Scalable Manufacturing of Oxazolylethanol Derivatives via Safe Aqueous Acylation and Reduction
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, particularly those serving as scaffolds for metabolic disease treatments. Patent CN1781915A discloses a groundbreaking methodology for the preparation of oxazolylethanol derivatives, specifically targeting the synthesis of compound (11), a potent intermediate for diabetes remedies. This invention addresses the severe limitations of prior art methods, such as those described in WO 95/18125 and the Journal of Medicinal Chemistry (1992), which relied on hazardous reagents and suffered from poor scalability. The core innovation lies in the strategic replacement of toxic solvents like methylene chloride with aqueous systems and the substitution of dangerous reducing agents with safer alternatives, resulting in a process that is not only environmentally superior but also economically viable for large-scale operations.
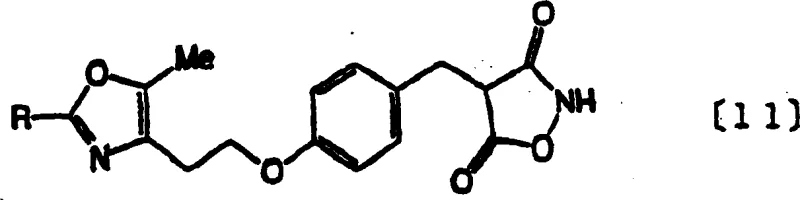
For procurement managers and supply chain directors, the implications of this technology are profound. By shifting the synthesis paradigm from laboratory-centric protocols to industrial-grade chemistry, the patent enables a reliable pharmaceutical intermediates supplier to offer high-purity compounds with drastically simplified purification workflows. The elimination of complex post-processing steps and the use of readily available mineral alkalis translate directly into cost reduction in API manufacturing, ensuring a stable supply chain for downstream drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these critical intermediates relied on 'Method A' and 'Method B', both of which presented significant bottlenecks for commercial production. As illustrated in the prior art pathways, the initial acylation step typically utilized methylene chloride as a solvent and triethylamine as a base. While effective on a gram scale, the industrial use of methylene chloride is heavily restricted due to environmental regulations regarding discharge, making it impractical for multi-ton production. Furthermore, the reduction step in conventional methods employed lithium aluminum hydride (LiAlH4) in diethyl ether. Both reagents are highly hazardous; LiAlH4 is pyrophoric and requires stringent safety protocols, while diethyl ether poses extreme flammability risks, creating substantial liability and operational costs for manufacturing facilities.
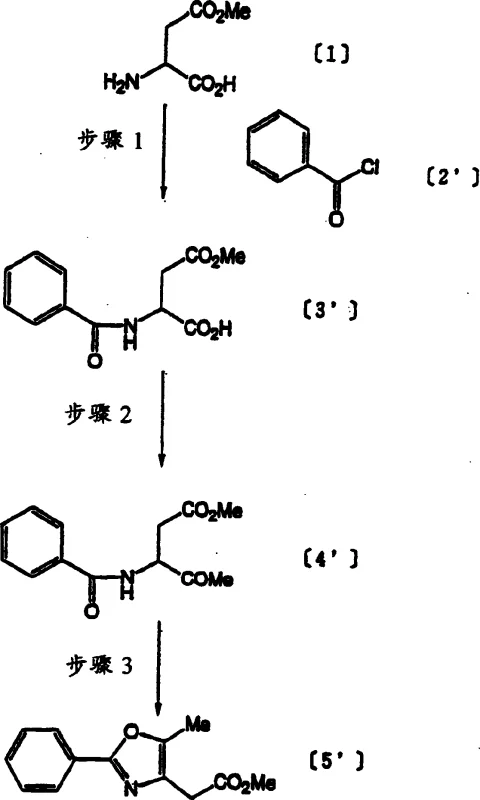
Beyond safety concerns, the chemical efficiency of traditional routes was suboptimal. The cyclization step often utilized phosphorus oxychloride (POCl3), a corrosive and toxic substance that complicates waste treatment and equipment maintenance. Additionally, the mesylation step using tosyl chloride (TsCl) frequently generated unwanted byproduct (15), significantly lowering the yield of the desired intermediate (7'). These cumulative inefficiencies resulted in an overall process that was costly, dangerous, and difficult to scale, failing to meet the rigorous demands of modern good manufacturing practice (GMP) standards.
The Novel Approach
The present invention introduces a transformative 'Novel Approach' that systematically dismantles these barriers through intelligent reagent selection and process optimization. Instead of organic solvents, the initial acylation is conducted in a water-based solvent system using inexpensive mineral alkalis like potassium carbonate or sodium carbonate. This simple switch not only resolves environmental compliance issues but surprisingly boosts the yield to an impressive 92-97%, a marked improvement over the prior art. The reduction step is revolutionized by using sodium borohydride (NaBH4) in tetrahydrofuran (THF) with methanol as an activator. This combination eliminates the fire hazards associated with LiAlH4 while achieving yields of 85-95%, ensuring a safer and more efficient transformation.
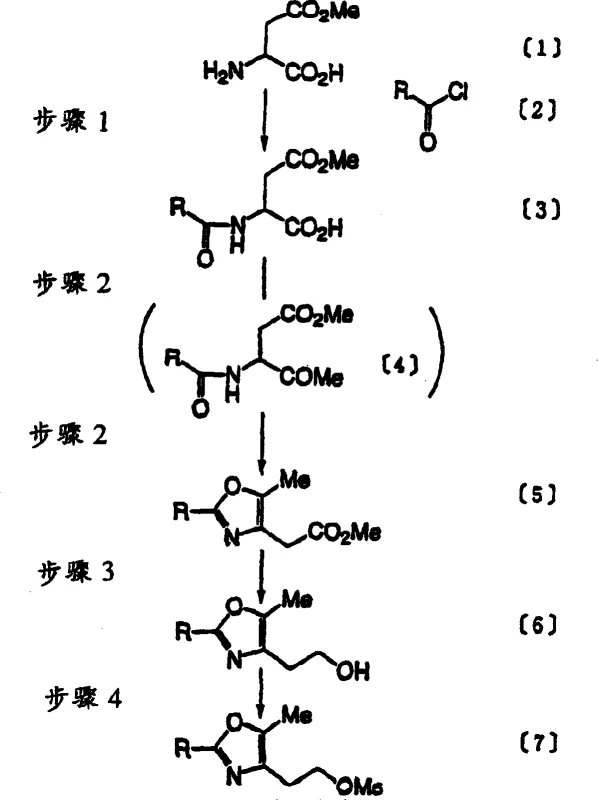
Furthermore, the novel process optimizes the mesylation step by replacing tosyl chloride with methanesulfonyl chloride (MsCl). This critical modification prevents the formation of byproduct (15), allowing for a near-quantitative yield of 99-100% for compound (7). The subsequent condensation and cyclization steps are also refined, utilizing pre-formed intermediates and optimized solvent systems to push the final yield of compound (11) to approximately 80%, compared to the unsatisfactory 40-65% yields of previous methods. This holistic redesign ensures that the commercial scale-up of complex pharmaceutical intermediates is both technically feasible and economically attractive.
Mechanistic Insights into Aqueous Acylation and Safe Reduction
The mechanistic success of this process hinges on the unique behavior of reactants in aqueous media and the specific activation of reducing agents. In the first step, the reaction of L-aspartic acid beta-methyl ester with an acyl chloride in the presence of mineral alkali proceeds efficiently despite the presence of water. The mineral alkali acts as a proton scavenger, neutralizing the hydrochloric acid byproduct and driving the equilibrium towards the formation of the amide bond in compound (3). The use of water as a solvent enhances the solubility of the inorganic salts and facilitates heat dissipation, which is crucial for controlling exothermic reactions during large-scale addition of acyl chlorides. This phase-transfer-like behavior allows for high conversion rates without the need for expensive phase-transfer catalysts in the initial stage.
Impurity control is another cornerstone of this mechanistic design, particularly evident in the mesylation and condensation stages. In conventional tosylation, the steric bulk and reactivity of TsCl can lead to side reactions forming compound (15), which is difficult to separate and drags down overall purity. By switching to MsCl, the reaction becomes cleaner and faster, effectively shutting down this impurity pathway. Similarly, in the condensation step, the instability of the aldehyde intermediate (13) in the old method led to decomposition and low yields. The new method circumvents this by pre-synthesizing the stable benzylidene malonate derivative (8) and reacting it directly with the mesylate (7). This 'telescoped' approach minimizes the exposure of unstable intermediates to reaction conditions, thereby preserving material integrity and maximizing the yield of the benzylidene derivative (9) to 80-85%.
How to Synthesize Oxazolylethanol Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to replicate the high yields reported in the patent. The process begins with the careful preparation of the aqueous reaction mixture, ensuring the mineral alkali is fully dissolved before the addition of the acyl chloride to prevent local overheating. Following the acylation, the subsequent cyclization and reduction steps must be monitored closely for temperature, as the decarboxylation and reduction reactions are sensitive to thermal variations. For detailed operational parameters, including exact stoichiometry, temperature ramps, and workup procedures, operators should refer to the standardized protocols derived from the patent examples.
- Perform acylation of L-aspartic acid beta-methyl ester in water using mineral alkali to obtain compound (3) with 92-97% yield.
- Cyclize compound (3) using acetic anhydride and tosic acid monohydrate in toluene to form oxazolyl acetate (5) without isolation of intermediate (4).
- Reduce compound (5) safely using sodium borohydride in THF/methanol to yield oxazolylethanol (6), avoiding hazardous lithium aluminum hydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain leaders, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple yield improvements. The shift away from regulated solvents like methylene chloride and hazardous reagents like lithium aluminum hydride fundamentally alters the cost structure of production. By eliminating the need for specialized containment systems, explosion-proof infrastructure, and complex waste treatment for halogenated solvents, the manufacturing overhead is significantly reduced. This allows for a more competitive pricing model for the final intermediate, providing downstream partners with a cost-effective source of high-quality material without compromising on safety or regulatory compliance.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents with commodity chemicals drives substantial cost savings. For instance, substituting lithium aluminum hydride with sodium borohydride reduces raw material costs drastically, as NaBH4 is cheaper, safer to handle, and generates less hazardous waste. Furthermore, the ability to perform the initial acylation in water removes the cost burden associated with purchasing, recovering, and disposing of large volumes of organic solvents. The high yields achieved at every step (92-97% for acylation, 99-100% for mesylation) mean that less raw material is wasted, directly improving the material throughput and reducing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The reliance on readily available, non-proprietary reagents ensures a robust and resilient supply chain. Mineral alkalis, sodium borohydride, and methanesulfonyl chloride are bulk commodities with stable global supply networks, unlike specialized catalysts or restricted solvents that may face supply disruptions. This stability guarantees consistent production schedules and reliable delivery times for clients. Additionally, the simplified purification processes reduce the lead time for batch release, enabling faster response to market demands and preventing stockouts of critical diabetes remedy intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale, addressing the 'poor practicability' noted in prior art. The use of toluene and water as primary solvents aligns with green chemistry principles, minimizing the environmental footprint of the manufacturing site. This compliance reduces the risk of regulatory fines and shutdowns, ensuring long-term operational continuity. The high thermal stability and safety profile of the reagents allow for scaling from pilot plant quantities (100 kgs) to full commercial production (100 MT) with minimal engineering changes, facilitating rapid capacity expansion to meet growing global demand for antidiabetic medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this novel method over traditional approaches.
Q: How does the new process improve safety compared to conventional methods?
A: The novel process replaces highly flammable diethyl ether and pyrophoric lithium aluminum hydride with tetrahydrofuran and sodium borohydride. Additionally, it eliminates corrosive phosphorus oxychloride (POCl3) in favor of tosic acid monohydrate for cyclization.
Q: What is the yield improvement for the mesylation step?
A: By switching from tosyl chloride (TsCl) to methanesulfonyl chloride (MsCl), the process avoids the formation of byproduct (15), achieving a near-quantitative yield of 99-100% for compound (7).
Q: Can this process be scaled for industrial production?
A: Yes, the substitution of methylene chloride with water and toluene, along with the use of stable mineral alkalis, significantly reduces environmental discharge restrictions and facilitates large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolylethanol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving diabetes therapies. Our technical team has extensively analyzed the methodologies described in CN1781915A and possesses the expertise to implement these advanced synthetic routes effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of oxazolylethanol derivatives meets the highest international standards for pharmaceutical applications.
We invite pharmaceutical partners to collaborate with us to leverage these process innovations for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to safety and efficiency can drive value for your organization.
