Scalable Chemical Synthesis of Calixanthomycin A Glycosyl Ligand for Oncology Drug Development
Introduction to Breakthrough Synthesis Technology
The pharmaceutical industry has long sought reliable access to complex polycyclic xanthone natural products due to their profound biological activity, particularly in oncology. Patent CN108558899B discloses a pioneering method for the chemical synthesis of the Calixanthomycin A glycosyl ligand, a critical precursor to the potent anticancer agent Calixanthomycin A. This natural product exhibits exceptional inhibitory activity against human colon cancer cells with an IC50 value as low as 0.43nM, yet its complex six-ring structure and high oxidation state have historically precluded total chemical synthesis. The disclosed technology represents the first successful chemical realization of this glycosyl ligand, offering a transformative pathway for drug development teams seeking to bypass the limitations of natural extraction. By establishing a robust synthetic route, this innovation enables the production of high-purity intermediates essential for generating Calixanthomycin A through subsequent simple glycosidation reactions.
This technological breakthrough addresses a significant gap in the availability of advanced pharmaceutical intermediates. Previously, reliance on fermentation processes using Streptomyces albus introduced variability and supply constraints that hindered clinical progression. The new methodology employs a modular synthetic strategy, constructing the complex core through convergent assembly of simpler aromatic building blocks. This approach not only secures the supply chain for this valuable therapeutic candidate but also opens avenues for creating structural analogs with improved pharmacokinetic profiles. For R&D directors and procurement specialists, this patent signifies a shift from unpredictable biological sourcing to precise, controllable chemical manufacturing, ensuring consistent quality and availability for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of Calixanthomycin A and its derivatives relied exclusively on isolation from natural sources, specifically the fermentation broth of Streptomyces albus. This biological production method suffers from inherent inefficiencies, including low titers, complex downstream purification requirements, and significant batch-to-batch variability influenced by microbial health and culture conditions. Furthermore, the structural complexity of the molecule, characterized by a highly oxidized polycyclic xanthone scaffold, made previous attempts at total chemical synthesis futile. The lack of a synthetic route meant that any modification to the core structure to enhance potency or reduce toxicity was nearly impossible, locking researchers into the properties of the naturally occurring variant. Supply chain managers faced constant risks of shortage, as scaling fermentation is capital-intensive and time-consuming, often failing to meet the demands of expanding clinical trials.
The Novel Approach
The novel approach detailed in the patent circumvents these biological bottlenecks by introducing a fully synthetic pathway that constructs the glycosyl ligand from commercially available starting materials. Instead of attempting a linear synthesis of the entire molecule at once, the inventors utilized a convergent strategy that assembles the tetracyclic core through key carbon-carbon bond-forming reactions. A standout feature of this method is the use of intramolecular radical cyclization to forge the challenging ring systems, a technique that offers superior regioselectivity compared to traditional ionic cyclizations. Additionally, the route incorporates a late-stage glycosylation strategy, where the synthesized ligand serves as a versatile platform. This modularity allows for the efficient generation of the final active pharmaceutical ingredient through a single, high-yielding glycosidation step, drastically simplifying the overall manufacturing process and reducing the number of purification stages required.
Mechanistic Insights into Radical Cyclization and Coupling
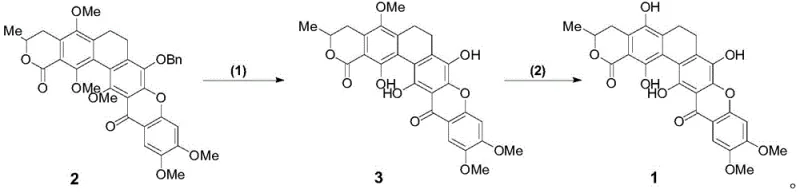
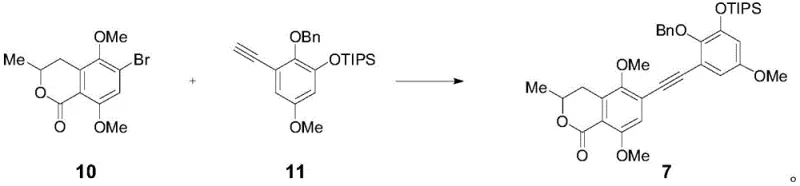
The core of this synthetic achievement lies in the sophisticated application of transition metal catalysis and photochemical radical processes to build molecular complexity. A pivotal step involves the Sonogashira coupling reaction between intermediate 10 and intermediate 11, facilitated by a palladium catalyst and cuprous iodide. This reaction efficiently links the two major aromatic fragments, establishing the carbon backbone necessary for the subsequent ring closures. The choice of catalyst system, typically bis-triphenylphosphine palladium dichloride, ensures high turnover and minimizes homocoupling byproducts, which is critical for maintaining high purity in such a complex molecule. Following the coupling, the resulting alkyne undergoes selective hydrogenation to set the stage for the crucial cyclization event, demonstrating precise control over oxidation states throughout the sequence.
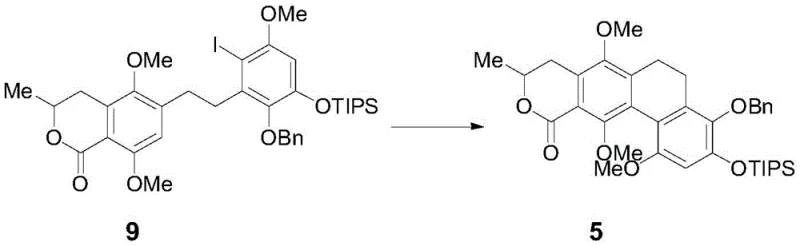
The most mechanistically distinct operation is the intramolecular radical cyclization of compound 9 to form compound 5, achieved under ultraviolet irradiation in the presence of an organic base. This photochemical transformation generates a radical species that attacks the adjacent aromatic ring, closing the central ring of the xanthone system with high fidelity. The use of UV light allows for the activation of the carbon-iodine bond without the need for harsh thermal conditions that might degrade sensitive functional groups. Impurity control is further enhanced in the final deprotection stages, where Lewis acids like boron trichloride are employed to selectively remove methyl and benzyl protecting groups. This chemoselectivity is vital, as it preserves the integrity of the lactone and ketone functionalities while exposing the necessary phenolic hydroxyl groups for the final glycosylation, ensuring the final product matches the natural structure exactly.
How to Synthesize Calixanthomycin A Glycosyl Ligand Efficiently
The synthesis of this high-value intermediate requires careful attention to reaction conditions, particularly during the photochemical cyclization and the final deprotection steps. The process begins with the preparation of key building blocks, followed by their convergence and structural elaboration. Operators must ensure strict exclusion of oxygen during the palladium-catalyzed steps to prevent catalyst deactivation and maintain high yields. The detailed standardized synthetic procedures, including specific molar ratios, solvent choices like hexafluoroisopropanol for Friedel-Crafts acylation, and temperature controls for the radical cyclization, are critical for reproducibility. For a comprehensive guide on executing these transformations in a GMP environment, please refer to the standardized protocol below.
- Prepare key intermediates 10 and 11 through multi-step functionalization including allylation, formylation, and Sonogashira coupling.
- Perform intramolecular radical cyclization on intermediate 9 under UV irradiation to construct the tetracyclic core structure 5.
- Execute final deprotection steps using Lewis acids and hypervalent iodine reagents to yield the target Calixanthomycin A glycosyl ligand (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from fermentation to chemical synthesis offers substantial strategic benefits beyond mere availability. The ability to produce the Calixanthomycin A glycosyl ligand synthetically decouples the supply of this critical oncology intermediate from the biological variabilities of microbial fermentation. This shift ensures a consistent, year-round production capability that can be scaled according to market demand without the long lead times associated with strain development and bioreactor optimization. Furthermore, the synthetic route utilizes commodity chemicals and standard reactor equipment, which significantly lowers the barrier to entry for manufacturing partners and reduces the overall cost of goods sold. By eliminating the need for complex extraction and purification from biomass, the process streamlines the supply chain, reducing logistics costs and environmental footprint associated with large-scale fermentation waste.
- Cost Reduction in Manufacturing: The synthetic route eliminates the expensive infrastructure required for sterile fermentation and downstream biological processing. By utilizing robust chemical transformations like Sonogashira coupling and radical cyclization, the process achieves high atom economy and reduces the number of unit operations. The use of recoverable palladium catalysts and common organic solvents further drives down operational expenditures. This efficiency translates into a more cost-effective supply of the intermediate, allowing pharmaceutical companies to allocate resources towards clinical development rather than raw material acquisition. The avoidance of low-yield natural extraction steps ensures that the final cost per gram is significantly optimized compared to traditional isolation methods.
- Enhanced Supply Chain Reliability: Chemical synthesis provides a predictable and controllable production schedule, immune to the biological contaminants or strain degradation issues that plague fermentation processes. The modular nature of the synthesis allows for the stocking of key intermediates, creating a buffer against supply disruptions. This reliability is crucial for maintaining continuous clinical trial supplies and meeting regulatory commitments. Suppliers can guarantee delivery timelines with greater confidence, as the production rate is determined by reactor capacity and chemical throughput rather than microbial growth rates. This stability mitigates the risk of project delays caused by raw material shortages, ensuring a steady flow of materials for drug product manufacturing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reactions that are well-understood in industrial organic chemistry. The absence of exotic reagents and the use of standard workup procedures facilitate easy scale-up from pilot plant to commercial production volumes. From an environmental perspective, the synthetic route generates less biological waste and avoids the disposal challenges associated with large volumes of fermentation broth. The implementation of efficient purification techniques, such as column chromatography and crystallization, minimizes solvent consumption and waste generation. This alignment with green chemistry principles supports corporate sustainability goals and simplifies regulatory compliance regarding environmental discharge and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the Calixanthomycin A glycosyl ligand. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your drug development pipeline. The responses cover aspects of purity, scalability, and the specific chemical advantages of this novel synthetic route.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: This chemical synthesis provides a reliable, scalable alternative to fermentation, eliminating batch-to-batch variability and supply chain dependencies associated with Streptomyces albus culture.
Q: How is the complex polycyclic structure constructed efficiently?
A: The route utilizes a strategic intramolecular radical cyclization under UV irradiation to close the rings, combined with robust Sonogashira coupling to join key aromatic fragments.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses standard industrial reagents and equipment, avoiding exotic catalysts, which facilitates commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calixanthomycin A Glycosyl Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for next-generation oncology therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle complex synthetic routes involving sensitive photochemical reactions and transition metal catalysis, adhering to stringent purity specifications required for clinical-grade materials. With rigorous QC labs and a commitment to process optimization, we guarantee that every batch of Calixanthomycin A glycosyl ligand meets the highest standards of identity and purity, enabling your team to focus on advancing therapeutic outcomes.
We invite you to collaborate with us to leverage this breakthrough synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep expertise in process chemistry and supply chain management. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the journey of this promising anticancer candidate from the lab to the clinic.
