Advanced Raney Nickel Hydrogenation for High-Trans Fragrance Intermediates: A Commercial Scale-Up Analysis
Advanced Raney Nickel Hydrogenation for High-Trans Fragrance Intermediates: A Commercial Scale-Up Analysis
The global fragrance and flavor industry constantly seeks efficient pathways to produce high-value olfactory compounds with precise stereochemical profiles. A pivotal advancement in this domain is detailed in patent CN1500766A, which discloses a robust method for the preparation of trimethylcyclohexyl-alkan-3-ols containing a high proportion of trans-isomers. These compounds, often associated with trade names like Timberol, are critical ingredients in fine perfumery due to their distinct woody and amber nuances. The technical breakthrough lies in the optimization of catalytic hydrogenation conditions, specifically utilizing Raney Nickel in the absence of copper chromite, to achieve superior trans-selectivity. This innovation addresses long-standing challenges in stereoselective synthesis, offering a viable route for the commercial scale-up of complex fragrance intermediates. By leveraging specific reaction parameters such as elevated temperature, pressure, and the addition of alkali promoters, manufacturers can now access these valuable scent molecules with improved efficiency and reduced environmental burden.
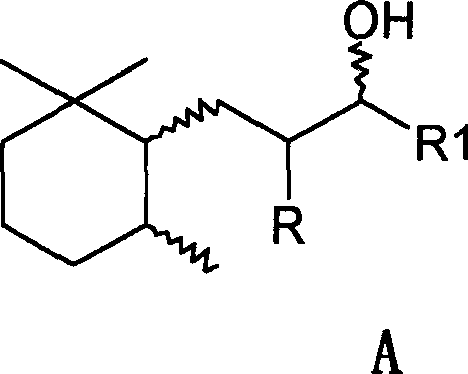
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these saturated alcohols from their unsaturated ketone precursors has been fraught with inefficiencies and selectivity issues. Prior art, such as DE 24 55 761C2, suggested that hydrogenation using Raney Nickel alone was insufficient, claiming that only minimal hydrogen absorption occurred without the addition of copper chromite. This reliance on chromium-based additives introduced significant toxicity concerns and complicated waste stream management, posing a severe liability for modern green chemistry initiatives. Furthermore, alternative approaches utilizing noble metal catalysts like Ruthenium, as described in DE 100 62 771A1, suffered from prohibitively slow reaction kinetics. Reports indicated reaction times extending up to 60 hours to achieve complete conversion, a timeframe that is economically unsustainable for large-scale industrial production. These conventional methods created a bottleneck for reliable fragrance intermediate suppliers, limiting the availability of high-purity trans-isomers and driving up costs due to low throughput and complex purification requirements necessitated by poor selectivity.
The Novel Approach
The methodology outlined in CN1500766A represents a paradigm shift by demonstrating that Raney Nickel, when used as the sole catalyst under optimized conditions, is highly effective. Contrary to previous beliefs, the inventors discovered that complete hydrogenation of the Formula B precursors is achievable without copper chromite. The process involves catalytic hydrogenation in the presence of a nickel catalyst, preferably Raney Nickel, at temperatures ranging from 40°C to 350°C, with a particularly advantageous window between 200°C and 300°C. Crucially, the addition of alkali promoters, such as sodium hydroxide or potassium hydroxide, was found to surprisingly enhance the formation of the desired trans-isomer (Formula D). This approach not only simplifies the catalyst system by removing toxic chromium components but also drastically accelerates the reaction rate, reducing processing time to merely 0.5 to 3 hours. This acceleration is vital for cost reduction in flavor and fragrance manufacturing, as it significantly increases reactor turnover rates and overall plant capacity without requiring additional capital investment in new equipment.
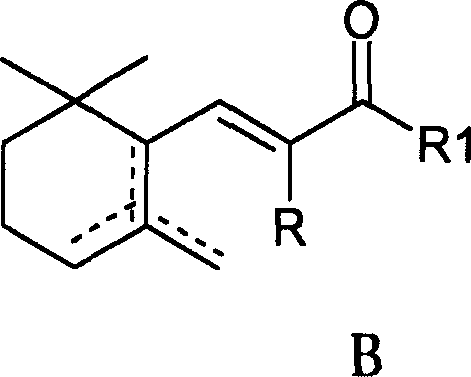
Mechanistic Insights into Raney Nickel-Catalyzed Stereoselective Hydrogenation
The core of this technological advancement lies in the intricate interplay between the heterogeneous catalyst surface, hydrogen pressure, and the thermodynamic equilibrium of the isomers. The hydrogenation of the unsaturated ketone (Formula B) proceeds via the adsorption of the substrate onto the active sites of the Raney Nickel surface. Under standard conditions, the reduction of the double bonds and the carbonyl group can yield a mixture of cis (Formula C) and trans (Formula D) isomers. However, the patent data reveals that the presence of alkali plays a critical mechanistic role in shifting this equilibrium. It is hypothesized that the basic environment facilitates the isomerization of the initially formed cis-alcohol or its intermediate alkoxide species into the thermodynamically more stable trans-configuration. This base-promoted isomerization occurs concurrently with or immediately following the hydrogenation step, effectively driving the product distribution towards the high-value trans-isomer.
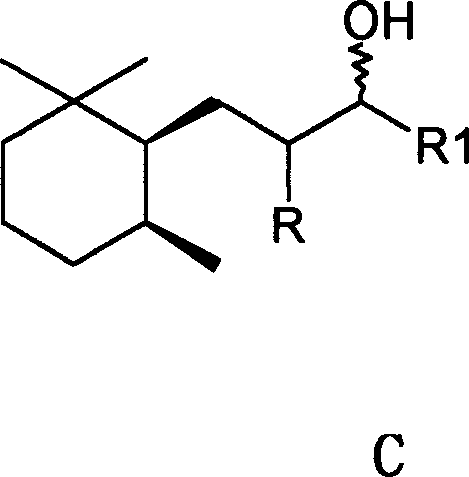
Furthermore, the physical dynamics of the reaction mixture, specifically mass transfer, are identified as key determinants of success. The patent emphasizes the use of high-speed stirring, particularly with gasification agitators, to enhance the solubility of hydrogen in the liquid phase and ensure uniform distribution of the solid catalyst. Experiments demonstrated that increasing stirring velocity from 400 rpm to 1600 rpm could double the amount of trans-isomer formed while simultaneously reducing reaction time from 24 hours to just 1 hour. This suggests that improved mass transfer prevents local depletion of hydrogen, which might otherwise favor kinetic products (cis-isomers) over thermodynamic products (trans-isomers). By rigorously controlling these parameters—temperature, pressure, alkali concentration, and agitation—the process achieves a trans/cis ratio of at least 15:85, and under optimal conditions, ratios as high as 35:65 or better. This level of control is essential for producing high-purity trimethylcyclohexyl-alkan-3-ols that meet the stringent sensory specifications of the luxury fragrance market.
How to Synthesize Trimethylcyclohexyl-Alkan-3-Ols Efficiently
The synthesis protocol derived from this patent offers a straightforward yet highly effective pathway for producing these valuable fragrance alcohols. The process begins with the charging of the unsaturated ketone precursor, such as methylionone or ethylionone derivatives, into a pressure-resistant reactor. Following the addition of the Raney Nickel catalyst and the alkali promoter, the system is pressurized with hydrogen and heated to the target reaction temperature. The reaction progress is monitored to ensure complete consumption of the starting material, typically achieved within a few hours. After the reaction is complete, the catalyst is filtered off, and the crude product is purified via distillation to isolate the final alcohol mixture. For a detailed breakdown of the specific operational parameters and safety considerations, please refer to the standardized guide below.
- Load the unsaturated ketone precursor (Formula B) into a stirred autoclave equipped with a gasification agitator.
- Add Raney Nickel catalyst (0.1-5% m/m) and an alkali promoter such as NaOH or KOH to the reaction mixture.
- Conduct hydrogenation at elevated temperatures (200-300°C) and pressures (10-50 bar) for 0.5 to 3 hours to maximize trans-isomer formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this Raney Nickel-based process offers compelling advantages over legacy technologies. The elimination of expensive noble metals like Ruthenium and toxic additives like copper chromite directly translates to significant raw material cost savings. Moreover, the drastic reduction in reaction time—from days to hours—means that existing manufacturing assets can produce substantially higher volumes of product within the same timeframe. This increase in throughput efficiency effectively lowers the fixed cost per kilogram of the final ingredient, providing a competitive edge in pricing negotiations. For supply chain managers, the robustness of the process ensures greater reliability in meeting delivery schedules, as the risk of batch failures due to slow kinetics or catalyst deactivation is minimized. This reliability is crucial for maintaining the continuity of supply for downstream perfume houses that depend on consistent quality and availability.
- Cost Reduction in Manufacturing: The substitution of costly Ruthenium catalysts with economical Raney Nickel eliminates a major expense driver in the production bill of materials. Additionally, the removal of copper chromite simplifies the downstream purification process, as there is no need for specialized heavy metal scavenging steps or complex waste treatment protocols associated with chromium disposal. The energy efficiency is also improved due to the shorter reaction cycles, leading to lower utility consumption per batch. These cumulative factors result in a leaner, more cost-effective manufacturing operation that can withstand market volatility in raw material pricing.
- Enhanced Supply Chain Reliability: The simplicity of the catalyst system enhances supply chain resilience. Raney Nickel is a widely available commodity chemical, reducing the risk of supply disruptions often associated with specialized noble metal catalysts. The shortened reaction cycle time allows for more flexible production scheduling, enabling manufacturers to respond rapidly to fluctuations in market demand. This agility is a critical asset in the fast-paced fragrance industry, where trends can shift quickly, and the ability to ramp up production of specific scent profiles without long lead times is highly valued by customers seeking a reliable fragrance intermediate supplier.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application in mind. The use of standard hydrogenation equipment and the avoidance of hazardous chromium compounds align perfectly with modern environmental, health, and safety (EHS) regulations. This compliance reduces the regulatory burden and potential liability for manufacturers, facilitating smoother audits and approvals. The ability to run the process in bulk or in solution with common solvents like alcohols or esters further simplifies the scale-up from pilot plant to full commercial production, ensuring that the technology can be deployed globally with minimal friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for R&D teams planning process validation and for procurement officers assessing the long-term viability of the supply source.
Q: Why is the trans-isomer preferred in fragrance applications like Timberol?
A: The trans-isomer (Formula D) possesses superior organoleptic properties, offering more intense woody and amber notes compared to the cis-isomer, which is often less desirable in high-end perfume compositions.
Q: How does this Raney Nickel method improve upon previous Ruthenium-catalyzed processes?
A: Previous Ruthenium-catalyzed methods required excessively long reaction times (up to 60 hours), making them industrially unfeasible. The new Raney Nickel process reduces reaction time to under 3 hours while maintaining high selectivity.
Q: Is copper chromite required for this hydrogenation process?
A: No, the patent explicitly demonstrates that copper chromite is not required. In fact, the process works effectively with Raney Nickel alone, especially when promoted with alkali, eliminating the need for toxic chromium additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethylcyclohexyl-Alkan-3-Ols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in defining the olfactory character of high-end fragrances. Our technical team has extensively analyzed the pathways described in CN1500766A and possesses the expertise to translate this laboratory-scale innovation into robust commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of reaction parameters required for high trans-selectivity is maintained at every batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every shipment meets the exacting standards required by the global flavor and fragrance industry.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. By leveraging our process engineering capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to advanced catalytic technologies can drive value and consistency for your fragrance formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
