Advanced Phosgene-Free Synthesis of 4,5-Dimethyl-1,3-dioxol-2-one for Commercial Scale-up
The pharmaceutical industry constantly seeks robust synthetic routes that balance efficiency with stringent safety standards, particularly for critical antibiotic precursors. Patent CN1261427C introduces a transformative chemical synthesis method for 4,5-dimethyl-1,3-dioxol-2-one, a vital intermediate used in the production of semi-synthetic penicillins such as Lenampicillin Hydrochloride. This innovation replaces the historically hazardous phosgene reagent with bis(trichloromethyl)carbonate, fundamentally altering the safety profile of the manufacturing process. By shifting from a gaseous toxicant to a manageable solid reagent, the technology addresses long-standing concerns regarding operator safety and environmental compliance. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while mitigating regulatory risks associated with controlled substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,5-dimethyl-1,3-dioxol-2-one relied heavily on the cyclization of 3-hydroxy-2-butanone using phosgene or diphosgene. While chemically effective, these traditional methods impose severe logistical and safety burdens on manufacturing facilities. Phosgene is a highly toxic gas that requires specialized containment systems, continuous monitoring, and extensive emergency response protocols, all of which drive up capital expenditure and operational costs. Furthermore, diphosgene, although a liquid, poses significant stability issues as it decomposes readily upon heating or contact with impurities, releasing phosgene gas unexpectedly. These inherent dangers complicate storage and transportation, creating bottlenecks in the supply chain and increasing the liability profile for any reliable pharmaceutical intermediates supplier attempting to scale production using legacy technologies.
The Novel Approach
The patented methodology offers a decisive break from these constraints by utilizing bis(trichloromethyl)carbonate (BTC) as a safe, solid alternative. This reagent allows for precise stoichiometric control and eliminates the need for complex gas-handling infrastructure. The process operates through a controlled two-stage temperature protocol, ensuring high conversion rates without the volatility associated with gaseous reagents. By removing the dependency on strictly controlled toxic gases, manufacturers can achieve cost reduction in antibiotic manufacturing through simplified facility requirements and reduced insurance premiums. This approach not only enhances the safety of the reaction environment but also streamlines the workflow, making it an ideal candidate for the commercial scale-up of complex heterocyclic intermediates in modern GMP facilities.
Mechanistic Insights into BTC-Mediated Cyclization
The core of this synthesis lies in the efficient activation of the hydroxyl group in 3-hydroxy-2-butanone by the carbonyl source provided by BTC. In the presence of an auxiliary agent such as N-methylpyrrole or triethylamine, the reaction initiates at low temperatures to form a chloroformate intermediate. This step is critical for minimizing side reactions and ensuring the structural integrity of the starting material. Subsequently, the reaction mixture undergoes a thermal cyclization phase where the temperature is elevated to facilitate ring closure. This mechanistic pathway ensures that the formation of the dioxolone ring proceeds with high selectivity, effectively suppressing the generation of polymeric byproducts or open-chain impurities that often plague less controlled acylation reactions.
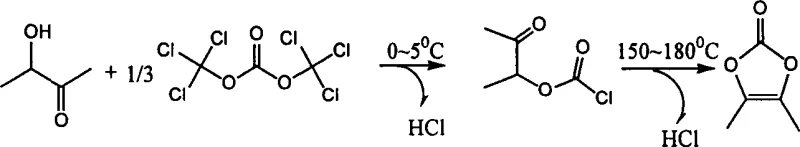
Impurity control is further enhanced by the specific solvent systems outlined in the patent, which include options like carbon tetrachloride or chloroform. These solvents provide the necessary solubility for the solid BTC while maintaining a stable reaction medium during the exothermic phases. The final product is isolated through a straightforward workup involving aqueous extraction and crystallization, yielding white crystals with exceptional purity. Analytical data from the patent examples confirms that this mechanism consistently delivers content levels above 99.0% as measured by HPLC. For quality assurance teams, this level of consistency is paramount, as it reduces the burden on downstream purification steps and ensures that the high-purity 4,5-dimethyl-1,3-dioxol-2-one meets the rigorous specifications required for subsequent antibiotic synthesis.
How to Synthesize 4,5-Dimethyl-1,3-dioxol-2-one Efficiently
Implementing this synthesis route requires careful attention to the thermal profile and reagent addition rates to maximize yield and safety. The process begins with the preparation of a reaction mixture containing the ketone substrate, a suitable organic solvent, and a base auxiliary. Once cooled to the initial low-temperature range, the BTC solution is introduced gradually to manage the evolution of hydrogen chloride. Following the initial acylation, the solvent is removed, and the residue is heated to induce cyclization. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature ramps that have been validated to produce optimal results.
- Mix 3-hydroxy-2-butanone with an organic solvent and auxiliary agent, then cool to 0-5°C.
- Dropwise add bis(trichloromethyl)carbonate solution and stir at 0-5°C for 2-3 hours.
- Remove solvent, heat residue to 150-180°C for cyclization, then purify via extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this phosgene-free technology offers substantial benefits that extend beyond mere chemical efficiency. The elimination of toxic gas handling significantly lowers the barrier to entry for production, allowing for a more diversified and resilient supplier base. This shift reduces the reliance on facilities with specialized hazardous gas permits, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to regulatory inspections or safety incidents. Furthermore, the use of solid reagents simplifies logistics, as transportation and storage do not require the same level of hazard mitigation as compressed gases, leading to smoother inventory management and reduced lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to bis(trichloromethyl)carbonate eliminates the need for expensive scrubbing systems and continuous gas monitoring equipment required for phosgene. This reduction in capital and operational expenditure translates directly into lower production costs. Additionally, the simplified workup procedure reduces solvent consumption and energy usage during the purification phase. By avoiding the complex safety protocols mandated for toxic gases, facilities can operate with greater efficiency, resulting in substantial cost savings that can be passed down the supply chain to benefit the final drug manufacturer.
- Enhanced Supply Chain Reliability: Sourcing solid reagents like BTC is inherently more stable than managing the supply of regulated toxic gases, which are often subject to strict transportation quotas and regional restrictions. This stability ensures a consistent flow of raw materials, preventing bottlenecks that could delay production schedules. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in environmental factors, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining the continuity of antibiotic production lines where interruptions can have significant clinical and commercial consequences.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, aligning with modern green chemistry principles and environmental regulations. The absence of phosgene residues simplifies waste treatment procedures, reducing the environmental footprint of the manufacturing site. Moreover, the reaction is highly scalable, as demonstrated by the patent examples which utilize standard laboratory glassware that can be directly translated to industrial reactor sizes. This scalability ensures that the method can meet increasing market demand without requiring disproportionate increases in safety infrastructure or waste management capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific technical disclosures and experimental data found within the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is bis(trichloromethyl)carbonate preferred over phosgene for this synthesis?
A: Bis(trichloromethyl)carbonate is a solid at room temperature, eliminating the severe safety hazards and complex containment infrastructure required for toxic phosgene gas, thereby reducing operational risk.
Q: What purity levels can be achieved with this novel cyclization method?
A: The patented process consistently achieves HPLC purity levels exceeding 99.0%, with specific examples demonstrating content up to 99.7% after standard crystallization workups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes common organic solvents and avoids gaseous reagents, making it highly scalable and easier to manage in standard chemical reactors compared to traditional phosgene routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Dimethyl-1,3-dioxol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that safe and efficient intermediates play in the global pharmaceutical supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN1261427C are translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4,5-dimethyl-1,3-dioxol-2-one meets the highest standards required for antibiotic synthesis. We are committed to delivering not just chemicals, but reliable solutions that enhance your production efficiency.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis route can benefit your specific applications. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless transition to this safer and more efficient manufacturing technology.
