Advanced Pyrimido-Fused Ring Compounds for Next-Generation Agrochemical Fungicides
The agricultural sector faces an escalating challenge in managing pathogenic fungi due to the rapid development of resistance against traditional fungicides. Addressing this critical gap, patent CN110964037B introduces a novel class of pyrimido-fused ring-containing compounds that demonstrate exceptional bactericidal potential. These molecules, characterized by a versatile general formula I, represent a significant leap forward in agrochemical intermediate technology, offering a robust solution for controlling devastating crop diseases such as downy mildew and powdery mildew. The structural innovation lies in the specific fusion of the isoxazole and pyrimidine rings, coupled with a highly modifiable side chain architecture that allows for precise tuning of biological activity. As a reliable agrochemical intermediate supplier, understanding the depth of this chemical space is vital for developing next-generation crop protection strategies that ensure food security and yield stability.
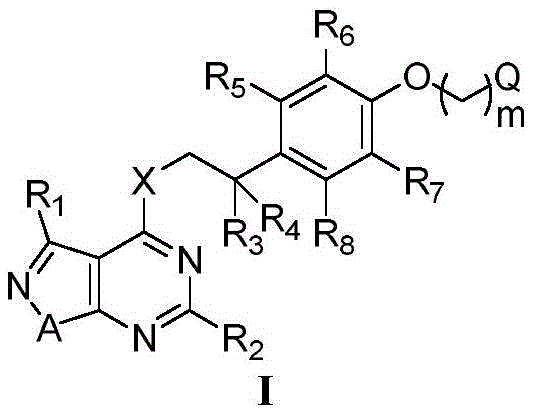
The development of effective fungicides often hinges on overcoming the limitations of prior art structures which may suffer from narrow spectra of activity or poor environmental profiles. Conventional methods relying on simpler pyrimidine scaffolds often fail to provide the necessary potency against resistant strains of oomycetes and basidiomycetes. Furthermore, older synthetic routes frequently involve harsh conditions or expensive transition metal catalysts that complicate purification and increase waste generation. In contrast, the novel approach detailed in the patent utilizes a convergent synthesis strategy that maximizes structural diversity while maintaining operational simplicity. By employing readily available halo-alkyl ethers and substituted phenethylamines, the process avoids complex catalytic cycles, thereby significantly reducing the impurity profile and enhancing the overall purity of the final active ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fungicidal chemistries, particularly those based on earlier generations of isoxazolo pyrimidines disclosed in literature such as Pest Management Science (2008), often exhibit restricted structural variability which limits their ability to combat evolving pathogen resistance. These legacy compounds typically rely on fixed substitution patterns that do not allow for the fine-tuning of physicochemical properties necessary for optimal plant uptake and translocation. Additionally, the synthesis of these older analogues frequently requires multi-step sequences with low atom economy, leading to higher production costs and significant environmental burdens due to solvent waste. The reliance on specific, hard-to-source intermediates can also create supply chain bottlenecks, making it difficult for manufacturers to scale production to meet global agricultural demands consistently and reliably.
The Novel Approach
The innovative methodology presented in this patent overcomes these hurdles by introducing a modular synthetic platform centered around the nucleophilic substitution of a chloro-pyrimido intermediate. This approach allows for the independent variation of up to ten different substituent positions (R1 through R10), enabling chemists to systematically explore structure-activity relationships to identify lead candidates with superior efficacy. The reaction conditions are notably mild, typically proceeding at temperatures between 20°C and 100°C in common organic solvents like tetrahydrofuran or DMF, which simplifies process control and reduces energy consumption. This flexibility not only accelerates the discovery of high-purity agrochemical intermediates but also facilitates the rapid adaptation of the synthesis to produce specific analogues required for regional pest management needs.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Etherification
The core chemical transformation driving the synthesis of these potent fungicides involves a sequential nucleophilic aromatic substitution followed by an etherification reaction. Initially, the electron-deficient chloro-pyrimido intermediate (Formula IV) acts as an electrophile, reacting with the nucleophilic amine group of a substituted phenethylamine (Formula VII). This step is facilitated by the presence of a base, such as triethylamine or potassium carbonate, which scavenges the generated hydrogen chloride, driving the equilibrium towards the formation of the amino-linked intermediate (Formula V). The electronic nature of the pyrimidine ring, activated by the adjacent nitrogen atoms and the fused isoxazole system, significantly lowers the activation energy for this displacement, allowing the reaction to proceed efficiently without the need for exotic catalysts.
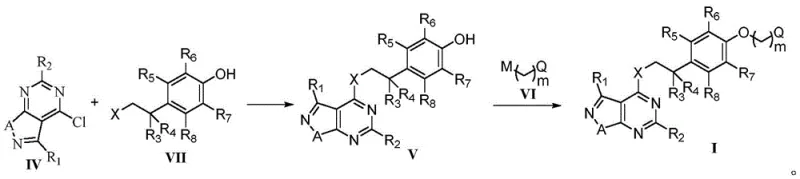
Following the formation of the amino-linkage, the second critical stage involves the etherification of the phenolic hydroxyl group on the intermediate. This is achieved by reacting the phenol with an alkyl halide (Formula VI) under basic conditions, effectively installing the diverse side chains that define the specific biological profile of each compound variant. The choice of base and solvent in this step is crucial for minimizing side reactions such as O-alkylation of the pyrimidine nitrogen or elimination reactions of the alkyl halide. By carefully controlling the stoichiometry and temperature, the process ensures high regioselectivity for the phenolic oxygen, resulting in a clean product profile that simplifies downstream purification. This mechanistic precision is key to achieving the stringent purity specifications required for commercial agrochemical registration and field application.
How to Synthesize Pyrimido-Fused Ring Compounds Efficiently
The synthesis of these high-value intermediates follows a logical three-stage protocol designed for scalability and reproducibility in an industrial setting. The process begins with the preparation of the key chloro-heterocycle, followed by coupling with the amine linker, and concludes with the installation of the terminal ether group. Each step has been optimized to balance reaction rate with selectivity, ensuring that impurities are kept to a minimum throughout the sequence. For detailed operational parameters, including specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized guide below which outlines the critical process controls necessary for successful manufacturing.
- Preparation of the key chloro-intermediate (Formula IV) via cyclization of amino-isoxazole precursors using phosphorus oxychloride.
- Nucleophilic substitution reaction between the chloro-intermediate and substituted phenethylamines (Formula VII) under basic conditions to form the amino-linked intermediate.
- Final etherification step reacting the phenolic intermediate with alkyl halides (Formula VI) to install the diverse side chains required for optimal biological activity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical performance. The reliance on commodity chemicals for the starting materials, such as benzyl chloride derivatives and common phenethylamines, mitigates the risk of raw material shortages and price volatility often associated with specialized reagents. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal removal steps, which are a significant cost driver in pharmaceutical and agrochemical manufacturing. This streamlined process translates directly into a more resilient supply chain capable of delivering consistent quality at a competitive cost structure.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for precious metal catalysts and complex purification protocols, leading to substantial cost savings in raw material and processing expenses. By utilizing straightforward nucleophilic substitutions in common solvents, the process reduces energy consumption and waste disposal costs associated with hazardous reagents. The high atom economy of the etherification step further contributes to lower production costs per kilogram of active ingredient, enhancing the overall profitability of the fungicide portfolio.
- Enhanced Supply Chain Reliability: The use of widely available commercial intermediates ensures a stable and secure supply of raw materials, reducing the risk of production delays caused by sourcing bottlenecks. The robustness of the reaction conditions allows for manufacturing in diverse geographic locations, facilitating regional production strategies that minimize logistics lead times. This reliability is crucial for meeting the seasonal demands of the agricultural market, ensuring that farmers have access to critical crop protection products exactly when they are needed most.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from laboratory bench scale to potential multi-ton commercial production without significant re-optimization. The avoidance of toxic heavy metals and the use of recyclable solvents align with increasingly stringent environmental regulations, reducing the regulatory burden and permitting risks for manufacturing sites. This environmental compatibility not only safeguards the ecosystem but also enhances the brand reputation of the end-product among environmentally conscious consumers and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrimido-fused ring compounds. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is essential for making informed decisions about integrating these intermediates into your existing product development pipelines.
Q: What is the primary biological advantage of these pyrimido-fused ring compounds?
A: These compounds exhibit broad-spectrum fungicidal activity, specifically showing over 80% control efficiency against cucumber downy mildew at concentrations as low as 6.25ppm, outperforming many conventional isoxazolo pyrimidines.
Q: Are the starting materials for this synthesis commercially viable?
A: Yes, the key intermediates such as the halo-alkyl ethers (Formula VI) and substituted phenethylamines (Formula VII) are largely commercially available or easily synthesized from common industrial feedstocks, ensuring supply chain stability.
Q: How does the structural flexibility impact formulation development?
A: The extensive variability in R-groups (R1 through R10) allows for fine-tuning of physicochemical properties like solubility and lipophilicity, facilitating the development of diverse formulations such as wettable powders, suspensions, and emulsions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimido-Fused Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective agrochemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrimido-fused ring compound meets the highest standards of quality and consistency required for global agricultural applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next generation of fungicidal products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
