the final oxidative step is a particular stroke of genius, simplifying the functionalization process and eliminating the need for separate etherification reagents. This streamlined logic results in a shorter reaction route with higher total yield, making it uniquely suited for the mass synthesis required by commercial supply chains.
Mechanistic Insights into Radical Cyclization and Oxidative Substitution
A deep understanding of the reaction mechanisms underpinning this synthesis is crucial for R&D teams aiming to optimize the process for large-scale manufacturing. The heart of this synthetic strategy is the radical cyclization step, where tricyclic ketone 12 is converted into the tetracyclic ketone 1 using tri-n-butyltin hydride and AIBN. This transformation is mechanistically fascinating as it involves the generation of a carbon-centered radical that undergoes an intramolecular addition to an aromatic ring, effectively forging the new carbon-carbon bond that completes the tetracyclic framework. The success of this step relies heavily on the precise conformational alignment of the substrate, which is pre-organized by the rigid tricyclic precursor. By controlling the radical kinetics, the process avoids unwanted side reactions such as polymerization or reduction, ensuring that the sterically demanding quaternary center is formed with the correct stereochemistry. This mechanistic precision is vital for maintaining the biological integrity of the final product, as the spatial arrangement of the chiral centers is directly linked to the compound's antitumor potency.
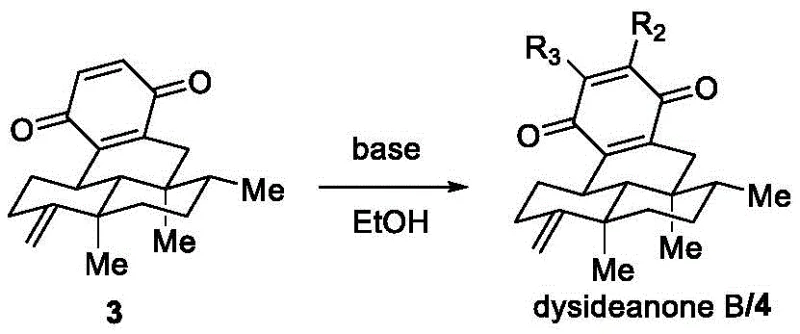
Equally important is the final mechanistic insight regarding the formation of the ethoxy-substituted quinone. The patent reveals that treating the tetracyclic quinone 3 with a base in ethanol under an oxygen atmosphere leads to the incorporation of the ethoxy group. Mechanistically, this likely proceeds via a nucleophilic attack of the ethoxide ion (generated in situ from ethanol and base) on the quinone system, facilitated by the electron-deficient nature of the oxidized ring. The presence of oxygen serves to re-oxidize any intermediate hydroquinone species back to the quinone state, driving the equilibrium towards the desired product. This solvent-participating mechanism is a powerful tool for process chemists, as it turns a common solvent into a reagent, thereby reducing the number of distinct chemical inputs required. Understanding this pathway allows for fine-tuning of the base strength and oxygen flow to maximize the ratio of Dysideanone B versus its analogue, providing a handle for impurity control that is essential for meeting stringent pharmaceutical purity specifications.
How to Synthesize Dysideanone B Efficiently
The synthesis of Dysideanone B described in this patent offers a clear roadmap for laboratories aiming to produce this valuable intermediate for biological testing or further derivatization. The process begins with readily available starting materials and proceeds through a series of well-defined transformations that have been optimized for yield and reproducibility. Key to the success of this route is the careful control of reaction temperatures and stoichiometry, particularly during the radical cyclization and final oxidation steps where side reactions can compete. By adhering to the specific conditions outlined, such as the use of THF for alkylation and toluene for the radical step, manufacturers can ensure consistent quality of the intermediate batches. The detailed experimental procedures provided in the patent serve as a robust foundation for scaling up, although process safety assessments regarding the use of tin reagents and oxidizers would be a standard prerequisite for industrial implementation. For those seeking to implement this chemistry, the following standardized guide outlines the critical operational phases.
- Initiate the synthesis by alkylating the Wieland-Miescher ketone derivative with a specific bromide to construct the initial tricyclic framework.
- Perform a methylenation reaction followed by hydrolysis and hydrogenation to prepare the substrate for the critical ring-closing step.
- Execute a radical cyclization using tributyltin hydride to form the congested tetracyclic core, followed by oxidation and ethoxy group installation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience. The primary advantage lies in the significant reduction of manufacturing complexity, as the short synthetic sequence minimizes the number of unit operations required to reach the final API intermediate. Fewer steps translate directly to lower labor costs, reduced equipment occupancy time, and decreased consumption of solvents and utilities, all of which contribute to a more cost-effective production model. Furthermore, the reliance on cheap and readily available raw materials, such as the Wieland-Miescher ketone derivative and common solvents like ethanol and toluene, mitigates the risk of supply disruptions associated with exotic or proprietary reagents. This accessibility ensures that the supply chain remains robust even in volatile market conditions, providing a stable source of material for long-term drug development projects.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive chiral catalysts and the high efficiency of key bond-forming steps. By achieving high yields in critical transformations, such as the 98% yield in the hydrolysis step and 87% in the Wittig olefination, the process minimizes material loss and the need for extensive recycling or reprocessing. Additionally, the use of ethanol as both solvent and reagent in the final step simplifies the bill of materials, removing the need for specialized alkylating agents. This streamlined approach reduces the overall cost of goods sold (COGS), making the commercial production of Dysideanone B analogues financially attractive for generic drug manufacturers or specialty chemical suppliers looking to enter the oncology intermediate market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are widely sourced from multiple global vendors. Unlike processes dependent on single-source biocatalysts or rare earth metals, this synthetic route utilizes standard organic reagents like tributyltin hydride and silver oxide, which, while requiring careful handling, are established items in the fine chemical inventory. The robustness of the reaction conditions, which operate at mild temperatures ranging from room temperature to moderate heating, reduces the energy burden on manufacturing facilities and lowers the risk of thermal runaways. This operational stability ensures that production schedules can be met consistently, reducing lead times for high-purity pharmaceutical intermediates and allowing downstream partners to plan their formulation and clinical trial timelines with greater confidence.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed to be adaptable to larger reactor volumes without significant re-engineering. The workup procedures involve standard extractions and column chromatography, techniques that are easily translated to preparative HPLC or crystallization protocols at the tonne scale. While the use of tin reagents necessitates appropriate waste management strategies to meet environmental regulations, the high atom economy of the overall sequence means less waste is generated per kilogram of product compared to less efficient routes. The ability to produce sufficient raw materials for biological evaluation and SAR studies positions this method as a sustainable solution for the long-term supply of Dysideanone B, aligning with the green chemistry principles increasingly demanded by regulatory bodies and corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the potential of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed technical disclosures within the patent. These answers address common concerns regarding structural integrity, synthetic flexibility, and the origin of specific functional groups which are critical for regulatory filing and quality control. Understanding these nuances helps in defining the critical quality attributes (CQAs) for the manufactured intermediate and ensures that the supplied material meets the rigorous standards required for preclinical and clinical investigations. The following insights are derived directly from the experimental data and mechanistic proposals provided in the intellectual property documentation.
Q: What is the key structural challenge in synthesizing Dysideanone B?
A: The primary challenge lies in constructing the unprecedented 6/6/6/6 fused tetracyclic all-carbon skeleton which contains five consecutive chiral centers, two of which are quaternary carbon centers, making the molecular backbone extremely crowded.
Q: How is the ethoxy group introduced in the final structure of Dysideanone B?
A: The patent reveals that the ethoxy group is derived directly from the solvent ethanol during the final reaction step, where the quinone precursor reacts with a base under an oxygen atmosphere.
Q: Why is this synthetic route considered superior for commercial manufacturing?
A: This route is superior because it utilizes cheap and readily available starting materials like Wieland-Miescher ketone derivatives, features mild reaction conditions, and achieves a short synthetic sequence with high yields in key transformation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dysideanone B Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN115197058A for the development of next-generation anticancer therapeutics. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of sesquiterpene quinone synthesis, including the safe management of radical reactions and oxidative processes. We are committed to delivering Dysideanone B and its analogues with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify structural identity and impurity profiles. By partnering with us, you gain access to a supply chain that is not only technically competent but also deeply aligned with your timeline and quality objectives.
We invite you to engage with our technical procurement team to discuss how this novel synthesis can be integrated into your drug development pipeline. Whether you require custom synthesis of specific analogues for SAR studies or bulk manufacturing of the parent compound for toxicology batches, we are prepared to provide a Customized Cost-Saving Analysis tailored to your project's unique constraints. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of our manufacturing capabilities with your regulatory strategy. Let us collaborate to accelerate the delivery of this promising anticancer candidate to the patients who need it most, leveraging our expertise to overcome the challenges of complex natural product synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
