Advanced Halogen-Fluorine Exchange Technology for Commercial Scale Fluorinated Intermediates
Advanced Halogen-Fluorine Exchange Technology for Commercial Scale Fluorinated Intermediates
The global demand for high-purity fluorinated intermediates continues to surge across the pharmaceutical and electronic materials sectors, driven by the unique physicochemical properties fluorine imparts to organic molecules. Patent CN1413177A introduces a transformative methodology for preparing these critical compounds through an improved halogen-fluorine exchange reaction, commonly known as the Halex reaction. This innovation specifically addresses the longstanding challenges of thermal instability and low space-time yields associated with conventional fluorination processes. By utilizing specific phosphonium-based catalysts of general formula (II), the process enables reactions to proceed at significantly milder temperatures, ranging from 40 to 260°C, compared to the harsh conditions traditionally required. This technical breakthrough not only enhances the purity profile of the resulting fluorinated aromatics but also opens new avenues for cost-effective manufacturing of complex intermediates used in liquid crystal mixtures and active pharmaceutical ingredients. For procurement and supply chain leaders, this represents a pivotal shift towards more sustainable and reliable production methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the nucleophilic substitution of halogen atoms with fluorine on aromatic rings has been plagued by severe operational constraints that hinder commercial scalability and efficiency. Traditional Halex reactions typically necessitate extremely high reaction temperatures, often falling between 200 and 300°C, to overcome the activation energy barriers inherent in breaking carbon-halogen bonds. These elevated thermal conditions frequently lead to the formation of considerable amounts of decomposition products, thereby compromising the purity of the final API intermediate or electronic chemical. Furthermore, conventional methods generally require the presence of solvents to maintain reaction homogeneity, which inevitably lowers the space-time yield compared to solvent-free preparations. The phase transfer catalysts historically employed, such as quaternary alkyl ammonium salts or crown ethers, often exhibit relatively low activity and suffer from moderate thermal stability at the requisite reaction temperatures. Consequently, manufacturers face significant challenges in managing byproduct profiles and ensuring consistent batch-to-batch quality without incurring excessive purification costs.
The Novel Approach
In stark contrast to legacy technologies, the novel approach detailed in the patent leverages the exceptional stability and catalytic activity of tetrakis(dialkylamino)phosphonium salts to drive the fluorination process with unprecedented efficiency. 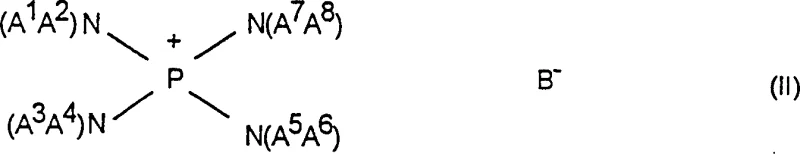 As illustrated by the general structure of the catalyst, the phosphonium center provides a robust framework that resists thermal degradation even under prolonged heating. It has been surprisingly found that the use of these formula (II) compounds results in an accelerated reaction rate, allowing the halogen-fluorine exchange to be conducted under considerably milder conditions. This acceleration translates directly into shorter reaction times and the ability to operate at lower temperatures, which substantially suppresses the formation of undesired thermal decomposition byproducts. Moreover, the versatility of this catalyst system allows for the possibility of running the reaction in the absence of solvents, thereby eliminating the need for extensive solvent recovery infrastructure and improving the overall volumetric productivity of the manufacturing plant. This technological leap offers a reliable agrochemical intermediate supplier or pharma partner a distinct competitive advantage in terms of process robustness and environmental compliance.
As illustrated by the general structure of the catalyst, the phosphonium center provides a robust framework that resists thermal degradation even under prolonged heating. It has been surprisingly found that the use of these formula (II) compounds results in an accelerated reaction rate, allowing the halogen-fluorine exchange to be conducted under considerably milder conditions. This acceleration translates directly into shorter reaction times and the ability to operate at lower temperatures, which substantially suppresses the formation of undesired thermal decomposition byproducts. Moreover, the versatility of this catalyst system allows for the possibility of running the reaction in the absence of solvents, thereby eliminating the need for extensive solvent recovery infrastructure and improving the overall volumetric productivity of the manufacturing plant. This technological leap offers a reliable agrochemical intermediate supplier or pharma partner a distinct competitive advantage in terms of process robustness and environmental compliance.
Mechanistic Insights into Phosphonium-Catalyzed Nucleophilic Substitution
The underlying mechanism of this enhanced fluorination process relies on the facilitation of nucleophilic attack by the fluoride ion on the electron-deficient aromatic ring system. In the context of aromatic compounds, particularly those activated by electron-withdrawing substituents, the reaction proceeds via a nucleophilic substitution pathway where the halogen leaving group is displaced. The phosphonium catalyst likely functions by effectively solvating or activating the fluoride source, such as potassium fluoride or cesium fluoride, thereby increasing its nucleophilicity in the reaction medium. This activation is crucial for overcoming the kinetic barriers associated with the substitution, especially when dealing with less reactive substrates like chlorobenzenes or chloropyridines that lack strong activating groups. The presence of the catalyst ensures that the fluoride ion remains available for the substitution event without being sequestered in unreactive lattice structures, which is a common issue in solid-state fluorinations.
Furthermore, the scope of substrates amenable to this transformation is remarkably broad, encompassing both activated and non-activated aromatic systems as defined by general formula (IV). 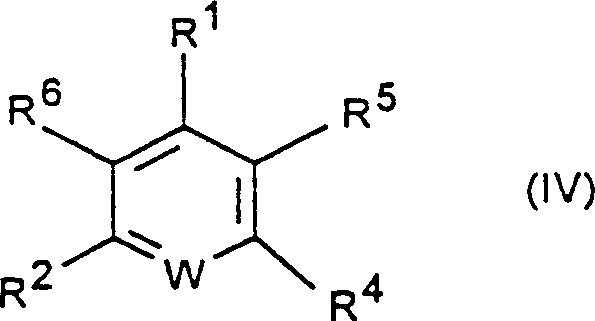 The patent delineates that compounds containing substituents such as nitro, cyano, trifluoromethyl, or carbonyl groups at ortho or para positions relative to the leaving halogen are particularly reactive due to the stabilization of the Meisenheimer complex intermediate. However, the catalytic system is sufficiently potent to facilitate exchange even in the absence of these strong activating groups, provided the temperature is adjusted within the optimal 120 to 200°C range. Impurity control is inherently improved because the milder thermal regime prevents the degradation of sensitive functional groups that might otherwise decompose under traditional Halex conditions. This mechanistic understanding allows process chemists to predict reactivity trends and optimize conditions for specific target molecules, ensuring high-purity fluorinated compounds suitable for stringent regulatory applications.
The patent delineates that compounds containing substituents such as nitro, cyano, trifluoromethyl, or carbonyl groups at ortho or para positions relative to the leaving halogen are particularly reactive due to the stabilization of the Meisenheimer complex intermediate. However, the catalytic system is sufficiently potent to facilitate exchange even in the absence of these strong activating groups, provided the temperature is adjusted within the optimal 120 to 200°C range. Impurity control is inherently improved because the milder thermal regime prevents the degradation of sensitive functional groups that might otherwise decompose under traditional Halex conditions. This mechanistic understanding allows process chemists to predict reactivity trends and optimize conditions for specific target molecules, ensuring high-purity fluorinated compounds suitable for stringent regulatory applications.
How to Synthesize Fluorinated Aromatics Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the fluoride source and the loading of the phosphonium catalyst to maximize conversion while minimizing waste. The patent outlines a straightforward procedure where the halo-aromatic starting material is combined with an alkali metal fluoride and the catalyst, optionally in a dipolar aprotic solvent like dimethyl sulfoxide or thiophene. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with the halo-aromatic substrate, alkali metal fluoride (e.g., KF), and the phosphonium catalyst (0.5-35 wt%).
- Heat the mixture to 120-200°C under inert atmosphere, optionally using dipolar aprotic solvents like DMSO or TMS, or run solvent-free.
- Monitor conversion via HPLC or GC, then cool, extract with dichloromethane, filter salts, and purify the fluorinated product by distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on optimizing their supply chain resilience and reducing the total cost of ownership for critical raw materials, this patented technology offers substantial strategic benefits. The shift towards a more thermally stable catalyst system directly addresses the volatility and inconsistency often seen in legacy fluorination processes, leading to a more predictable manufacturing timeline. By enabling operations at lower temperatures and potentially without solvents, the process significantly reduces the energy consumption and waste treatment burdens associated with high-temperature organic synthesis. These efficiencies translate into a more robust supply chain capable of meeting tight delivery schedules without the risk of batch failures due to thermal runaway or catalyst decomposition. Ultimately, adopting this methodology positions a company as a cost reduction in electronic chemical manufacturing leader by streamlining the production of high-value fluorinated building blocks.
- Cost Reduction in Manufacturing: The elimination of expensive and thermally unstable phase transfer catalysts, such as crown ethers or specialized ammonium salts, results in direct material cost savings. Additionally, the potential for solvent-free operation removes the capital and operational expenditures related to solvent recovery, distillation, and disposal systems. The higher space-time yields achieved through accelerated reaction kinetics mean that existing reactor assets can produce more product per unit of time, effectively lowering the fixed cost allocation per kilogram of output. Qualitative analysis suggests that the reduced formation of decomposition products also minimizes the load on purification units, further driving down processing costs.
- Enhanced Supply Chain Reliability: The use of readily available starting materials, such as common chloro-aromatics and alkali metal fluorides, ensures that raw material sourcing remains stable and unaffected by niche supply constraints. The robustness of the phosphonium catalyst against thermal degradation means that the process is less susceptible to variations in heating rates or minor temperature fluctuations, leading to consistent batch quality. This reliability is critical for maintaining continuous production schedules and avoiding delays that could impact downstream formulation or drug substance manufacturing. Suppliers leveraging this technology can offer greater assurance of continuity of supply for high-purity fluorinated intermediates.
- Scalability and Environmental Compliance: The ability to conduct the reaction under milder conditions simplifies the engineering requirements for scale-up, as there is less demand on cooling systems and pressure containment compared to traditional high-temperature processes. Reduced solvent usage or the option for solvent-free synthesis aligns with green chemistry principles, significantly lowering the volume of hazardous waste generated per unit of product. This environmental advantage facilitates easier regulatory compliance and reduces the carbon footprint of the manufacturing operation. Commercial scale-up of complex fluorinated intermediates becomes more feasible as the safety risks associated with high-temperature exotherms are mitigated by the controlled nature of the catalytic system.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this fluorination technology, based on the specific data and embodiments provided in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of transferring this process to pilot or commercial scales. The answers reflect the empirical findings regarding catalyst stability, substrate scope, and reaction conditions observed during the development of this method.
Q: What are the thermal stability advantages of phosphonium catalysts over traditional ammonium salts?
A: Unlike conventional quaternary ammonium salts which decompose at high Halex temperatures (200-300°C), the phosphonium catalysts described in CN1413177A exhibit superior thermal stability, showing minimal decomposition (e.g., only 0.2% carbonaceous products) even after 10 hours at 230°C.
Q: Can this process be operated without solvents to reduce waste?
A: Yes, the patent explicitly demonstrates that the reaction can be successfully carried out in the absence of solvents. This solvent-free capability significantly simplifies downstream processing and reduces the environmental burden associated with solvent recovery and disposal.
Q: Does this method work for non-activated aromatic substrates?
A: While electron-withdrawing groups enhance reactivity, the method is effective for a broad range of substrates including non-activated chloropyridines and chlorobenzenes, provided the reaction temperature is adjusted appropriately within the 40-260°C range.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Aromatics Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical and specialty chemical markets. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the phosphonium-catalyzed Halex reaction are successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorinated intermediate meets the exacting standards required for API synthesis and electronic material applications. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced fluorination technology can be integrated into your supply chain to drive efficiency and quality. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits applicable to your target molecules. Our experts are ready to provide specific COA data and route feasibility assessments to support your next project milestone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
