Advanced Non-Phosgene Synthesis of 1,5-Naphthalene Diisocyanate for High-Performance Polyurethane Manufacturing
The chemical industry is currently witnessing a significant paradigm shift towards greener and safer synthesis methodologies, particularly in the production of high-value isocyanates. Patent CN1394851A introduces a groundbreaking approach for the synthesis of 1,5-naphthalene diisocyanate (NDI), a critical monomer for advanced polyurethane applications. This technology addresses the longstanding challenges associated with traditional phosgene-based routes, offering a non-phosgene, one-step synthesis pathway that utilizes bis(trichloromethyl)carbonate (BTC) as a safe, solid alternative. For R&D directors and procurement specialists, this patent represents a viable solution to complex process engineering issues, promising high purity and operational safety. The method employs a liquid phase reaction and microemulsion technique, ensuring that the reaction conditions remain mild while achieving exceptional conversion rates. By transitioning away from hazardous gaseous reagents, manufacturers can significantly mitigate environmental risks and streamline their production workflows, aligning with modern regulatory standards for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the global production of 1,5-naphthalene diisocyanate has relied heavily on the phosgene method, a process fraught with significant technical and safety hurdles. Conventional phosgene routes require the use of excessive amounts of highly toxic gaseous and liquid phosgene, necessitating extreme caution in handling and storage. These processes typically operate under high temperature and pressure conditions, which demand sophisticated and expensive equipment designed to withstand such harsh environments. Furthermore, the technological complexity of managing phosgene reactions often leads to lengthy process flows that are difficult to control precisely. A major drawback of these traditional methods is the formation of polymeric by-products that are challenging to eliminate and separate from the final product, compromising purity. The severe pollution associated with toxic phosgene emissions creates a harsh operating environment, posing serious health risks to personnel and complicating waste management protocols. Consequently, the storage, transportation, and usage of phosgene are extremely inconvenient and regulated, creating bottlenecks in the supply chain.
The Novel Approach
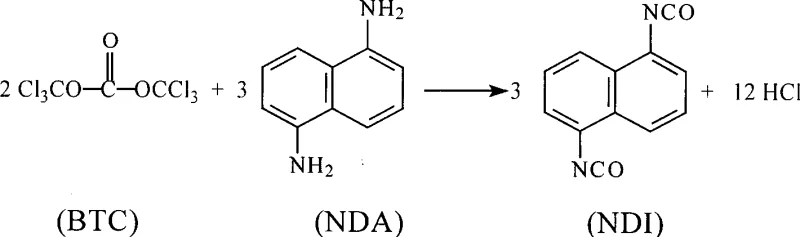
In stark contrast to the hazardous legacy methods, the novel approach detailed in the patent utilizes bis(trichloromethyl)carbonate (BTC), often referred to as triphosgene, as a stable and non-toxic solid crystalline reagent. This substitution fundamentally transforms the safety profile of the manufacturing facility by eliminating the need for complex phosgene generation and storage infrastructure. The new method facilitates a one-step synthesis directly from 1,5-diaminonaphthalene (NDA) and BTC in an inert solvent, drastically simplifying the process flow. Operationally, this route is characterized by mild reaction conditions that do not require the extreme temperatures or pressures seen in phosgene methods, allowing for the use of conventional reaction vessels. The high reactivity of BTC ensures accurate reaction stoichiometry and short reaction times, leading to substantially improved efficiency. Moreover, the process is designed to be pollution-free and environmentally friendly, as the solid nature of BTC prevents the release of toxic gases during storage and transportation. This innovation not only enhances operator safety but also reduces the overall operational costs associated with safety compliance and waste treatment.
Mechanistic Insights into BTC-Mediated Isocyanate Formation
The core of this technological advancement lies in the precise mechanistic interaction between 1,5-diaminonaphthalene and bis(trichloromethyl)carbonate within an inert organic solvent system. The reaction proceeds through a nucleophilic attack where the amino groups of the naphthalene derivative interact with the carbonyl carbon of the BTC molecule. This interaction is carefully managed by controlling the addition temperature between -10°C and 80°C, followed by a reaction phase at 15°C to 150°C under a nitrogen atmosphere. The use of a microemulsion reaction system enhances the contact area between the reactants, facilitating a more uniform and rapid conversion. This specific catalytic environment ensures that the transformation from diamine to diisocyanate occurs with high selectivity, minimizing the formation of unwanted side products. The process is robust enough to handle the inevitable formation of small amounts of 1,5-diaminonaphthalene hydrochloride, which further reacts with BTC to contribute to the final yield. This mechanistic efficiency is critical for maintaining high product quality and ensuring that the synthesis remains economically viable on a commercial scale.
Impurity control is a paramount concern for R&D directors focusing on the quality of polyurethane intermediates, and this synthesis route offers distinct advantages in this regard. The high conversion rate and selectivity of the reaction mean that the process approximates a quantitative proportioning reaction, leaving very little unreacted starting material. Aside from the manageable formation of 1,5-diaminonaphthalene hydrochloride, no other significant side reaction products are detected during the process. This hydrochloride salt is not merely a waste product; it actively participates in the reaction with BTC to generate additional 1,5-naphthalene diisocyanate, thereby maximizing atom economy. The absence of complex polymeric by-products, which are common in phosgene methods, simplifies the downstream purification steps significantly. The resulting product exhibits a sharp melting point range, indicative of high structural purity, which is essential for the performance of the final polyurethane materials. By minimizing impurities at the source, the need for extensive and costly purification processes is reduced, further enhancing the economic attractiveness of this synthetic route.
How to Synthesize 1,5-Naphthalene Diisocyanate Efficiently
Implementing this synthesis route requires a systematic approach to reagent preparation and reaction control to ensure optimal yields and safety. The process begins with the precise配制 of inert organic solutions of both 1,5-diaminonaphthalene and bis(trichloromethyl)carbonate at specific concentrations to maintain reaction homogeneity. Detailed standardized synthesis steps are crucial for replicating the high yields reported in the patent data, particularly regarding temperature gradients and stirring speeds. The following guide outlines the critical operational parameters necessary for successful execution of this non-phosgene pathway. Adhering to these protocols ensures that the reaction proceeds smoothly without the formation of excessive by-products or safety incidents.
- Preparation of inert organic solutions of 1,5-diaminonaphthalene and bis(trichloromethyl)carbonate at specific concentrations.
- Controlled addition of amine solution to BTC solution under nitrogen atmosphere at -10 to 80°C followed by reaction at 15 to 150°C.
- Distillation of reaction liquid, solvent recovery, and filtration to isolate high-purity 1,5-naphthalene diisocyanate solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this BTC-based synthesis method offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of gaseous phosgene from the supply chain removes a significant logistical burden, as BTC is a stable solid that is far easier and safer to transport and store. This shift drastically simplifies the regulatory compliance landscape, reducing the costs associated with hazardous material handling and emergency response planning. The simplified process flow, which operates under mild conditions, allows for the use of standard industrial equipment rather than specialized high-pressure reactors, leading to significant capital expenditure savings. Furthermore, the high yield and selectivity of the reaction minimize raw material waste, directly contributing to cost reduction in manufacturing operations. The ability to recycle solvents and recover by-products like HCl for chlorine regeneration enhances the overall sustainability profile, appealing to environmentally conscious stakeholders. These factors collectively create a more resilient and cost-effective supply chain for high-purity polyurethane intermediates.
- Cost Reduction in Manufacturing: The adoption of this non-phosgene route eliminates the need for expensive phosgene generation equipment and the rigorous safety infrastructure required to handle toxic gases. By utilizing solid BTC, facilities can avoid the high operational costs associated with gas containment systems and continuous monitoring. The high reaction yield, which can exceed 98% under optimized conditions, ensures that raw material utilization is maximized, reducing the cost per unit of production. Additionally, the simplified downstream processing due to fewer impurities lowers the energy and resource consumption required for purification. These cumulative effects result in a leaner manufacturing process with a significantly improved margin structure.
- Enhanced Supply Chain Reliability: Sourcing and handling solid bis(trichloromethyl)carbonate is inherently less risky and more reliable than managing gaseous phosgene supplies. The stability of BTC allows for longer storage times and more flexible transportation schedules, reducing the pressure on just-in-time delivery systems. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream polyurethane manufacturers. The reduced regulatory hurdles associated with non-phosgene reagents also mean fewer delays in customs and logistics, ensuring a smoother flow of materials. Consequently, supply chain managers can achieve greater predictability and stability in their raw material procurement strategies.
- Scalability and Environmental Compliance: The mild reaction conditions and use of conventional equipment make this process highly scalable from pilot plants to full commercial production without significant re-engineering. The environmental benefits are profound, as the process generates minimal pollution and avoids the release of toxic phosgene into the atmosphere. The by-product HCl can be electrolyzed to recover chlorine, which can then be recycled to prepare more BTC, creating a closed-loop system that approaches zero emission. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly strict environmental regulations. Such scalability and compliance ensure long-term viability and reduce the risk of production shutdowns due to regulatory non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the feasibility and benefits of the BTC route. Understanding these details is essential for stakeholders evaluating the potential integration of this method into their existing production frameworks. The information provided here underscores the technical robustness and commercial viability of the process.
Q: What are the primary safety advantages of using BTC over phosgene for NDI synthesis?
A: The use of bis(trichloromethyl)carbonate (BTC) eliminates the need for handling toxic gaseous phosgene, significantly reducing operational risks and equipment complexity.
Q: What yield can be expected from this non-phosgene synthesis method?
A: According to patent data, the process achieves high conversion rates with yields reaching over 98% under optimized conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions and conventional equipment, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Naphthalene Diisocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this non-phosgene synthesis route for the production of high-performance polyurethane intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,5-naphthalene diisocyanate meets the highest quality standards. We are committed to leveraging this advanced technology to provide our partners with a reliable and sustainable source of critical chemical intermediates. Our team is dedicated to optimizing the process parameters to maximize yield and minimize environmental impact, aligning with the global push for greener manufacturing practices.
We invite you to collaborate with us to explore how this synthesis method can enhance your supply chain efficiency and product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to reach out to request specific COA data and route feasibility assessments to determine the best fit for your operations. By partnering with us, you gain access to not just a product, but a comprehensive solution that addresses both technical and commercial challenges in the specialty chemical sector. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
