Advanced In-Situ Synthesis of Diisopinocampheylchloroborane for High-Purity Chiral Alcohols
Advanced In-Situ Synthesis of Diisopinocampheylchloroborane for High-Purity Chiral Alcohols
The landscape of asymmetric synthesis in the pharmaceutical industry is constantly evolving, driven by the need for safer, more cost-effective, and scalable processes. A pivotal advancement in this domain is detailed in patent CN1051087C, which discloses an improved process for the in-situ preparation of diisopinocampheylchloroborane. This chiral reducing agent is critical for transforming prochiral ketones into their corresponding alcohols with exceptionally high optical purity. Unlike legacy methods that rely on hazardous reagents and complex isolation steps, this innovation leverages the reaction of sodium borohydride and boron trichloride with alpha-pinene. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the manufacturing of key pharmaceutical intermediates, particularly those required for leukotriene antagonists and other high-value therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of diisopinocampheylchloroborane has been fraught with significant operational challenges and safety hazards. Traditional protocols, such as those described in earlier literature, often necessitate the use of corrosive hydrogen chloride gas or the preparation of unstable monochloroborane etherates. These reagents are not only dangerous to handle on a large scale but also require specialized equipment resistant to severe corrosion. Furthermore, previous methods, including those cited in United States Patent 5,043,479, often required the crystallization and isolation of intermediate species like diisopinocampheylchloroborane base borane. These intermediates are notoriously sensitive to oxygen and moisture, making their separation and purification complex, costly, and prone to yield losses. The reliance on expensive borane reagents and the need for rigorous exclusion of air and water further compounded the economic burden, rendering these conventional routes less attractive for industrial-scale applications.
The Novel Approach
The methodology outlined in CN1051087C offers a transformative solution by enabling the generation of the active reducing agent directly within the reaction vessel, effectively bypassing the need for isolation. By reacting sodium borohydride and boron trichloride with alpha-pinene in an inert organic solvent, the process generates the diisopinocampheylchloroborane in situ. This approach eliminates the handling of corrosive hydrogen chloride gas and avoids the precarious isolation of oxygen-sensitive intermediates. The result is a streamlined workflow that is inherently safer and more robust. Moreover, this method demonstrates remarkable tolerance regarding the optical purity of the starting alpha-pinene; even pinene with an enantiomeric excess of approximately 70% can yield final alcohol products with optical purities around 94%. This flexibility allows manufacturers to utilize more cost-effective starting materials without sacrificing the stringent quality standards required for pharmaceutical intermediates.
Mechanistic Insights into In-Situ Chiral Borane Generation
The core of this technological breakthrough lies in the efficient generation of the chiral borane species. The reaction initiates with the interaction between sodium borohydride and boron trichloride in the presence of alpha-pinene, typically within a polyether solvent such as diglyme or triglyme. The stoichiometry is carefully controlled, with preferred mole ratios of sodium borohydride to boron trichloride around 1:1.1, and alpha-pinene present in excess to ensure complete complexation. The reaction temperature is meticulously managed, starting below 0°C during the addition of boron trichloride to control exothermicity, and subsequently warming to facilitate the formation of the active chloroborane complex. This in-situ generation ensures that the reactive species is immediately available for the reduction step, minimizing decomposition pathways that often plague isolated borane reagents.
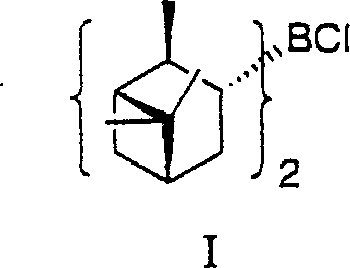
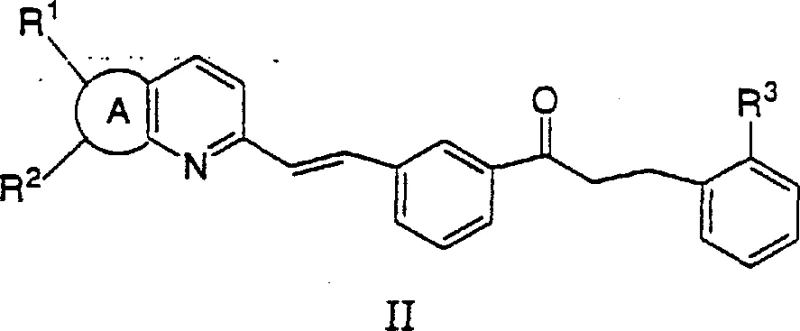
Once formed, the diisopinocampheylchloroborane acts as a potent stereoselective reducing agent. The bulky chiral environment created by the isopinocampheyl groups directs the hydride transfer to the prochiral ketone substrate with high fidelity. As illustrated in the patent data, this system is particularly effective for reducing ketones with structures akin to Formula II, converting them into chiral alcohols of Formula III. The mechanism relies on the rigid coordination of the boron center with the chiral ligands, which creates a specific spatial arrangement that favors the formation of one enantiomer over the other. This precise control is essential for producing pharmaceutical intermediates where even minor deviations in enantiomeric excess can render a batch useless. The ability to achieve enantiomeric excess values exceeding 99% in the final product, as demonstrated in the examples, underscores the efficacy of this catalytic system in maintaining stereochemical integrity throughout the transformation.
How to Synthesize Diisopinocampheylchloroborane Efficiently
Implementing this synthesis route requires careful attention to solvent selection and atmospheric conditions to maximize yield and safety. The process is preferably conducted under an inert atmosphere, such as nitrogen, to prevent oxidation of the sensitive borane species. Solvents like 1,2-dimethoxyethane (glyme) are favored for the initial generation of the reagent, while tetrahydrofuran (THF) is often preferred for the subsequent reduction step due to its excellent solubility properties for diverse ketone substrates. The reaction timeline is relatively short, with the formation of the reducing agent completing within approximately 30 minutes to 2 hours, followed by the reduction of the ketone which may proceed over several hours depending on the specific substrate and temperature profile.
- Combine sodium borohydride and alpha-pinene in an inert polyether solvent like glyme under nitrogen atmosphere.
- Slowly add boron trichloride solution while maintaining low temperatures to generate the active chloroborane species in situ.
- Introduce the prochiral ketone substrate to the reaction mixture to effect asymmetric reduction to the corresponding chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this in-situ methodology presents compelling economic and logistical benefits. The primary advantage stems from the drastic simplification of the process flow. By eliminating the crystallization and isolation steps associated with traditional methods, manufacturers can significantly reduce solvent consumption, energy usage, and processing time. This reduction in unit operations translates directly into lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry principles. Furthermore, the avoidance of corrosive hydrogen chloride gas reduces the maintenance costs associated with reactor vessels and piping, extending the lifespan of capital equipment and minimizing downtime for repairs.
- Cost Reduction in Manufacturing: The substitution of expensive, specialized borane reagents with commodity chemicals like sodium borohydride and boron trichloride offers a substantial decrease in raw material costs. Additionally, the ability to use alpha-pinene with lower optical purity without compromising the final product quality allows for the sourcing of more affordable starting materials. The elimination of intermediate isolation steps further reduces labor and utility costs, creating a leaner and more cost-efficient production model for high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, specifically sodium borohydride and alpha-pinene, are widely available commodities with stable global supply chains. Unlike specialized chiral catalysts that may have limited suppliers and long lead times, these materials can be sourced from multiple vendors, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent output and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The in-situ nature of this reaction makes it highly amenable to scale-up. Without the need for complex purification of sensitive intermediates, the process can be easily transferred from laboratory to pilot and eventually to commercial production scales. The simplified workup procedure, which involves quenching with benign reagents like benzaldehyde and aqueous potassium carbonate, generates waste streams that are easier to treat and dispose of compared to those containing heavy metals or highly corrosive acids. This facilitates compliance with increasingly stringent environmental regulations and reduces the overall cost of waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: How does this method improve safety compared to traditional borane preparations?
A: Traditional methods often require handling corrosive hydrogen chloride gas or unstable monochloroborane etherates. This patented process utilizes sodium borohydride and boron trichloride, eliminating the need for gaseous HCl and avoiding the isolation of highly oxygen-sensitive intermediates, thereby significantly enhancing operational safety.
Q: Can lower purity alpha-pinene be used without compromising product quality?
A: Yes, the process demonstrates remarkable robustness. Even when using alpha-pinene with an enantiomeric excess as low as 70%, the resulting chiral alcohols can achieve optical purities around 94%, reducing the dependency on expensive, ultra-high purity starting materials.
Q: What are the scalability advantages of this in-situ technique?
A: By avoiding the crystallization and isolation of the intermediate diisopinocampheylchloroborane base borane, the process removes complex purification steps that are difficult to scale. This leads to a more continuous workflow, reduced solvent consumption, and faster throughput for large-scale API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diisopinocampheylchloroborane Supplier
The synthesis of complex chiral intermediates like those derived from diisopinocampheylchloroborane requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this field, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling sensitive organometallic reactions under strict inert atmospheres, ensuring that the stringent purity specifications required for pharmaceutical applications are consistently met. With our rigorous QC labs and commitment to process optimization, we provide a secure and reliable supply chain for critical API intermediates.
We invite R&D and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. Our technical team is ready to evaluate the feasibility of adapting this in-situ reduction technology to your existing workflows, potentially unlocking significant efficiency gains. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with high-quality, cost-effective chiral building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
