Revolutionizing Agrochemical Intermediate Production: Scalable Rhodium-Catalyzed Synthesis of High-Purity Furanoisoquinolinone Compounds for Global Supply Chains
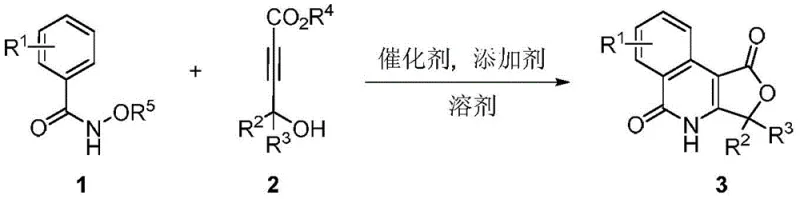
Patent CN108148070B introduces a groundbreaking one-pot tandem synthesis method for furanone isoquinolone compounds, representing a significant advancement in the field of agrochemical intermediate manufacturing. This innovative approach utilizes N-alkoxy aryl formamide compounds and 4-hydroxy-2-alkynoate compounds to construct complex heterocyclic structures through a rhodium-catalyzed cascade reaction under mild conditions. The methodology addresses longstanding challenges in traditional synthesis routes by eliminating multiple isolation steps while maintaining excellent regioselectivity and broad substrate compatibility. With reaction temperatures ranging from 80-120°C and operational simplicity that enables straightforward scale-up, this patent provides a commercially viable pathway for producing high-value agrochemical intermediates with superior purity profiles. The process demonstrates remarkable versatility across diverse substituent patterns while maintaining consistent high yields, making it particularly valuable for manufacturers seeking reliable production of bioactive heterocyclic compounds for agricultural applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for furanone isoquinolone compounds have historically suffered from multiple critical limitations that hinder commercial viability and scalability. Conventional approaches typically require multi-step procedures with intermediate isolations, significantly increasing production costs and reducing overall process efficiency. These methods often employ harsh reaction conditions, including elevated temperatures exceeding 150°C or highly acidic/basic environments that necessitate specialized equipment and stringent safety protocols. Furthermore, the narrow substrate scope of existing methodologies restricts their applicability to only specific molecular architectures, limiting the structural diversity of final products. The poor regioselectivity observed in many conventional syntheses results in complex product mixtures that require extensive purification, leading to substantial yield losses and increased solvent consumption. Additionally, the reliance on stoichiometric amounts of expensive reagents rather than catalytic systems creates significant economic barriers for large-scale manufacturing, while the generation of hazardous byproducts complicates waste management and environmental compliance.
The Novel Approach
The patented methodology overcomes these limitations through an elegant one-pot tandem reaction that simultaneously constructs both the nitrogen and oxygen heterocyclic components in a single operation. By utilizing N-alkoxy aryl formamide compounds and 4-hydroxy-2-alkynoate compounds as starting materials, the process eliminates the need for intermediate isolations while maintaining excellent control over regioselectivity. The rhodium-catalyzed system operates under remarkably mild conditions (80-120°C) in common solvents such as DME or methanol, significantly reducing energy consumption and equipment requirements compared to traditional high-temperature processes. The broad substrate compatibility demonstrated across various substituent patterns—including halogens, alkyl groups, and aryl moieties—enables the production of diverse furanone isoquinolone derivatives without process modification. Crucially, the catalytic nature of the rhodium system minimizes reagent costs while the high yields and simplified workup procedure substantially reduce waste generation and purification complexity, making this approach uniquely suited for industrial-scale implementation.
Mechanistic Insights into Rhodium-Catalyzed Tandem Cyclization
The catalytic cycle begins with oxidative addition of the rhodium catalyst [RhCp*Cl2]2 to the alkyne moiety of the 4-hydroxy-2-alkynoate compound, forming a vinyl-rhodium intermediate that undergoes nucleophilic attack by the hydroxyl group to generate a key enol ether species. This intermediate then coordinates with the N-alkoxy aryl formamide through the carbonyl oxygen, facilitating intramolecular cyclization via C-H activation at the ortho position of the aryl ring. The resulting rhodacycle undergoes reductive elimination to form the initial heterocyclic structure, followed by a second cyclization event where the carbonyl oxygen attacks the activated alkyne to close the furanone ring system. The precise control of regioselectivity stems from the steric and electronic properties of the rhodium catalyst, which directs the cyclization pathway to favor the desired isomer while suppressing potential side reactions. The mild reaction conditions prevent decomposition of sensitive functional groups commonly found in agrochemical intermediates.
Impurity control is achieved through multiple mechanisms inherent to this tandem process. The one-pot nature eliminates intermediate purification steps where impurities could accumulate, while the high regioselectivity minimizes formation of structural isomers that would require difficult separation. The rhodium catalyst's ability to operate effectively at moderate temperatures prevents thermal degradation pathways that typically generate byproducts in conventional syntheses. Additionally, the use of potassium fluoride as an additive promotes clean conversion by facilitating proton transfer steps without introducing new impurities. The solvent system (DME or similar) provides optimal polarity for both reaction stages while enabling straightforward product isolation through simple filtration and chromatography. This combination of factors results in consistently high purity profiles without requiring specialized purification techniques, making the process particularly attractive for manufacturing agrochemical intermediates with strict quality specifications.
How to Synthesize Furanoisoquinolinone Efficiently
This patented synthesis route represents a significant advancement in manufacturing efficiency for furanoisoquinolinone compounds, offering a streamlined alternative to traditional multi-step approaches. The methodology leverages readily available starting materials and common laboratory equipment while maintaining exceptional control over product quality and yield consistency. By eliminating intermediate isolations and operating under mild conditions, this process reduces both production time and resource consumption compared to conventional methods. The following standardized procedure provides a reliable framework for implementing this technology at scale, with detailed operational parameters optimized for consistent commercial production outcomes.
- Dissolve N-alkoxy aryl formamide compound (1 equivalent) and 4-hydroxy-2-alkynoate compound (1-2 equivalents) in appropriate solvent (DME, methanol, or similar)
- Add rhodium catalyst [RhCp*Cl<sub>2</sub>]<sub>2</sub> (0.025 equivalents) and additive (cesium acetate, KF, or similar at 1-2 equivalents) under nitrogen atmosphere
- Stir reaction mixture at 80-120°C for 12 hours, then cool to room temperature before filtration and chromatographic purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in agrochemical intermediate supply chains by delivering a more robust and economically viable manufacturing solution. The process eliminates multiple unit operations required in traditional routes while maintaining consistent product quality across diverse molecular structures, significantly enhancing supply reliability for procurement teams managing complex ingredient portfolios. By utilizing common solvents and commercially available catalysts under moderate conditions, the technology reduces dependency on specialized equipment and hazardous reagents that often create supply bottlenecks in conventional manufacturing.
- Cost Reduction in Manufacturing: The elimination of multiple isolation steps and purification procedures substantially reduces solvent consumption and labor requirements throughout the production cycle. The catalytic nature of the rhodium system minimizes reagent costs compared to stoichiometric approaches, while maintaining high material conversion efficiency maximizes raw material utilization without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The broad substrate compatibility enables consistent production across diverse molecular variants using identical process parameters, reducing qualification timelines for new product variants. The use of readily available starting materials from multiple global suppliers mitigates single-source dependency risks common in specialized chemical manufacturing while enhancing batch-to-batch consistency through simplified process flow.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production has been demonstrated across multiple examples in patent documentation with consistent performance maintenance at larger volumes. The reduced number of processing steps minimizes waste generation compared to conventional multi-step syntheses while facilitating regulatory compliance through elimination of hazardous reagents commonly used in traditional routes.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation potential for agrochemical intermediate manufacturing. These insights have been developed through comprehensive evaluation of the reaction mechanism, scalability data, and comparative process economics presented in patent documentation.
Q: How does this methodology improve upon traditional synthesis routes for furanone isoquinolone compounds?
A: The patented one-pot tandem reaction eliminates multiple isolation steps required in conventional methods while maintaining excellent regioselectivity under milder conditions (80-120°C versus >150°C). This approach significantly reduces production complexity, improves yield consistency across diverse substrates, and minimizes waste generation compared to traditional multi-step syntheses that often require harsh reaction conditions.
Q: What are the key advantages for supply chain reliability when implementing this technology?
A: The broad substrate compatibility allows consistent production across diverse molecular variants using identical process parameters, reducing qualification timelines for new product variants. The use of readily available starting materials from multiple global suppliers mitigates single-source dependency risks, while the simplified process flow with fewer critical control points enhances batch-to-batch consistency.
Q: How does this process support environmental compliance in chemical manufacturing?
A: The reduced number of processing steps minimizes waste generation by eliminating intermediate isolations and purifications required in conventional routes. The catalytic nature of the rhodium system reduces reagent consumption compared to stoichiometric approaches, while operation at moderate temperatures lowers energy requirements and associated carbon footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furanoisoquinolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds for global agrochemical manufacturers. Our state-of-the-art facilities are equipped with rigorous QC labs that ensure stringent purity specifications are consistently met across all production batches, with comprehensive analytical capabilities including advanced chromatography and spectroscopy systems for thorough impurity profiling. With deep expertise in rhodium-catalyzed transformations and heterocyclic chemistry, we provide end-to-end development support from route scouting through commercial manufacturing, ensuring seamless technology transfer and robust process validation for reliable supply chain integration.
For procurement teams seeking to optimize their agrochemical intermediate sourcing strategy, we invite you to request a Customized Cost-Saving Analysis that demonstrates how this patented methodology can enhance your specific supply chain economics. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your production requirements and quality specifications.
