Advanced Two-Step Synthesis of Alpha-Hydroxy Aromatic Ketones for Industrial UV Photoinitiators
The global demand for high-efficiency ultraviolet (UV) photoinitiators has driven significant innovation in the synthesis of alpha-hydroxy aromatic ketones, a critical class of compounds widely used in water-based curing systems. Patent CN102249832A introduces a groundbreaking two-step synthetic methodology that addresses the longstanding inefficiencies of traditional routes. This novel approach combines Friedel-Crafts acylation and hydrohalogenation addition into a single pressurized step, followed by a streamlined alkaline hydrolysis using phase transfer catalysis. By eliminating the need for complex protection-deprotection sequences and cryogenic conditions, this technology offers a robust pathway for the commercial scale-up of complex photoinitiator intermediates. For R&D directors and procurement specialists alike, understanding this shift from multi-step laboratory curiosities to streamlined industrial processes is essential for securing a reliable photoinitiator supplier capable of meeting stringent quality and volume requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-hydroxy ketones like Irgacure 2959 has been plagued by inefficient multi-step sequences that hinder industrial viability. One prominent conventional method involves the reaction of acetone with hydrogen cyanide to form a cyanohydrin, followed by hydroxyl protection using dihydropyran. This protected intermediate must then react with phenylmagnesium bromide before undergoing acidic hydrolysis to remove the protecting group. As illustrated in the reaction scheme below, this pathway is fraught with chemical pitfalls; the acidic reflux conditions required for deprotection often trigger unwanted elimination reactions, drastically reducing the overall yield to approximately 30%. Furthermore, the use of toxic cyanides and the generation of substantial waste streams make this route environmentally unsustainable and economically unviable for large-scale manufacturing.
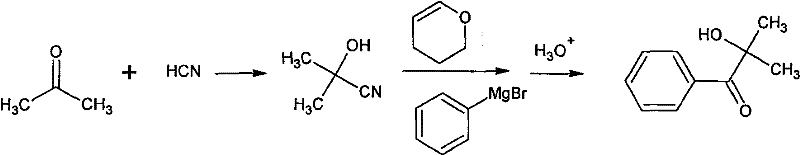
Another traditional approach utilizes organolithium reagents, reacting acetone cyanohydrin trimethylsilane with phenyllithium. While chemically feasible, this method imposes severe operational constraints, requiring reaction temperatures as low as -78°C. Maintaining such cryogenic conditions on a metric-ton scale demands specialized, energy-intensive refrigeration infrastructure that significantly inflates capital and operational expenditures. Additionally, organolithium reagents are notoriously expensive, moisture-sensitive, and hazardous to handle, creating supply chain vulnerabilities. These factors collectively render conventional methods unsuitable for the cost-sensitive and safety-critical environment of modern fine chemical production, necessitating a paradigm shift towards more robust catalytic technologies.
The Novel Approach
The methodology disclosed in CN102249832A represents a decisive break from these legacy constraints by merging acylation and addition reactions into a unified, pressurized step. Instead of building the carbon skeleton through fragile cyanohydrin intermediates, this process directly reacts substituted or unsubstituted aromatic hydrocarbons with alpha-substituted acryloyl halides in the presence of anhydrous hydrogen halides. Conducted in a closed system under moderate pressure (0.01-10 atm) with Lewis acid catalysts, this step efficiently generates the alpha-halo-alpha-substituted aromatic acetone intermediate. This consolidation of reaction steps not only simplifies the workflow but also inherently improves atom economy. The subsequent hydrolysis step utilizes a biphasic organic solvent-water system enhanced by phase transfer catalysts, allowing for mild alkaline conditions that preserve the sensitive alpha-hydroxy ketone structure while ensuring high conversion rates.
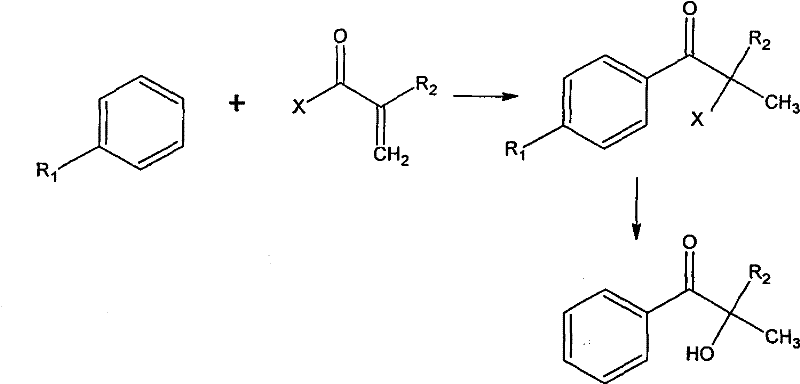
This novel strategy effectively bypasses the thermal instability issues associated with acidic deprotection in older methods. By operating under controlled pressure and utilizing specific metal halide catalysts (such as aluminum, iron, or antimony chlorides), the process achieves a total yield exceeding 60% with product purity reaching 98%. The elimination of cryogenic requirements and the use of readily available bulk chemicals like aromatic hydrocarbons and acryloyl chlorides significantly lower the barrier to entry for production. For supply chain managers, this translates to a more resilient sourcing model where raw material availability is high, and the dependency on exotic, single-source reagents is minimized, ensuring consistent cost reduction in photoinitiator manufacturing.
Mechanistic Insights into Pressurized Friedel-Crafts Acylation and Hydrolysis
The core innovation of this synthesis lies in the mechanistic synergy between electrophilic aromatic substitution and nucleophilic addition under pressurized conditions. In the first step, the Lewis acid catalyst activates the alpha-substituted acryloyl halide, generating a highly reactive acylium ion species. Simultaneously, the presence of anhydrous hydrogen halide facilitates the Markovnikov addition across the double bond of the acryloyl moiety. This dual activation allows the aromatic ring to attack the electrophilic center while the halide ion captures the carbocation intermediate, forming the stable alpha-halo ketone in a single pot. The use of a closed, pressurized system is critical here; it maintains the concentration of gaseous hydrogen halides in the liquid phase, driving the equilibrium forward and preventing the loss of volatile reagents, which is a common failure point in atmospheric reactions.
Impurity control is meticulously managed through the choice of hydrolysis conditions in the second step. Traditional acidic hydrolysis often leads to dehydration of the alpha-hydroxy group, forming alpha,beta-unsaturated ketones as major impurities. In contrast, this patent employs a phase transfer catalyst (such as quaternary ammonium salts or crown ethers) to shuttle hydroxide ions into the organic phase. This creates a localized alkaline environment that promotes nucleophilic substitution of the alpha-halogen without inducing elimination. The result is a clean conversion to the target alpha-hydroxy ketone. This mechanistic precision ensures that the final high-purity photoinitiator meets the rigorous specifications required for UV curing applications, where trace impurities can severely impact color stability and curing speed.
How to Synthesize Alpha-Hydroxy Aromatic Ketones Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the reaction vessel, ensuring it is rated for the specified pressure range and equipped with efficient cooling and heating capabilities. The selection of the catalyst is paramount; while aluminum trichloride is effective, other metal halides like ferric chloride or antimony trichloride offer alternative profiles regarding solubility and workup ease. The hydrolysis step demands careful monitoring of pH and temperature to prevent emulsion formation during the phase transfer process. Detailed standard operating procedures regarding reagent addition rates, pressure maintenance, and quenching protocols are essential for safe scale-up. The following guide outlines the critical operational phases derived from the patent examples.
- React substituted aromatic hydrocarbons with alpha-substituted acryloyl halides and anhydrous hydrogen halides under pressure (0.01-10 atm) with a metal catalyst to form an alpha-halo intermediate.
- Subject the intermediate to hydrolysis in an organic solvent-water system using a phase transfer catalyst and alkaline base.
- Perform extraction and distillation or recrystallization to isolate the final alpha-hydroxy aromatic ketone with purity exceeding 98%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers transformative benefits for procurement strategies and supply chain resilience. The primary driver for cost reduction in fine chemical manufacturing is the simplification of the process flow. By reducing the number of unit operations and eliminating the need for expensive protecting groups and cryogenic infrastructure, the overall production cost per kilogram is significantly lowered. The ability to operate at near-atmospheric or moderate pressures (up to 10 atm) means that standard stainless steel reactors can be utilized, avoiding the need for specialized Hastelloy equipment often required for highly corrosive or extreme condition processes. This compatibility with existing infrastructure accelerates time-to-market and reduces capital expenditure for manufacturers.
- Cost Reduction in Manufacturing: The elimination of organolithium reagents and cryogenic cooling represents a massive saving in raw material and utility costs. Organolithiums are not only expensive but also require specialized handling and storage, adding hidden logistical costs. By replacing these with commodity chemicals like hydrogen chloride and acryloyl chlorides, the variable cost of goods sold (COGS) is drastically reduced. Furthermore, the higher yield (>60% vs. 30% in old methods) means less raw material is wasted per unit of product, directly improving the gross margin profile for the final photoinitiator.
- Enhanced Supply Chain Reliability: The raw materials for this process—benzene derivatives, acryloyl chlorides, and common metal halides—are produced on a massive global scale for various industries. This abundance ensures that supply disruptions are rare compared to niche reagents like phenyllithium or specific silylating agents. For supply chain heads, this diversification of the supplier base mitigates risk. The robustness of the chemistry also means that production campaigns can be run continuously with minimal downtime for cleaning or catalyst regeneration, ensuring a steady flow of inventory to meet just-in-time delivery schedules.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard batch processing techniques familiar to chemical engineers. The use of phase transfer catalysis minimizes the volume of organic solvents required compared to traditional homogeneous reactions, reducing the load on solvent recovery systems. Additionally, the avoidance of cyanide reagents removes a significant regulatory burden and safety hazard from the plant floor. This alignment with green chemistry principles facilitates easier permitting and reduces the long-term liability associated with hazardous waste disposal, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-hydroxy aromatic ketone synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the balance between chemical efficiency and operational practicality that defines this technology.
Q: What are the key advantages of this synthesis over the cyanohydrin route?
A: This novel method eliminates the need for hydroxyl protection and deprotection steps required in the cyanohydrin route, avoiding acidic reflux conditions that cause elimination side reactions and low yields (approx. 30%).
Q: Does this process require cryogenic conditions?
A: No, unlike organolithium-based methods that require -78°C, this process operates at reflux temperatures (e.g., 80°C) under moderate pressure, significantly reducing energy consumption and equipment complexity.
Q: What is the expected purity of the final photoinitiator?
A: The patent data indicates that the content of alpha-hydroxy aromatic ketones can reach 98%, with a total yield exceeding 60%, making it highly suitable for high-performance UV curing applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Aromatic Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this pressurized acylation route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-hydroxy aromatic ketones meets the exacting standards required for high-performance UV photoinitiators. Our commitment to quality ensures that your downstream curing applications perform consistently, free from the variability that plagues lesser suppliers.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. By leveraging our expertise in process intensification and cost-effective manufacturing, we can help you achieve significant efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can become a strategic asset for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
