Revolutionizing S-Adenosylmethionine Production with Advanced Recombinant Enzyme Engineering
Revolutionizing S-Adenosylmethionine Production with Advanced Recombinant Enzyme Engineering
The pharmaceutical and nutraceutical industries continuously demand more efficient, cost-effective, and high-purity production methods for critical bioactive molecules. A significant technological breakthrough in this domain is detailed in Chinese patent CN111235084A, which discloses a novel recombinant Escherichia coli engineering bacterium designed for the preparation of S-adenosylmethionine (SAM). This innovation addresses long-standing bottlenecks in SAM manufacturing by integrating microbial fermentation advantages with precise enzymatic catalysis. By leveraging a dual-expression system that co-produces S-adenosylmethionine synthase and ATP synthase, this technology offers a transformative pathway for producing high-purity pharmaceutical intermediates. The implications for global supply chains are profound, promising a shift away from costly, low-yield traditional methods toward a streamlined, sustainable biocatalytic process that enhances both economic viability and product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of S-adenosylmethionine has been plagued by significant technical and economic hurdles across three primary methodologies. Chemical synthesis routes often suffer from complex product configurations and low yields, generating substantial impurity profiles that require rigorous and expensive purification steps to meet pharmaceutical standards. Traditional microbial fermentation, while widely used, typically relies on yeast cells where SAM accumulates intracellularly. This necessitates complex cell wall disruption procedures to release the product, leading to increased processing time, higher energy consumption, and potential product degradation during extraction. Furthermore, conventional enzymatic catalysis, although specific, has been limited by the prohibitive cost of adenosine triphosphate (ATP), a key substrate that can cost between 600 to 700 yuan per kilogram, rendering the process economically unfeasible for large-scale applications without an efficient regeneration system.
The Novel Approach
The methodology outlined in patent CN111235084A introduces a sophisticated genetic engineering solution that effectively bypasses these traditional constraints. By constructing a recombinant E. coli strain capable of co-expressing S-adenosylmethionine synthase alongside a suite of ATP regeneration enzymes—including adenosine kinase (AK), adenylate kinase (ADK), and polyphosphate kinase (PPK)—the process creates a self-sustaining catalytic cycle. This ingenious design allows for the in situ regeneration of ATP from cheaper precursors like adenosine and polyphosphate, drastically reducing raw material costs. The engineered strain facilitates the secretion of SAM into the extracellular medium, eliminating the need for harsh cell disruption and simplifying downstream purification. This holistic approach combines the robustness of fermentation with the precision of enzymatic synthesis, establishing a new benchmark for efficiency in fine chemical manufacturing.
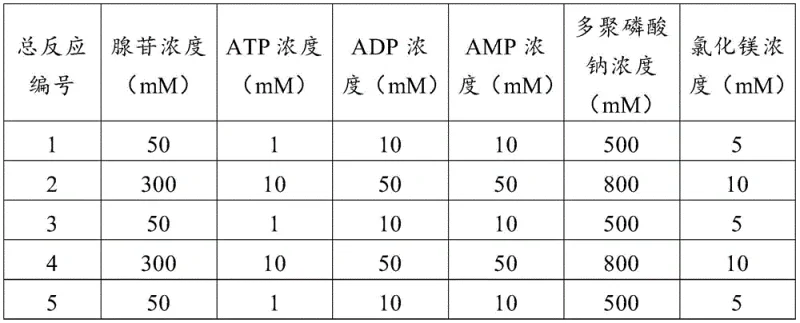
Mechanistic Insights into Dual-Enzyme Co-Expression Catalysis
At the heart of this technological advancement lies a meticulously engineered metabolic pathway that optimizes the thermodynamic and kinetic parameters of SAM synthesis. The core mechanism involves the synergistic action of two distinct enzymatic systems within a single host organism. The S-adenosylmethionine synthase, specifically mutated at key amino acid sites such as C9R and K224R to enhance stability and activity, catalyzes the transfer of the adenosyl group from ATP to L-methionine. Simultaneously, the co-expressed ATP synthase system drives a regeneration loop where adenosine is phosphorylated back to ATP using polyphosphate as an energy donor. This coupling ensures that the concentration of ATP remains sufficient to drive the SAM synthesis reaction forward without the need for continuous external supplementation, effectively solving the cost barrier associated with stoichiometric ATP usage in vitro.
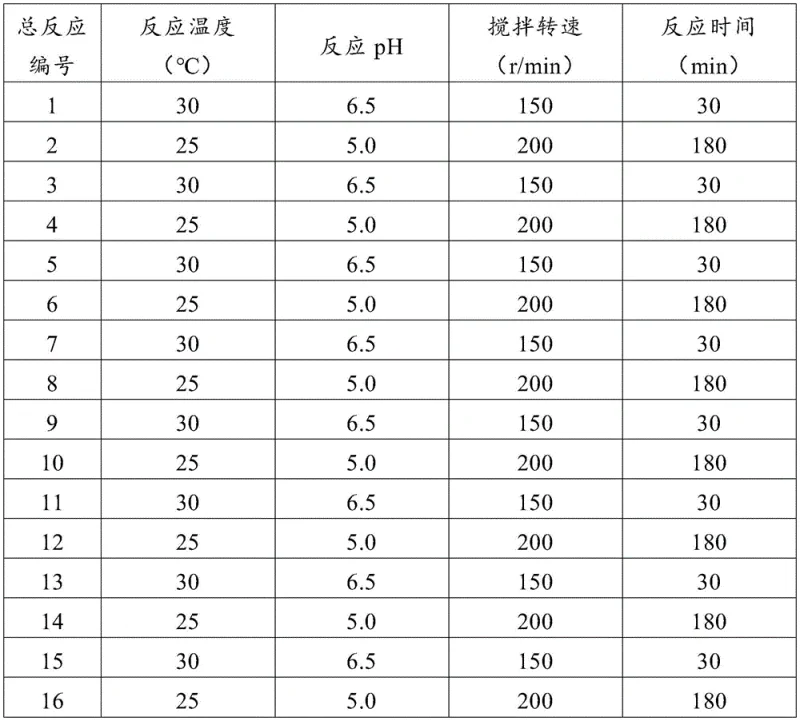
From a quality control perspective, this recombinant system offers superior impurity management capabilities crucial for regulatory compliance. The use of specific protein mutants ensures high stereoselectivity, favoring the production of the bioactive (S,S)-SAM isomer while minimizing the formation of inactive or potentially harmful stereoisomers. Furthermore, because the reaction occurs in a controlled whole-cell catalytic environment, side reactions common in chemical synthesis are largely suppressed. The extracellular secretion of the product means that the final broth contains significantly fewer intracellular contaminants such as nucleic acids and complex proteins, which typically complicate purification in traditional fermentation. This results in a cleaner crude product profile, reducing the burden on chromatographic separation steps and ensuring consistent batch-to-batch quality essential for pharmaceutical grade intermediates.
How to Synthesize S-Adenosylmethionine Efficiently
The implementation of this recombinant technology follows a logical, scalable workflow designed for industrial adoption. The process begins with the fermentation of the engineered E. coli strain in a defined medium, followed by a mild permeabilization step to allow substrate entry and product exit without destroying cell integrity. The subsequent biocatalytic reaction is divided into two optimized stages: an initial ATP regeneration phase followed by the SAM synthesis phase, each conducted under specific pH and temperature conditions to maximize enzyme turnover. This structured approach ensures that the expensive cofactors are utilized with maximum efficiency. For detailed operational parameters and standardized protocols regarding media composition and reaction kinetics, please refer to the technical guide below.
- Ferment the recombinant E. coli strain co-expressing SAM synthase and ATP synthase genes in TB medium with appropriate antibiotics.
- Harvest cells by centrifugation and perform permeabilization treatment using Triton X-100 and EDTA in phosphate buffer.
- Conduct a two-stage enzymatic reaction: first regenerate ATP using adenosine and polyphosphate, then synthesize SAM by adding L-methionine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recombinant E. coli technology translates directly into tangible strategic benefits that enhance overall business resilience. The most immediate impact is observed in the structure of the bill of materials, where the elimination of stoichiometric ATP addition removes a major cost driver from the production equation. By regenerating ATP internally using low-cost polyphosphates, the process decouples production economics from the volatile pricing of high-purity nucleotides. This structural cost advantage allows for more competitive pricing models and improved margin stability, even in fluctuating raw material markets. Additionally, the simplified downstream processing resulting from extracellular product secretion reduces the consumption of solvents and resins, further contributing to substantial cost savings in manufacturing operations.
- Cost Reduction in Manufacturing: The integration of an ATP regeneration cycle fundamentally alters the cost structure of SAM production by removing the dependency on expensive exogenous ATP. This biochemical efficiency means that the process consumes significantly less high-value raw material per unit of output, leading to a drastic reduction in variable production costs. Furthermore, the avoidance of complex cell disruption equipment and the reduction in purification steps lower both capital expenditure and operating expenses. These cumulative efficiencies create a leaner manufacturing model that is inherently more profitable and less sensitive to supply chain disruptions affecting specialty chemical inputs.
- Enhanced Supply Chain Reliability: Utilizing a robust E. coli expression system offers superior scalability and reliability compared to yeast-based fermentation or chemical synthesis. E. coli strains are well-characterized, grow rapidly, and are less susceptible to the phage infections or metabolic instabilities that can plague other microbial hosts. The short fermentation cycle, reported to be under 24 hours, enables faster turnaround times and more responsive production scheduling. This agility allows suppliers to react quickly to changes in market demand, ensuring consistent availability of high-purity intermediates for downstream drug manufacturers and reducing the risk of stockouts.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by reducing waste generation and energy consumption. The absence of harsh cell breaking steps and the reduced need for organic solvents in purification align with increasingly stringent environmental regulations globally. This eco-friendly profile not only minimizes waste disposal costs but also enhances the sustainability credentials of the supply chain, a factor of growing importance to multinational pharmaceutical clients. The simplicity of the two-stage reaction setup facilitates easy scale-up from pilot to commercial volumes without the need for complex reactor modifications, ensuring a smooth transition to mass production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for industry stakeholders. Understanding these details is critical for evaluating the technology's fit within existing manufacturing frameworks and supply chain strategies.
Q: How does this recombinant method reduce production costs compared to traditional fermentation?
A: The method eliminates the need for expensive exogenous ATP addition by co-expressing ATP regeneration enzymes (AK, ADK, PPK), significantly lowering raw material costs.
Q: What are the purity advantages of this enzymatic approach?
A: The use of specific SAM synthase mutants ensures high stereoselectivity for the bioactive (S,S)-SAM isomer, while extracellular secretion simplifies purification and reduces impurity profiles.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes robust E. coli hosts with short fermentation cycles (<24h) and avoids complex cell disruption steps, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Adenosylmethionine Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a technically proficient manufacturer is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced biocatalytic technologies like the one described in patent CN111235084A to deliver superior products. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of S-adenosylmethionine meets the highest international standards for safety and efficacy.
We invite you to explore how our innovative production capabilities can optimize your supply chain and reduce your overall procurement costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced recombinant methods can serve as a reliable foundation for your long-term production goals.
