Advanced Synthesis of Triazolinthionone Derivatives for Scalable Fungicide Production
Advanced Synthesis of Triazolinthionone Derivatives for Scalable Fungicide Production
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of heterocyclic intermediates, specifically triazoline thione derivatives which serve as critical active compounds in crop protection. Patent CN1515558A introduces a groundbreaking methodology that fundamentally alters the production landscape for these valuable molecules. Unlike traditional routes that rely on harsh sulfurization or complex multi-step hydrolysis, this novel approach utilizes a streamlined condensation of hydrazine derivatives with carbonyl compounds and thiocyanates. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this technology represents a significant leap forward in process efficiency and cost-effectiveness. By leveraging mild reaction conditions and ubiquitous raw materials, the patent outlines a pathway that not only improves yield but also simplifies the supply chain for high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazoline thione derivatives has been plagued by significant technical hurdles that hinder commercial viability. Prior art methods, such as those referenced in WO-A96-16048, often require the use of highly basic conditions followed by complex hydrolysis steps, or direct reaction with elemental sulfur at elevated temperatures. These conventional pathways suffer from inherently low yields and generate substantial waste streams, making them economically unattractive for large-scale cost reduction in agrochemical manufacturing. Furthermore, alternative methods disclosed in literature, such as those involving phenylhydrazine and sodium thiocyanate in the presence of hydrochloric acid, are characterized by excessively long reaction times and an inability to produce unsubstituted derivatives efficiently. The reliance on hazardous reagents like n-Butyl Lithium at cryogenic temperatures (-70°C), as seen in comparison examples, further exacerbates safety risks and operational costs, rendering these older technologies obsolete for modern, sustainable chemical production.
The Novel Approach
The methodology detailed in CN1515558A offers a robust solution by employing a three-component condensation strategy that operates under significantly milder and more controllable conditions. The process begins with the reaction of a 1-chloro-2-hydroxyethane derivative or an oxirane derivative with hydrazine hydrate, followed by condensation with a carbonyl compound and a thiocyanate salt. This sequence allows for the highly selective formation of the triazolidinethione ring, which is subsequently cyclized using formic acid to yield the final triazoline thione structure. As illustrated in the reaction scheme below, this route avoids the need for extreme temperatures or exotic catalysts, facilitating a smoother transition from laboratory bench to pilot plant.
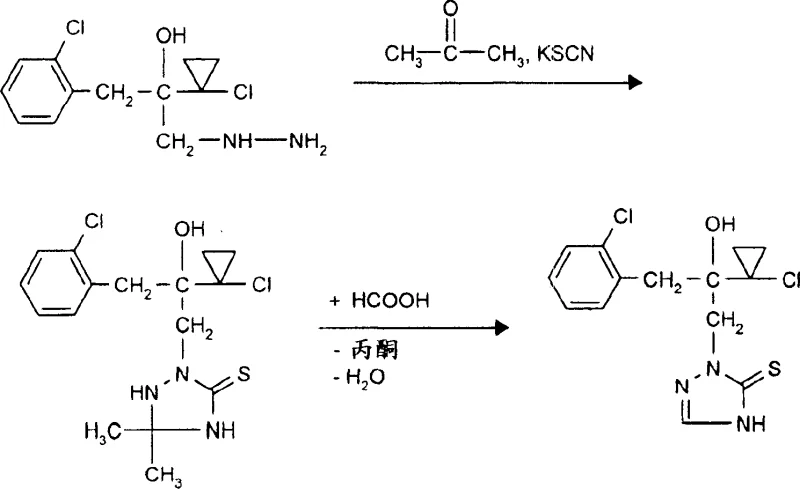
This novel approach not only enhances the commercial scale-up of complex agrochemical intermediates but also ensures a cleaner impurity profile. The use of common solvents like toluene or alcohols, combined with inexpensive reagents such as acetone and potassium thiocyanate, drastically reduces the raw material cost base. Moreover, the ability to telescope certain steps or perform reactions without isolating unstable intermediates provides a distinct advantage in reducing lead time and minimizing handling losses, making it an ideal candidate for companies aiming to optimize their fungicide supply chains.
Mechanistic Insights into Thiocyanate-Mediated Cyclization
The core of this synthetic innovation lies in the efficient construction of the 1,2,4-triazole ring system through a nucleophilic attack mechanism. In the first stage, the hydrazine derivative acts as a bidentate nucleophile, attacking the electrophilic carbon of the carbonyl compound to form an initial hydrazone intermediate. The presence of the thiocyanate ion (SCN-) is crucial, as it facilitates the subsequent cyclization by providing the sulfur and nitrogen atoms necessary to close the five-membered ring. This step is typically catalyzed by mild acids such as hydrochloric acid or acetic acid, which protonate the carbonyl oxygen to enhance electrophilicity without degrading the sensitive hydrazine moiety. The reaction proceeds with high regioselectivity, ensuring that the sulfur atom is incorporated at the correct position to form the thione tautomer, which is the biologically active form for many fungicidal applications.
Following the formation of the triazolidinethione intermediate, the final aromatization step involves treatment with formic acid. This dehydration reaction removes a molecule of water to establish the double bond within the triazoline ring, stabilizing the structure and locking in the desired stereochemistry. The patent data indicates that this step can be performed at temperatures between 80°C and 150°C, which is well within the operating range of standard glass-lined steel reactors. From an impurity control perspective, the mild acidity of formic acid prevents the degradation of the chloro-cyclopropyl and chloro-phenyl substituents, which are prone to hydrolysis under stronger acidic or basic conditions. This preservation of functional groups is critical for maintaining the biological activity of the final API intermediate.
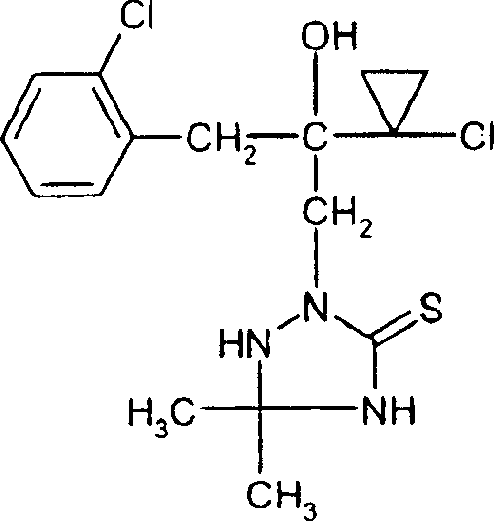
Furthermore, the mechanistic pathway allows for significant flexibility in substrate scope. The R1 and R2 groups on the hydrazine backbone can be varied extensively, including alkyl, cycloalkyl, aryl, and heteroaryl substituents, without compromising the reaction efficiency. This modularity enables the rapid generation of diverse libraries of triazoline thione derivatives for structure-activity relationship (SAR) studies. For process chemists, understanding this mechanism is key to troubleshooting potential side reactions, such as the over-alkylation of the nitrogen atoms or the hydrolysis of the chloro-substituents, ensuring that the final product meets stringent purity specifications required for regulatory approval in the agrochemical sector.
How to Synthesize Triazolinthionone Derivatives Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize byproduct formation. The process generally involves dissolving the hydrazine derivative in a suitable solvent such as toluene or ethanol, followed by the sequential addition of the carbonyl compound and the thiocyanate salt. Acid catalysis is then introduced to drive the cyclization, with the reaction mixture typically stirred at ambient or slightly elevated temperatures for several hours. The resulting triazolidinethione precipitate can be isolated by filtration, washed to remove inorganic salts, and then subjected to the formic acid treatment for final ring closure. Detailed standard operating procedures for each unit operation are essential to ensure reproducibility and safety during scale-up.
- React 1-chloro-2-hydroxyethane or oxirane derivatives with hydrazine hydrate to form the hydrazine intermediate.
- Condense the hydrazine derivative with a carbonyl compound and potassium thiocyanate in the presence of acid to form the triazolidinethione ring.
- Cyclize the triazolidinethione derivative using formic acid, optionally with a catalyst, to yield the final triazoline thione product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and hazardous reagents like n-Butyl Lithium or elemental sulfur with commodity chemicals such as acetone, potassium thiocyanate, and formic acid, manufacturers can achieve substantial cost savings in raw material procurement. These commodities are globally available with stable pricing, reducing the risk of supply disruptions that often plague specialty chemical markets. Additionally, the elimination of cryogenic cooling requirements (down to -70°C in older methods) significantly lowers energy consumption and capital expenditure on specialized refrigeration equipment, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The new process eliminates the need for transition metal catalysts and expensive organolithium reagents, which are not only costly but also require rigorous removal steps to meet heavy metal specifications in the final product. By utilizing a metal-free organic synthesis pathway, the downstream purification process is streamlined, reducing solvent usage and waste disposal costs. The high yields reported in the patent examples, often exceeding 70-80%, mean that less raw material is wasted per kilogram of product, directly improving the gross margin for fungicide manufacturing. This efficiency gain allows producers to offer more competitive pricing in the global market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures a resilient supply chain that is less susceptible to geopolitical fluctuations or single-source bottlenecks. Unlike specialized catalysts that may have long lead times, reagents like hydrazine hydrate and thiocyanates are produced by multiple vendors worldwide, providing procurement teams with greater negotiating power and flexibility. Furthermore, the robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-validation, ensuring supply continuity even in the face of unexpected production interruptions at a single facility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to traditional sulfurization techniques. The avoidance of elemental sulfur dust and hydrogen sulfide gas generation reduces the burden on scrubber systems and minimizes the risk of workplace exposure incidents. The aqueous workup steps generate waste streams that are easier to treat compared to the complex organic residues from organometallic reactions. This alignment with green chemistry principles facilitates faster regulatory approvals and supports corporate sustainability goals, making the technology attractive for companies aiming to reduce their environmental footprint while scaling up production of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazoline thione synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear picture of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the key advantages of this new synthesis method over conventional routes?
A: The novel method described in CN1515558A avoids the harsh conditions of traditional sulfurization or highly basic hydrolysis. It operates under milder temperatures (0°C-120°C) and utilizes readily available reagents like acetone and potassium thiocyanate, resulting in significantly higher yields (up to 82% in examples) compared to the low yields and difficult scale-up of prior art methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It eliminates the need for expensive transition metal catalysts or hazardous reagents like n-Butyl Lithium found in comparison examples. The workup involves standard washing and crystallization steps, making it highly compatible with existing multipurpose reactor infrastructure for agrochemical intermediate production.
Q: What is the purity profile of the resulting triazoline thione derivatives?
A: The method demonstrates high selectivity, particularly in the displacement of the methylene group during the second step. Experimental data shows the formation of colorless crystalline solids with sharp melting points (e.g., 138-139°C), indicating high chemical purity and minimal byproduct formation, which is critical for downstream fungicide efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinthionone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the competitiveness of the agrochemical industry. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1515558A are fully realized in practical manufacturing settings. We possess the infrastructure and technical expertise to handle the specific challenges of heterocyclic chemistry, including the management of exothermic condensation reactions and the precise control of crystallization parameters to meet stringent purity specifications. Our rigorous QC labs are equipped to analyze complex impurity profiles, guaranteeing that every batch of triazolinthionone derivative meets the highest standards of quality required for downstream formulation.
We invite you to collaborate with us to optimize your supply chain for fungicide intermediates. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us help you transform innovative patent chemistry into a reliable, cost-effective reality for your business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
