Scalable Total Synthesis of Penicillide Racemate for Advanced Pharmaceutical Applications
Scalable Total Synthesis of Penicillide Racemate for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methods for producing bioactive natural products and their analogs, particularly those exhibiting potent biological activities such as cholesteryl ester transfer protein (CETP) inhibition. Patent CN103044383A discloses a highly efficient, convergent synthetic methodology for preparing the racemate of Penicillide, a natural product originally isolated from fungal sources. This innovative approach addresses the critical bottlenecks associated with natural extraction, such as low abundance and complex purification, by establishing a fully synthetic route that relies on readily available commercial starting materials. By dissecting the complex molecular architecture into two manageable fragments, Compound 12 and Compound 20, the process enables precise control over stereochemistry and functional group installation, offering a viable pathway for the commercial scale-up of complex natural product analogs. For R&D teams and procurement specialists alike, this technology represents a significant leap forward in securing a stable supply of high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Penicillide and related bioactive metabolites has been plagued by severe supply chain inefficiencies inherent to natural product isolation. The content of Penicillide in fungal plants is extremely low, making the extraction process economically unviable for large-scale applications and resulting in inconsistent batch-to-batch quality. Furthermore, traditional semi-synthetic approaches often suffer from linear reaction sequences that accumulate impurities at every stage, leading to diminished overall yields and requiring extensive, costly purification efforts. The reliance on specific biological sources also introduces volatility into the reliable pharmaceutical intermediate supplier network, as fermentation conditions and harvest times can drastically alter output. These factors collectively hinder the comprehensive research and development of Penicillide derivatives for therapeutic use, creating an urgent demand for a versatile, fully synthetic alternative that decouples production from biological constraints.
The Novel Approach
The methodology outlined in the patent data revolutionizes the production landscape by employing a convergent synthesis strategy that utilizes cheap and easy-to-get starting materials, specifically 2,3-dihydroxybenzaldehyde and 5-amino-2-methylphenol. This route is characterized by its operational simplicity, high selectivity, and the use of common reagents that are inexpensive and widely accessible in the global chemical market. Instead of a lengthy linear progression, the synthesis constructs two distinct aromatic fragments independently before joining them via a robust ether linkage, a tactic that significantly mitigates risk and enhances throughput. The process demonstrates excellent yield and selectivity across multiple steps, including lithiation-carboxylation and copper-catalyzed coupling, ensuring that the final high-purity Penicillide racemate is obtained with minimal waste. This strategic design not only simplifies separation but also lays the groundwork for substantial cost reduction in API manufacturing by eliminating the need for rare precursors.
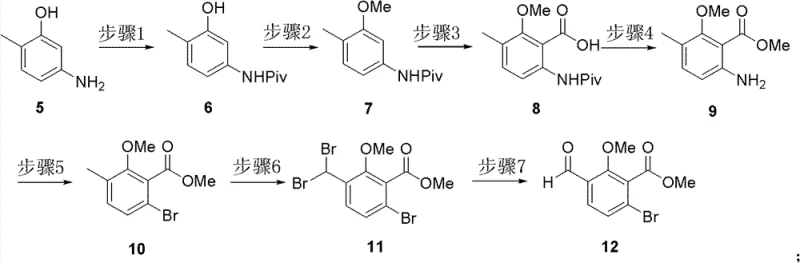
The synthesis of the first key fragment, Compound 12, exemplifies the efficiency of this novel approach. Starting from 5-amino-2-methylphenol, the route employs a series of well-controlled transformations including pivaloyl protection, methylation, and directed ortho-lithiation followed by carboxylation. The subsequent conversion of the amino group to a bromide via diazotization allows for further functionalization, ultimately yielding the aldehyde-bearing aromatic core. This sequence highlights the precision required to install substituents in specific positions without affecting sensitive functional groups, a challenge often faced in pharmaceutical intermediates production. The use of standard workup procedures such as extraction and silica gel chromatography ensures that the intermediate meets stringent quality standards before proceeding to the coupling stage.
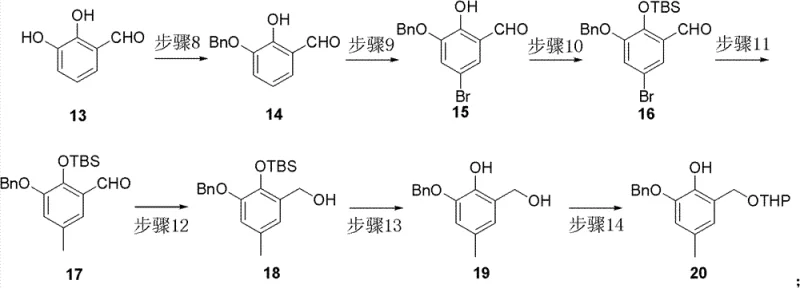
Parallel to the first fragment, the second critical building block, Compound 20, is derived from 2,3-dihydroxybenzaldehyde through a carefully orchestrated series of protection and substitution reactions. The strategy involves selective benzylation of one phenolic hydroxyl group, followed by regioselective bromination and silyl protection of the remaining phenol. A palladium-catalyzed cross-coupling reaction introduces a methyl group, setting the stage for the reduction of the aldehyde to a primary alcohol. The final protection of this alcohol as a tetrahydropyranyl (THP) ether completes the fragment, rendering it stable for the subsequent coupling reaction. This modular approach allows for the independent optimization of each fragment, ensuring that any issues with yield or purity can be resolved before the valuable fragments are combined, thereby maximizing the overall efficiency of the commercial scale-up of complex polymer additives or pharmaceutical precursors.
Mechanistic Insights into Copper-Catalyzed Etherification and Macrolactonization
The heart of this synthetic achievement lies in the coupling of the two aromatic fragments via a copper-catalyzed etherification, a reaction that forms the biaryl ether backbone essential to the Penicillide structure. In the presence of a base such as DMAP and a copper source like copper powder or cupric oxide, the phenolic oxygen of one fragment attacks the aryl bromide of the other, forging the critical C-O bond. This Ullmann-type coupling is notoriously challenging due to potential side reactions, but the specific conditions described—utilizing acetonitrile as a solvent and optimized stoichiometry—ensure high conversion and selectivity. Following the coupling, the removal of protecting groups reveals the necessary functionalities for ring closure. The subsequent macrolactonization step, mediated by activated esters or similar coupling agents, closes the macrocyclic ring, creating the core scaffold of the natural product. Understanding these mechanistic nuances is vital for R&D directors aiming to replicate or modify the process for analog synthesis.
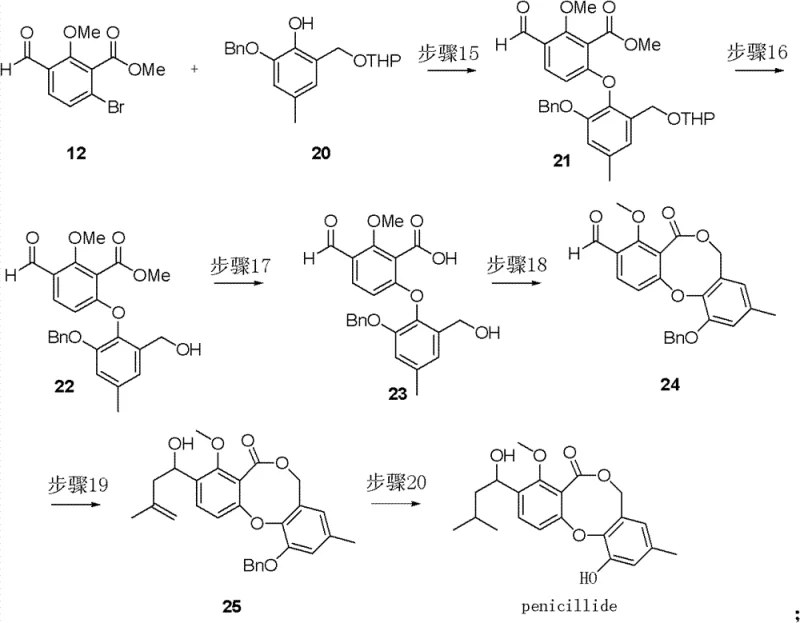
Following the formation of the macrocyclic core, the synthesis proceeds with the installation of the side chain via a Grignard addition to the aldehyde moiety, introducing the requisite carbon framework with high fidelity. The final step involves a transition metal-catalyzed hydrogenation, typically using palladium on carbon, to remove benzyl protecting groups and saturate any remaining unsaturation, yielding the final Penicillide racemate. This final deprotection is crucial as it unveils the free phenolic hydroxyl groups responsible for the molecule's biological activity. The entire sequence is designed to minimize the formation of difficult-to-remove impurities, a key consideration for maintaining stringent purity specifications in pharmaceutical grade materials. The robustness of these reactions underpins the feasibility of transferring this laboratory-scale success to multi-ton production environments.
How to Synthesize Penicillide Racemate Efficiently
The execution of this synthetic route requires meticulous attention to reaction conditions, particularly regarding temperature control during lithiation and the exclusion of moisture during organometallic steps. The patent provides a detailed roadmap that guides chemists through twenty distinct steps, each optimized for yield and ease of purification. From the initial protection of amines and phenols to the final hydrogenolysis, every operation is designed to be reproducible and scalable. For technical teams looking to implement this process, the availability of standardized protocols for each transformation reduces the risk of failure and accelerates the timeline from bench to pilot plant. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive framework for achieving consistent results.
- Synthesize the left-hand aromatic fragment (Compound 12) starting from 5-amino-2-methylphenol via protection, lithiation-carboxylation, and diazotization-bromination sequences.
- Prepare the right-hand aromatic fragment (Compound 20) from 2,3-dihydroxybenzaldehyde through selective benzylation, bromination, silyl protection, and methylation.
- Couple the two fragments via copper-catalyzed etherification, followed by deprotection, macrolactonization, Grignard addition, and final hydrogenation to yield Penicillide racemate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic methodology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from unpredictable natural extraction to a defined chemical synthesis, organizations can secure a consistent supply of Penicillide intermediates, insulating themselves from the volatility of agricultural or fermentation-based sourcing. The reliance on commodity chemicals rather than bespoke reagents means that raw material costs are inherently lower and more stable, facilitating better budget forecasting and cost reduction in electronic chemical manufacturing or pharmaceutical sectors. Furthermore, the simplicity of the workup procedures, which primarily involve extraction and filtration, reduces the burden on waste management systems and lowers the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of abundant copper salts and the use of inexpensive starting materials like 2,3-dihydroxybenzaldehyde drastically lowers the bill of materials. Additionally, the high selectivity of the reactions minimizes the formation of byproducts, reducing the need for costly and time-consuming purification steps such as preparative HPLC. This streamlined approach translates to significant operational savings, allowing for more competitive pricing of the final active pharmaceutical ingredient without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing starting materials that are commercially available in bulk quantities ensures that production schedules are not disrupted by raw material shortages. The synthetic route's robustness means that lead times can be accurately predicted and shortened, providing a distinct advantage in fast-paced drug development cycles. By establishing a domestic or regional supply chain for these key intermediates, companies can mitigate geopolitical risks and logistics delays, ensuring uninterrupted access to critical materials for clinical and commercial needs.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram and ton-scale production without fundamental changes to the chemistry. The use of common solvents and reagents simplifies regulatory compliance regarding hazardous materials, and the efficient atom economy of the convergent strategy reduces the volume of chemical waste generated. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Penicillide synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity for stakeholders evaluating the feasibility of this route. Understanding these details is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the key advantages of this convergent synthesis route for Penicillide?
A: The primary advantage lies in the use of commercially available and inexpensive starting materials like 2,3-dihydroxybenzaldehyde and 5-amino-2-methylphenol. The convergent strategy allows for the independent optimization of two key fragments before coupling, significantly improving overall yield and purity compared to linear synthesis methods.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes common reagents such as sodium bicarbonate, methyl iodide, and copper powder, avoiding exotic catalysts. The purification steps rely on standard extraction and column chromatography techniques which are easily adaptable to industrial preparative chromatography or crystallization processes.
Q: How does this method address the supply chain challenges of natural product extraction?
A: Natural extraction of Penicillide from fungal sources is limited by extremely low content and difficult separation. This total synthesis method bypasses biological variability, ensuring a consistent, reliable supply of high-purity material independent of seasonal or fermentation constraints, thereby securing the supply chain for downstream drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Penicillide Racemate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex pharmaceutical intermediates like Penicillide racemate. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the highest industry standards for safety and efficacy. Our expertise in handling sensitive chemistries, such as the organometallic and coupling reactions described in this patent, allows us to deliver high-quality materials that accelerate your drug development timelines.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall expenditure. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities align with your strategic goals for reducing lead time for high-purity pharmaceutical intermediates. Together, we can bring innovative therapies to market faster and more efficiently.
