Advanced Synthesis of Ferrocenyl Coumarin Intermediates for Commercial Antioxidant Applications
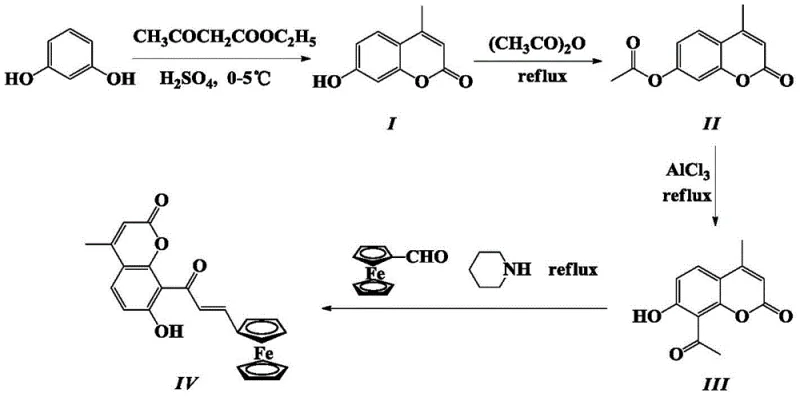
The pharmaceutical and fine chemical industries are constantly seeking novel molecular scaffolds that offer enhanced biological activity alongside manufacturability. Patent CN113512074A introduces a significant breakthrough in this domain with the disclosure of (E)-4-methyl-7-hydroxy-8-(3-(ferrocenyl)acryloyl)coumarin. This unique compound integrates three distinct pharmacophores: the coumarin backbone, the chalcone alpha-beta unsaturated ketone system, and the redox-active ferrocenyl group. The synthesis described in the patent represents a robust four-step pathway starting from the commodity chemical resorcinol. By strategically combining Knoevenagel condensation, esterification, Fries rearrangement, and a final aldol condensation, the inventors have created a molecule with superior antioxidant properties compared to its individual structural components. For R&D directors and procurement specialists, this patent offers a tangible route to high-purity ferrocenyl coumarin that avoids the pitfalls of traditional strong-base catalysis, ensuring better yield stability and product integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chalcone derivatives often relies on strong alkaline catalysts such as sodium hydroxide, potassium hydroxide, or sodium methoxide. While effective for simple ketone-aldehyde condensations, these harsh conditions pose a severe threat to the structural stability of coumarin derivatives. The lactone ring within the coumarin framework is highly susceptible to hydrolytic ring-opening when exposed to strong bases, leading to significant degradation of the starting material and a drastic reduction in overall yield. Furthermore, ferrocenyl compounds are known to be sensitive to various catalytic environments, often decomposing under aggressive reaction conditions. This sensitivity limits the choice of catalysts and reaction parameters, creating a bottleneck in the efficient production of ferrocenyl-coumarin hybrids. Consequently, many conventional routes fail to deliver the target molecule with the necessary purity and yield required for commercial scale-up of complex organic intermediates.
The Novel Approach
The methodology outlined in patent CN113512074A circumvents these traditional limitations through a carefully designed multi-step sequence that protects sensitive functional groups until the final stage. Instead of subjecting the coumarin core to harsh bases during the introduction of the ferrocenyl moiety, the process utilizes a mild amine-catalyzed aldol condensation. By first establishing the coumarin skeleton and installing the acetyl group via a Fries rearrangement, the synthesis ensures that the reactive sites are correctly positioned before the final coupling. This approach not only preserves the integrity of the lactone ring but also accommodates the sensitivity of the ferrocene unit. The result is a streamlined process that achieves high yields across all four steps, demonstrating a clear path for cost reduction in pharmaceutical intermediates manufacturing by minimizing waste and purification burdens associated with side reactions.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Aldol Condensation
The core of this synthesis lies in the precise manipulation of reaction mechanisms to achieve regioselectivity and high conversion. The initial step involves a Pechmann-like condensation where resorcinol reacts with ethyl acetoacetate in concentrated sulfuric acid. This acid-catalyzed process facilitates the formation of the coumarin ring system through dehydration and cyclization, yielding 4-methyl-7-hydroxycoumarin. Following this, the 7-hydroxyl group is protected via esterification with acetic anhydride. This protection is crucial for the subsequent Fries rearrangement, where anhydrous aluminum trichloride acts as a Lewis acid to migrate the acetyl group from the oxygen to the 8-position of the aromatic ring. This rearrangement is thermally driven, typically requiring temperatures between 140°C and 180°C, and is followed by acid hydrolysis to restore the phenolic hydroxyl group. The final step is the aldol condensation between the 8-acetyl coumarin and ferrocene carboxaldehyde. Here, the use of organic amines like piperidine or triethylamine as catalysts ensures a mild environment that promotes the formation of the alpha-beta unsaturated ketone linkage without compromising the ferrocene or coumarin structures.
Impurity control is inherently built into this synthetic design through the use of recrystallization at each stage. The patent specifies recrystallization from 95% ethanol for every intermediate and the final product, which serves as a powerful purification tool to remove unreacted starting materials and side products. For instance, the Fries rearrangement can produce isomeric byproducts, but the specific solubility differences in ethanol allow for the isolation of the desired 8-acetyl isomer. Additionally, the choice of solvent in the final condensation step, such as ethanol or isopropanol, aids in driving the equilibrium towards the product while keeping impurities in solution. This rigorous attention to purification ensures that the final (E)-4-methyl-7-hydroxy-8-(3-(ferrocenyl)acryloyl)coumarin meets stringent purity specifications, a critical factor for its application as a high-purity ferrocenyl coumarin in sensitive biological assays or pharmaceutical formulations.
How to Synthesize (E)-4-methyl-7-hydroxy-8-(3-(ferrocenyl)acryloyl)coumarin Efficiently
The synthesis of this advanced antioxidant intermediate requires precise control over reaction temperatures and stoichiometry to maximize efficiency. The process begins with the careful addition of resorcinol and ethyl acetoacetate into cooled concentrated sulfuric acid, maintaining the temperature below 10°C to prevent polymerization or degradation. Following the initial cyclization, the intermediate is subjected to reflux with acetic anhydride, a standard exothermic reaction that requires controlled heating to ensure complete esterification. The subsequent Fries rearrangement is the most energy-intensive step, necessitating an oil bath at 140-180°C under anhydrous conditions to facilitate the migration of the acyl group. Finally, the aldol condensation is performed under reflux in an alcoholic solvent with a catalytic amount of amine. Detailed standardized synthesis steps see the guide below.
- Perform Knoevenagel condensation of resorcinol with ethyl acetoacetate in concentrated sulfuric acid at low temperature to form 4-methyl-7-hydroxycoumarin.
- Execute 7-hydroxy esterification using acetic anhydride under reflux to protect the hydroxyl group and prepare for rearrangement.
- Conduct Fries rearrangement with anhydrous aluminum trichloride at elevated temperatures to introduce the acetyl group at the 8-position.
- Finalize with aldol condensation using ferrocene carboxaldehyde and an amine catalyst to yield the target ferrocenyl coumarin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers significant advantages due to its reliance on readily available commodity chemicals and standard processing equipment. The primary raw material, resorcinol, is a bulk chemical with a stable global supply, reducing the risk of raw material shortages that often plague specialty synthesis. Furthermore, the reaction conditions do not require exotic high-pressure equipment or cryogenic temperatures beyond standard ice-bath cooling, making the process highly adaptable to existing manufacturing infrastructure. This adaptability translates directly into reduced lead time for high-purity antioxidants, as production lines can be configured quickly without extensive retooling. The robustness of the four-step sequence ensures consistent output, which is vital for maintaining supply continuity for downstream pharmaceutical or agrochemical applications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the final coupling step significantly lowers the raw material cost profile. Traditional cross-coupling reactions often require palladium or other precious metals, which not only increase direct material costs but also necessitate expensive removal steps to meet residual metal specifications. By utilizing simple amine catalysts and aluminum chloride, this process avoids these costs entirely. Additionally, the high yields reported in the patent examples, ranging from 72% to 97% across individual steps, minimize the loss of valuable intermediates. This efficiency reduces the overall cost of goods sold, allowing for more competitive pricing in the market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The use of common solvents such as ethanol, acetic anhydride, and sulfuric acid ensures that the supply chain is not vulnerable to niche chemical shortages. These reagents are produced at massive scales globally, ensuring consistent availability and price stability. Moreover, the solid nature of the intermediates and the final product facilitates easy storage and transportation, reducing logistical complexities associated with liquid or unstable compounds. This stability enhances the reliability of the supply chain, ensuring that procurement managers can secure long-term contracts with confidence, knowing that the production process is resilient to minor fluctuations in reagent quality or availability.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from the laboratory to the pilot plant and finally to commercial production. The waste streams generated are primarily acidic or organic in nature, which can be managed through standard wastewater treatment protocols common in the fine chemical industry. The absence of heavy metals simplifies the environmental compliance burden, reducing the costs associated with hazardous waste disposal. This environmental friendliness aligns with modern green chemistry principles, making the manufacturing process more sustainable and acceptable to regulatory bodies, thereby smoothing the path for commercial approval and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this ferrocenyl coumarin derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility of the synthesis and the potential value proposition for industrial partners looking to integrate this molecule into their product pipelines. Understanding these details is crucial for making informed decisions about process adoption and investment.
Q: Why is this ferrocenyl coumarin synthesis superior to traditional chalcone methods?
A: Traditional methods often use strong bases like NaOH or KOH which can hydrolyze the sensitive lactone ring in coumarins. This patent utilizes a mild amine-catalyzed aldol condensation in the final step, preserving the structural integrity of the coumarin core while successfully integrating the ferrocenyl moiety.
Q: What are the primary industrial applications of this compound?
A: The compound demonstrates exceptional free radical scavenging activity against ABTS, DPPH, and galvinoxyl radicals, outperforming standard ferrocenyl chalcones. This makes it a high-value candidate for developing advanced antioxidant pharmaceutical intermediates and specialty fine chemicals.
Q: Is the synthetic route scalable for commercial production?
A: Yes, the process relies on commodity raw materials like resorcinol and standard unit operations such as reflux, filtration, and recrystallization. The absence of exotic catalysts and the use of robust reaction conditions facilitate straightforward commercial scale-up of complex organic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-4-methyl-7-hydroxy-8-(3-(ferrocenyl)acryloyl)coumarin Supplier
The technical potential of this ferrocenyl coumarin synthesis is immense, offering a pathway to high-performance antioxidants with superior stability and activity. NINGBO INNO PHARMCHEM stands ready to support the commercialization of this technology through our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the exacting standards required by global pharmaceutical and fine chemical clients. We understand the complexities of organometallic synthesis and are uniquely positioned to handle the specific nuances of ferrocene chemistry, ensuring a seamless transition from patent to production.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that will validate the economic and technical viability of this intermediate for your applications. Let us help you optimize your supply chain with a reliable partner dedicated to quality and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
