Revolutionizing Lusutrombopag Intermediate Production via High-Efficiency Carbonyl Reductase Catalysis
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing complex chiral intermediates, particularly for high-value therapeutic agents like Lusutrombopag. A significant breakthrough in this domain is documented in Chinese patent CN112852769A, which discloses a novel method for preparing (S)-1-(2-methoxy-3-bromophenyl) ethanol, a critical chiral building block for this antiplatelet medication. This patent introduces a highly stereoselective carbonyl reductase capable of transforming the corresponding ketone precursor with exceptional efficiency, achieving yields not lower than 95% and enantiomeric excess values exceeding 99%. Unlike traditional chemical methods that rely on harsh reagents, this biocatalytic approach operates under mild physiological conditions, utilizing recombinant cells as whole-cell biocatalysts to drive the reaction. For global procurement leaders and R&D directors, this technology represents a paradigm shift towards greener, safer, and potentially more cost-effective manufacturing strategies for thrombocytopenia treatments, addressing both supply chain sustainability and regulatory compliance regarding solvent residues and heavy metal contamination.
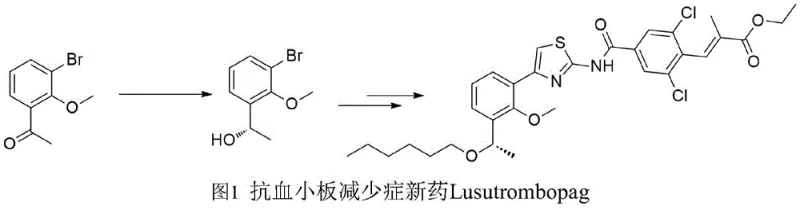
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric reduction of carbonyl groups to generate chiral alcohols, such as the key intermediate for Lusutrombopag, has relied heavily on stoichiometric chemical reducing agents or transition metal-catalyzed hydrogenation. The background art specifically highlights the use of borane reagents, such as borane-tetrahydrofuran or borane-dimethylsulfide complexes, often in conjunction with chiral oxazaborolidine catalysts like (R)- or (S)-2-methyl-CBS-oxazole borane. While these chemical methods can achieve high stereoselectivity, they impose severe limitations on large-scale manufacturing due to significant safety hazards and environmental burdens. Borane reagents are pyrophoric and moisture-sensitive, requiring stringent anhydrous conditions and specialized handling equipment that drastically increase capital expenditure and operational complexity. Furthermore, the use of organic solvents like tetrahydrofuran or toluene generates substantial volatile organic compound (VOC) emissions, complicating waste management and increasing the carbon footprint of the production facility. From a supply chain perspective, the reliance on expensive chiral catalysts and hazardous reagents creates vulnerability to raw material price fluctuations and regulatory scrutiny regarding residual impurities in the final Active Pharmaceutical Ingredient (API).
The Novel Approach
In stark contrast to the hazardous chemical protocols, the novel approach described in the patent leverages the power of enzyme engineering to achieve the same transformation with remarkable elegance and efficiency. By employing a specific carbonyl reductase derived from Novosphingobium, the process facilitates the asymmetric reduction of 1-(2-methoxy-3-bromophenyl) ethanone directly in an aqueous environment, effectively eliminating the need for dangerous borane reagents and anhydrous organic solvents. This biocatalytic system is designed to function at normal temperature and pressure, typically between 25°C and 32°C, which significantly reduces energy consumption compared to the heating or cooling often required in chemical synthesis. The integration of a cofactor regeneration system, utilizing either Glucose Dehydrogenase (GDH) or Formate Dehydrogenase (FDH), ensures that the expensive NADH cofactor is recycled continuously within the reaction mixture, allowing for catalytic rather than stoichiometric usage. This innovation not only simplifies the downstream processing by avoiding complex metal removal steps but also aligns perfectly with the principles of green chemistry, offering a reliable pharmaceutical intermediate supplier pathway that is both economically and ecologically superior.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise molecular recognition and catalytic activity of the engineered carbonyl reductase (CBR). The enzyme functions by binding the prochiral ketone substrate, 1-(2-methoxy-3-bromophenyl) ethanone, within its active site, where specific amino acid residues facilitate the transfer of a hydride ion from the reduced nicotinamide adenine dinucleotide (NADH) cofactor to the carbonyl carbon. The stereochemical outcome, favoring the (S)-enantiomer with an ee value greater than 99%, is dictated by the chiral environment of the enzyme's active pocket, which sterically hinders the approach of the hydride from the Re-face, forcing addition exclusively from the Si-face. This level of intrinsic selectivity is difficult to replicate with small-molecule catalysts without extensive optimization and often requires cryogenic temperatures to maintain fidelity. In this biological system, the enzyme maintains high fidelity even at elevated substrate concentrations, demonstrating a robust kinetic profile that is essential for industrial viability. The structural stability of the recombinant enzyme, expressed in E. coli BL21(DE3), allows it to withstand the presence of organic co-solvents like methanol, ethanol, or DMF, which are necessary to solubilize the hydrophobic aromatic substrate in the aqueous reaction medium.
Furthermore, the sustainability of the process is underpinned by the efficient cofactor regeneration mechanism, which is critical for preventing the accumulation of oxidized NAD+ that would otherwise halt the reaction. When Glucose Dehydrogenase is employed, it oxidizes glucose to gluconolactone, simultaneously reducing NAD+ back to NADH, thereby driving the equilibrium towards product formation. Alternatively, the Formate Dehydrogenase system oxidizes sodium formate to carbon dioxide, a gas that easily escapes the reaction mixture, irreversibly pulling the reaction forward according to Le Chatelier's principle. This dual-enzyme cascade ensures that the costly cofactor is used in catalytic amounts, drastically reducing the raw material cost per kilogram of product. The ability to operate at high substrate loadings, demonstrated in the patent examples up to 150g/L, indicates that the enzyme possesses high specific activity and tolerance to product inhibition. This mechanistic efficiency translates directly into higher space-time yields, meaning that manufacturers can produce more product in less time using smaller reactor volumes, a key metric for evaluating the commercial feasibility of any new synthetic route in the competitive API market.
How to Synthesize (S)-1-(2-methoxy-3-bromophenyl) ethanol Efficiently
Implementing this biocatalytic route requires a systematic approach to strain construction, fermentation, and biotransformation to maximize yield and purity. The process begins with the genetic engineering of the host organism, where the gene encoding the specific carbonyl reductase is cloned into an expression vector alongside the gene for the cofactor regeneration enzyme. Following transformation into a suitable host like E. coli, the recombinant cells are cultivated in a controlled fermentation environment to induce high-level protein expression. Once the biomass is harvested, it serves as the whole-cell biocatalyst for the reduction step. The reaction is conducted in a buffered aqueous system where pH control is vital to maintain enzyme activity and stability throughout the conversion. Detailed standard operating procedures regarding the specific ratios of enzymes, buffer compositions, and induction conditions are critical for reproducibility. For a comprehensive understanding of the exact experimental parameters and step-by-step execution required to replicate these results in a GMP setting, please refer to the standardized synthesis guide below.
- Construct recombinant E. coli strains co-expressing Carbonyl Reductase (CBR) and a cofactor regeneration enzyme such as Glucose Dehydrogenase (GDH) or Formate Dehydrogenase (FDH) to ensure sustainable NADH supply.
- Prepare the biocatalyst by fermenting the recombinant cells in LB medium, inducing expression with IPTG at optimal temperatures (25°C), and harvesting the biomass via centrifugation.
- Conduct the asymmetric reduction by resuspending the cells in phosphate buffer, adding the ketone substrate 1-(2-methoxy-3-bromophenyl) ethanone at high concentrations (up to 150g/L), and maintaining pH 7.0 until conversion exceeds 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical to biocatalytic synthesis offers profound strategic advantages that extend beyond simple yield metrics. The elimination of hazardous borane reagents removes a significant safety liability from the manufacturing site, reducing insurance costs and the need for specialized containment infrastructure. Moreover, the shift to an aqueous-based process significantly lowers the volume of organic solvents required, which directly correlates to reduced costs for solvent purchase, recovery, and disposal. This reduction in solvent intensity also simplifies the regulatory filing process, as residual solvent limits are easier to meet with water and ethanol compared to chlorinated or aromatic solvents. The high substrate tolerance of the enzyme means that production capacity can be maximized without expanding facility footprint, allowing for significant cost reduction in API manufacturing through improved asset utilization. Additionally, the robustness of the biological catalyst ensures consistent quality batch after batch, minimizing the risk of costly production failures or out-of-specification results that can disrupt supply continuity.
- Cost Reduction in Manufacturing: The economic benefits of this enzymatic route are driven primarily by the simplification of the input material list and the reduction of waste treatment expenses. By replacing expensive chiral chemical catalysts and stoichiometric borane reagents with recyclable whole-cell biocatalysts and inexpensive co-substrates like glucose or formate, the variable cost of goods sold is substantially lowered. The process avoids the need for complex downstream purification steps often required to remove heavy metals or boron impurities, which typically involve expensive scavenging resins or multiple crystallization cycles. Furthermore, the high space-time yield reported in the patent implies that the same reactor volume can produce significantly more product per day compared to traditional methods, effectively spreading fixed overhead costs over a larger output. These factors combine to create a leaner, more cost-efficient production model that enhances margin potential for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable raw materials, such as glucose and sodium formate, which are commodity chemicals with secure global supply lines, unlike specialized chiral ligands that may have single-source suppliers. The mild reaction conditions reduce the dependency on complex utility systems, such as deep-freeze cooling or high-pressure hydrogenation equipment, making the process easier to transfer between different manufacturing sites or contract manufacturing organizations (CMOs). This flexibility is crucial for mitigating risks associated with geopolitical instability or localized disruptions. The high conversion rates and minimal byproduct formation ensure that the throughput is predictable and reliable, allowing for more accurate demand forecasting and inventory management. Consequently, partners can rely on a steady flow of high-purity intermediates without the volatility often associated with complex chemical syntheses.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently safer and more straightforward than scaling exothermic chemical reductions involving pyrophoric reagents. The aqueous nature of the reaction minimizes fire hazards and explosion risks, facilitating easier approval from environmental health and safety (EHS) departments for capacity expansion. The process generates significantly less hazardous waste, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The reduction in VOC emissions and hazardous waste disposal needs not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a responsible producer. This environmental advantage is becoming a key differentiator in tender processes where pharmaceutical companies are mandated to prioritize green suppliers, ensuring long-term commercial viability and market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology for Lusutrombopag intermediate production. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value proposition of this synthetic route. The clarity provided here aims to bridge the gap between academic innovation and industrial application, ensuring all stakeholders have a unified understanding of the process capabilities.
Q: What are the advantages of using carbonyl reductase over chemical reduction for Lusutrombopag intermediates?
A: The enzymatic approach eliminates the need for hazardous borane reagents and expensive chiral catalysts like CBS, operating under mild aqueous conditions with superior stereoselectivity (ee > 99%) and significantly reduced environmental impact.
Q: Can this biocatalytic process handle high substrate concentrations for industrial scale-up?
A: Yes, the patented method demonstrates exceptional robustness, successfully converting substrate concentrations as high as 150g/L while maintaining yields above 95%, which indicates strong potential for cost-effective commercial manufacturing.
Q: How is the cofactor NADH regenerated in this synthetic route?
A: The process utilizes a coupled enzyme system where Glucose Dehydrogenase (GDH) or Formate Dehydrogenase (FDH) regenerates NADH from NAD+ using inexpensive co-substrates like glucose or sodium formate, ensuring catalytic efficiency without stoichiometric cofactor costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-(2-methoxy-3-bromophenyl) ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern pharmaceutical manufacturing and are fully equipped to bring this advanced technology to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that consistency is paramount in the pharmaceutical supply chain; therefore, our facilities are operated under stringent purity specifications and supported by rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity, potency, and chiral purity. Whether you require custom development of the enzymatic route or large-scale supply of the finished intermediate, our infrastructure is designed to meet the demanding requirements of global regulatory agencies, guaranteeing a supply of high-quality materials that support your drug development timelines.
We invite you to collaborate with us to leverage this innovative synthetic route for your Lusutrombopag projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this enzymatic process for your unique volume requirements. We encourage potential partners to contact us directly to obtain specific COA data from our pilot batches and to discuss detailed route feasibility assessments tailored to your production needs. Let us help you optimize your supply chain with a solution that delivers superior quality, sustainability, and value.
