Advanced Copper-Catalyzed Synthesis of Benzothiazole Esters for Industrial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective synthetic routes for high-value intermediates, particularly those containing privileged scaffolds like benzothiazole. Patent CN110156716B, published in August 2021, introduces a groundbreaking methodology for the synthesis of benzothiazole ester derivatives that addresses critical pain points in modern process chemistry. This innovation leverages a synergistic copper and silver catalytic system to achieve direct C-H acyloxylation, bypassing the need for pre-functionalized substrates. For R&D directors and process chemists, this represents a paradigm shift from precious metal dependency to abundant base metal catalysis. The technology enables the efficient construction of complex ester linkages on the benzothiazole core, a structural motif ubiquitous in bioactive molecules ranging from anticancer agents to fluorescent probes. By operating under ambient air conditions and utilizing inexpensive reagents, this patent lays the foundation for a more sustainable and economically viable supply chain for these essential chemical building blocks.
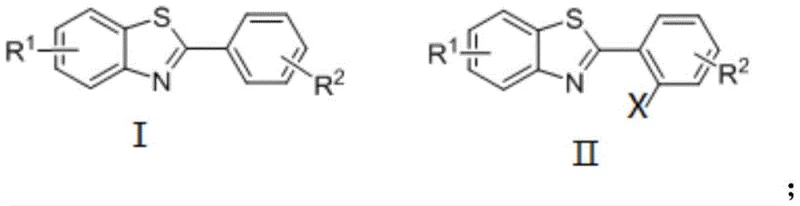
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of benzothiazole rings to introduce ester groups has relied heavily on transition metal catalysis, predominantly featuring palladium complexes. Literature precedents, such as those reported by the Wu and Ding groups, often utilize palladium acetate (Pd(OAc)2) in conjunction with strong oxidants like ammonium ceric nitrate (CAN). While effective on a laboratory scale, these conventional methods suffer from severe drawbacks when translated to industrial manufacturing. The primary limitation is the exorbitant cost associated with palladium and ruthenium catalysts, which directly inflates the Cost of Goods Sold (COGS) for the final active pharmaceutical ingredient (API). Furthermore, these reactions frequently require stringent inert atmosphere conditions, necessitating specialized equipment and increasing operational risks. The use of toxic oxidants and the generation of heavy metal waste streams also pose significant environmental compliance challenges, complicating waste treatment protocols and increasing the overall ecological footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in CN110156716B utilizes a cost-effective copper catalyst system, specifically employing species such as cuprous oxide (Cu2O), copper acetate, or cuprous iodide. This substitution of noble metals with base metals is a strategic masterstroke for cost reduction in fine chemical manufacturing. The reaction proceeds smoothly in an air atmosphere, eliminating the need for nitrogen or argon purging, which drastically simplifies the reactor setup and operational procedures. The method employs silver salts, such as silver carbonate, as oxidants, which are more manageable and less hazardous than traditional strong oxidants. This new pathway not only reduces the financial burden of catalyst procurement but also streamlines the downstream purification process by minimizing heavy metal contamination. The result is a cleaner reaction profile with fewer side products, higher target yields, and a significantly reduced environmental impact, making it an ideal candidate for green chemistry initiatives in large-scale production facilities.
Mechanistic Insights into Copper-Catalyzed C-H Acyloxylation
The core of this technological advancement lies in the mechanistic efficiency of the copper-silver dual catalytic cycle. The reaction initiates with the activation of the C(sp2)-H bond at the ortho-position of the benzothiazole ring by the copper catalyst. Unlike palladium mechanisms that often require directing groups or harsh conditions, the copper species here facilitates a coordinated insertion that is highly selective for the desired position. The presence of the silver salt serves a dual purpose: it acts as an oxidant to regenerate the active copper species and potentially assists in the activation of the carboxylic acid substrate. This synergistic interaction ensures that the catalytic turnover number remains high throughout the reaction duration of 18 to 24 hours. The mechanism avoids the formation of unstable intermediates that typically lead to polymerization or decomposition, thereby ensuring a high degree of chemoselectivity even in the presence of sensitive functional groups on the aromatic ring.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical quality assurance. The avoidance of palladium eliminates the risk of residual Pd contamination, a critical specification limit in API manufacturing that often requires expensive scavenging resins to meet regulatory standards (e.g., ICH Q3D guidelines). The reaction conditions, specifically the temperature range of 130°C to 140°C in solvents like chlorobenzene or p-xylene, are optimized to maximize kinetic energy for bond formation while minimizing thermal degradation of the substrate. The use of air as the terminal oxidant source further simplifies the redox balance, preventing the accumulation of reduced metal species that could catalyze unwanted side reactions. Consequently, the crude reaction mixture exhibits a cleaner impurity profile, facilitating easier isolation of the high-purity benzothiazole ester derivatives through standard extraction and chromatography techniques.
How to Synthesize Benzothiazole Ester Derivatives Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility, key factors for any reliable pharmaceutical intermediate supplier. The process begins with the precise weighing of the benzothiazole derivative and the chosen organic carboxylic acid, maintaining a molar ratio that favors the acid slightly (1:1.5 to 1:2.2) to drive the equilibrium towards product formation. The catalyst loading is remarkably low, typically ranging from 0.05 to 0.2 equivalents relative to the substrate, demonstrating the high efficiency of the copper system. The reaction is conducted in a standard heated vessel without the need for pressure reactors or glove boxes. Following the heating period, the workup involves a straightforward liquid-liquid extraction using ethyl acetate, followed by drying and concentration. The final purification is achieved via silica gel column chromatography using a petroleum ether and ethyl acetate gradient, yielding the target ester in high purity.
- Mix benzothiazole derivatives, organic carboxylic acid, copper catalyst (e.g., Cu2O), and silver salt (e.g., Ag2CO3) in an organic solvent like chlorobenzene.
- Heat the reaction mixture to 130-140°C under air atmosphere for 18-24 hours to facilitate the acyloxylation reaction.
- Extract the crude product with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits beyond mere technical feasibility. The shift from precious metals to base metals fundamentally alters the cost structure of the supply chain, mitigating the volatility associated with the global market prices of palladium and rhodium. This stability allows for more accurate long-term budgeting and pricing contracts with downstream pharmaceutical clients. Additionally, the simplified operational requirements—specifically the ability to run reactions under air—reduce the capital expenditure needed for specialized inert atmosphere reactors, allowing existing infrastructure to be utilized more effectively. This flexibility enhances supply chain resilience, as production can be ramped up quickly without waiting for complex equipment installation or certification.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts results in substantial cost savings per kilogram of produced intermediate. Since copper salts are orders of magnitude cheaper than their noble metal counterparts, the raw material cost contribution of the catalyst becomes negligible. Furthermore, the removal of heavy metal scavenging steps from the downstream processing reduces the consumption of auxiliary materials and lowers waste disposal costs. This comprehensive reduction in both direct material costs and processing overheads significantly improves the gross margin potential for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade chemicals such as cuprous oxide and silver carbonate ensures a stable supply of reagents. Unlike specialized ligands or organometallic complexes that may have single-source suppliers and long lead times, these copper and silver salts are produced globally in massive quantities. This abundance minimizes the risk of supply disruptions due to geopolitical issues or manufacturer outages. Consequently, production schedules become more predictable, reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its robust nature and tolerance to air. Scaling from gram to ton quantities does not require fundamental changes to the reaction engineering, as the heat transfer and mixing requirements are standard for batch reactors. Moreover, the greener profile of the reaction, characterized by the absence of toxic oxidants and reduced heavy metal waste, aligns perfectly with increasingly stringent environmental regulations. This compliance reduces the administrative burden of environmental reporting and permits, facilitating smoother operations in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and reliability for decision-makers. Understanding these nuances is crucial for evaluating the fit of this technology within your specific production portfolio.
Q: Why is the copper-catalyzed method superior to traditional palladium catalysis for benzothiazole synthesis?
A: The copper-catalyzed method eliminates the need for expensive noble metals like palladium or ruthenium, significantly reducing raw material costs. Furthermore, it operates effectively under air atmosphere without requiring inert gas protection, simplifying operational complexity and equipment requirements for large-scale production.
Q: What represents the optimal reaction conditions for maximizing yield in this synthesis?
A: According to the patent data, optimal yields are achieved by maintaining a reaction temperature between 130°C and 140°C for a duration of 18 to 24 hours. The molar ratio of benzothiazole derivative to organic carboxylic acid should be maintained around 1:1.5 to 1:2.2, utilizing copper(I) oxide and silver carbonate as the preferred catalyst and oxidant system.
Q: Is this synthetic route suitable for diverse substrate scopes in industrial applications?
A: Yes, the method demonstrates high efficiency and a wide application range. It is compatible with various substituted benzothiazoles (including 6-methoxy variants) and a broad spectrum of organic carboxylic acids, including aromatic acids, cinnamic acids, and heterocyclic acids like thiophene-2-carboxylic acid, ensuring versatility for different pharmaceutical intermediate needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole Ester Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis described in CN110156716B for the production of high-value benzothiazole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this process, while our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a secure and optimized supply solution.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthetic route for their next-generation drug candidates. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in benzothiazole chemistry can accelerate your project timelines and enhance your competitive advantage in the marketplace.
