Advanced Spirophosphoramidite Ligands for High-Efficiency Asymmetric Catalytic Hydrogenation
Advanced Spirophosphoramidite Ligands for High-Efficiency Asymmetric Catalytic Hydrogenation
The landscape of asymmetric synthesis is continually evolving, driven by the demand for higher purity and more robust catalytic systems in the production of active pharmaceutical ingredients. A pivotal advancement in this field is detailed in patent CN1156482C, which discloses a novel class of spirocyclic phosphoramidites characterized by their unique axial chirality and exceptional stability. These compounds represent a significant departure from traditional bidentate phosphine ligands, offering a monodentate architecture that maintains high stereoselectivity while overcoming the notorious sensitivity issues associated with earlier generations of catalysts. For R&D directors and process chemists, understanding the structural nuances of these ligands is critical, as they provide a versatile platform for the hydrogenation of dehydroamino acids, enamines, and itaconic acid derivatives. The core innovation lies in the spirobiindane backbone, which imparts rigid conformational control, ensuring that the chiral information is effectively transferred to the substrate during the catalytic cycle. 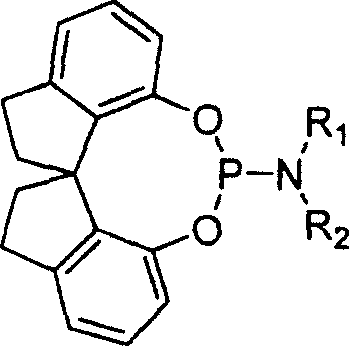
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial standard for asymmetric hydrogenation has relied heavily on C2-symmetric bidentate ligands such as BINAP, DIOP, and DuPHOS. While these compounds have proven effective in numerous academic and pilot-scale settings, they suffer from inherent limitations that pose significant challenges for large-scale manufacturing. The primary drawback is their chemical instability; many of these phosphines are highly susceptible to oxidation, necessitating rigorous exclusion of oxygen and moisture throughout the entire synthesis and usage lifecycle. This requirement for strict inert atmospheres, often involving complex nitrogen purging systems and specialized glovebox techniques, drastically increases operational complexity and cost. Furthermore, the synthesis of these traditional ligands often involves multi-step sequences with low overall yields, creating bottlenecks in the supply chain that can delay project timelines and inflate the cost of goods sold for the final API.
The Novel Approach
In contrast, the spirophosphoramidite technology introduced in the patent data offers a streamlined and robust alternative that directly addresses these stability and cost concerns. By utilizing a spirocyclic diphenol (SPINOL) precursor reacting with substituted phosphoramidites, the synthesis becomes a direct, one-step condensation that is far easier to control and scale. 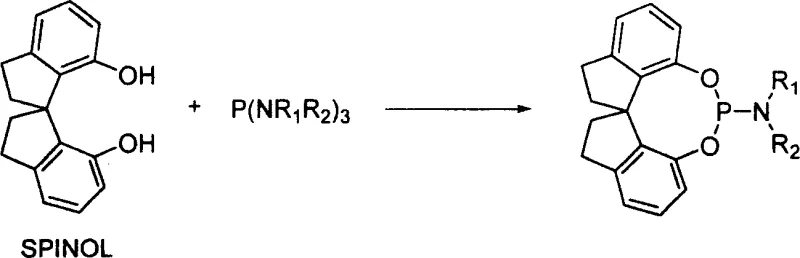
This novel approach eliminates the need for sensitive phosphine bonds that are prone to degradation, replacing them with a more stable phosphoramidite linkage. The resulting ligands retain the necessary chiral environment through the rigid spiro-indane structure but do so with a resilience that allows for easier handling and storage. For procurement managers, this translates to a reduction in the specialized infrastructure required for catalyst management, while for supply chain heads, it意味着 a more reliable source of chiral inducers that are less likely to degrade during transit or storage, thereby ensuring consistent batch-to-batch quality in the final drug substance.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The efficacy of these spirophosphoramidites stems from their ability to form well-defined complexes with rhodium(I) centers, creating a chiral pocket that dictates the facial selectivity of hydrogen addition. When coordinated with Rh(I), the monodentate nature of the ligand allows for a dynamic equilibrium that can adapt to the substrate, yet the bulky spiro-backbone provides sufficient steric hindrance to enforce high enantioselectivity. This mechanism is particularly effective for substrates containing olefinic bonds adjacent to electron-withdrawing groups, such as dehydroamino acids and enamines. The axial chirality of the spiro-indane framework ensures that the catalyst-substrate complex adopts a specific conformation that favors the formation of one enantiomer over the other, leading to the reported e.e. values exceeding 99%. 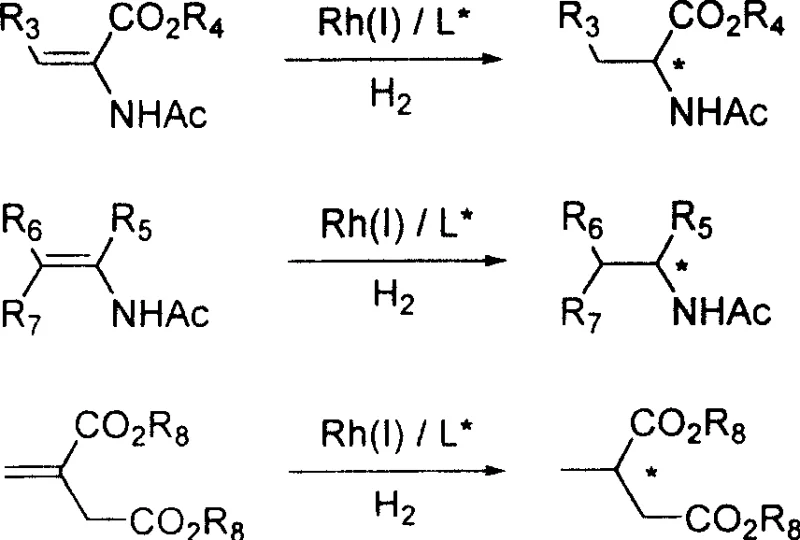
From an impurity control perspective, the high selectivity of this system minimizes the formation of the unwanted enantiomer, which is often the most difficult impurity to remove during downstream purification. In traditional processes, low selectivity might require costly recrystallization steps or chiral chromatography to meet regulatory purity standards. However, the intrinsic selectivity of the spirophosphoramidite-Rhodium system reduces the burden on purification units, simplifying the overall process flow. This mechanistic advantage is crucial for regulatory filings, as it demonstrates a robust control strategy for chiral purity, a key quality attribute for any chiral pharmaceutical intermediate intended for global markets.
How to Synthesize Spirophosphoramidite Efficiently
The synthesis protocol outlined in the patent data provides a clear roadmap for producing these high-value ligands with excellent yields and purity. The process involves reacting spirocyclic diphenol with a substituted phosphoramidite in a non-polar solvent such as toluene or benzene under a nitrogen stream. The reaction conditions are remarkably mild, typically requiring temperatures between 80-150°C for a duration of 1 to 10 hours, depending on the specific substituents used. This thermal stability allows for flexible processing windows, reducing the risk of thermal runaway or decomposition that can plague more sensitive organometallic syntheses. Following the reaction, the crude product can be purified using standard silica gel column chromatography, yielding white crystals or viscous liquids with high purity suitable for immediate use in catalytic applications.
- Combine spirocyclic diphenol (SPINOL) and substituted phosphoramidite in a reactor with a molar ratio of 1: 1 to 1:2.
- Add anhydrous toluene or benzene as the solvent and maintain a nitrogen atmosphere to prevent oxidation.
- Heat the reaction mixture to a temperature range of 80-150°C for 1-10 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of spirophosphoramidite ligands offers compelling economic and logistical benefits. The shift from unstable, multi-step bidentate ligands to this stable, single-step condensation product fundamentally alters the cost structure of chiral synthesis. By eliminating the need for expensive metal scavengers often required to remove degraded phosphine byproducts, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. Furthermore, the robustness of the ligand means that inventory can be held for longer periods without degradation, providing a buffer against supply chain disruptions and allowing for more strategic purchasing decisions.
- Cost Reduction in Manufacturing: The simplified synthetic route for the ligand itself, combined with its high turnover number and selectivity in the hydrogenation step, leads to substantial cost savings. The elimination of complex protection-deprotection sequences found in traditional ligand synthesis reduces raw material consumption and waste generation. Additionally, the ability to operate under less stringent atmospheric conditions lowers the capital expenditure required for specialized reactor setups, making the technology accessible for both pilot and commercial scales without prohibitive upfront investment.
- Enhanced Supply Chain Reliability: The chemical stability of spirophosphoramidites ensures that the material remains potent during storage and transportation, reducing the lead time for high-purity chiral ligands. Unlike air-sensitive catalysts that require cold chain logistics or argon-blanketed shipping, these ligands can be handled with standard precautions, simplifying the logistics network. This reliability is critical for maintaining continuous production schedules in API manufacturing, where any delay in catalyst delivery can halt the entire production line and result in significant revenue loss.
- Scalability and Environmental Compliance: The process utilizes common solvents like toluene and operates at moderate temperatures, facilitating easy scale-up from gram to ton quantities. The high atom economy of the condensation reaction and the absence of heavy metal contaminants in the ligand structure align well with green chemistry principles. This environmental compatibility simplifies waste disposal and regulatory compliance, reducing the administrative burden on EHS teams and minimizing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of spirophosphoramidite technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, offering a factual basis for decision-making. Understanding these details helps bridge the gap between academic potential and commercial reality, ensuring that all stakeholders have a clear view of the technology's capabilities and limitations.
Q: How does the stability of spirophosphoramidites compare to traditional ligands like BINAP?
A: Unlike traditional bidentate ligands such as BINAP or DIOP which often require strict inert atmospheres due to poor stability, spirophosphoramidites exhibit significantly enhanced stability, allowing for more flexible handling and storage conditions without compromising catalytic performance.
Q: What enantiomeric excess (ee) values can be achieved using these ligands?
A: When applied to the asymmetric catalytic hydrogenation of dehydroamino acids, enamines, and itaconic acids, these ligands demonstrate exceptional stereoselectivity, with documented e.e. values consistently reaching above 99% in optimized conditions.
Q: Are these ligands suitable for large-scale pharmaceutical manufacturing?
A: Yes, the synthesis utilizes readily available raw materials like SPINOL and operates under relatively mild thermal conditions (80-150°C), making the process highly scalable and economically viable for commercial production of chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirophosphoramidite Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of spirophosphoramidite meets the exacting standards required for GMP manufacturing. Our commitment to quality ensures that the high enantioselectivity promised by the patent data is realized consistently in your production runs.
We invite you to collaborate with us to optimize your chiral synthesis routes and achieve your cost and efficiency targets. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning, ensuring that you have a reliable partner for your complex chemical requirements.
