Scalable Synthesis of High-Efficiency Bipolar Blue OLED Host Materials for Next-Gen Displays
Scalable Synthesis of High-Efficiency Bipolar Blue OLED Host Materials for Next-Gen Displays
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit superior electroluminescent efficiency but also possess the thermal and morphological stability required for commercial manufacturing. Patent CN103450191A introduces a groundbreaking organic semiconductor material designed specifically to address the bottlenecks in blue phosphorescent device performance. This novel compound, chemically defined as 3,6-bis-pyrenes-9,9-bis-(4-(two-p-methylphenyl) aminophenyl)-1,8-carboline, represents a sophisticated integration of three distinct functional moieties: triphenylamine, carboline, and pyrene. By strategically combining these units, the material achieves a rare bipolar character, facilitating both hole injection and electron transport while maintaining a high triplet energy level essential for hosting blue phosphorescent dopants. For R&D directors and procurement specialists in the display sector, understanding the synthetic accessibility and structural advantages of this molecule is critical for securing a reliable supply chain for next-generation flat panel displays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional host materials for OLED applications often suffer from unbalanced charge transport properties, leading to efficiency roll-off at high brightness levels. Many conventional hosts are purely hole-transporting or electron-transporting, which necessitates complex device architectures with multiple additional layers to balance the carrier flux. Furthermore, standard carbazole-based hosts frequently lack sufficient thermal stability, resulting in crystallization or degradation during the high-temperature vacuum deposition processes used in mass production. This instability can significantly shorten the operational lifetime of the final display panel. Additionally, the synthesis of complex host molecules often involves harsh conditions or low-yielding steps that generate difficult-to-remove impurities, complicating the purification process and driving up the cost of goods sold for the final electronic chemical product.
The Novel Approach
The synthetic strategy outlined in the patent data offers a robust solution by constructing a rigid, sterically hindered molecular framework that inherently balances charge transport. The core of this approach involves the formation of a 9,9-disubstituted carboline scaffold, which serves as an excellent electron-transporting unit due to its nitrogen-containing heterocyclic structure. This core is flanked by triphenylamine derivatives, known for their exceptional hole-transporting capabilities, and terminated with pyrene groups that provide high fluorescence quantum yields and further enhance electron mobility. This molecular design effectively creates a bipolar host material that simplifies device architecture by eliminating the need for separate charge transport layers. The synthetic route utilizes efficient condensation and cross-coupling reactions that are amenable to scale-up, ensuring that the material can be produced in quantities sufficient for industrial application without compromising on purity or performance metrics.
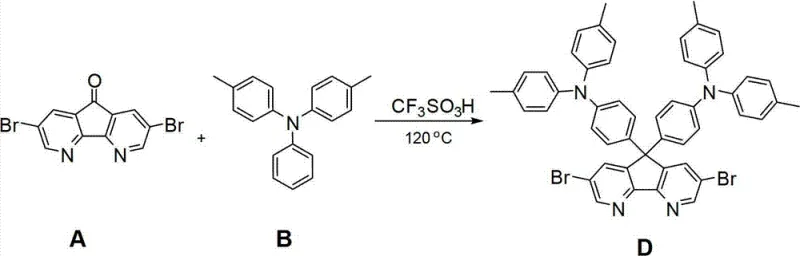
Mechanistic Insights into Acid-Catalyzed Condensation and Suzuki Coupling
The synthesis of the key intermediate, Compound D (9,9-bis-(4-(two-p-methylphenyl) aminophenyl)-3,6-bis-bromo-1,8-carboline), relies on a highly efficient acid-catalyzed condensation reaction. In this step, 3,6-dibromo-1,8-phenodiazine Fluorenone reacts with 4,4'-dimethyltrianiline in the presence of trifluoromethanesulfonic acid (CF3SO3H) at elevated temperatures ranging from 110°C to 120°C. The strong Brønsted acid catalyst activates the carbonyl group of the fluorenone derivative, facilitating a nucleophilic attack by the amine groups of the triphenylamine derivative. This is followed by a dehydration and cyclization sequence that constructs the rigid carboline core. The use of trifluoromethanesulfonic acid is particularly advantageous as it promotes the reaction to completion with high conversion rates, evidenced by the reported isolated yield of 91% for Compound D. This high yield at the intermediate stage is crucial for overall process economics, minimizing waste and reducing the burden on downstream purification steps.
The final assembly of the target OLED material involves a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction between the dibromo-intermediate Compound D and pyrene-1-boronic acid pinacol ester (Compound C). This transformation is critical for installing the pyrene functionalities which dictate the optical and electronic properties of the final product. The reaction mechanism proceeds through the standard catalytic cycle of oxidative addition, transmetallation, and reductive elimination. The patent explores various catalyst systems, including tetrakis(triphenylphosphine)palladium(0) and the more advanced Pd2dba3/S-phos system. The use of bulky, electron-rich phosphine ligands like S-phos stabilizes the monomeric palladium species, accelerating the oxidative addition of the sterically hindered aryl bromide bonds in Compound D. This mechanistic optimization allows the reaction to proceed efficiently even with the bulky pyrene boronate coupling partner, achieving yields up to 78% under optimized conditions using potassium carbonate in tetrahydrofuran.
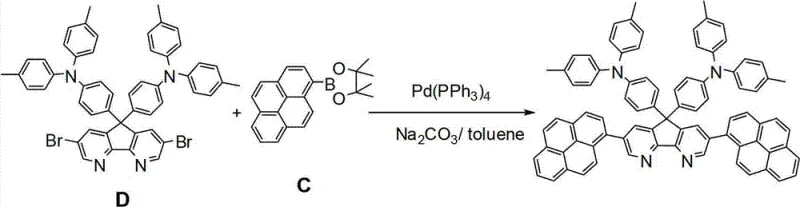
How to Synthesize 3,6-bis-pyrenes-9,9-bis-(4-(two-p-methylphenyl) aminophenyl)-1,8-carboline Efficiently
The preparation of this high-performance organic semiconductor requires strict control over reaction parameters to ensure the high purity necessary for electronic applications. The process begins with the rigorous drying of all solvents and reagents to prevent catalyst deactivation during the palladium-coupling step. The initial condensation to form the carboline core must be monitored closely to prevent over-reaction or polymerization, utilizing trifluoromethanesulfonic acid as a potent promoter. Following the isolation of the dibromo-intermediate, the subsequent Suzuki coupling demands an inert atmosphere to protect the sensitive palladium catalyst from oxidation. Detailed standardized synthesis steps for this specific pathway are provided in the guide below to assist process chemists in replicating these results.
- Synthesize the core intermediate Compound D by reacting 3,6-dibromo-1,8-phenodiazine Fluorenone with 4,4'-dimethyltrianiline using trifluoromethanesulfonic acid catalyst at 120°C.
- Perform Suzuki-Miyaura cross-coupling between Compound D and pyrene pinacol borate using a palladium catalyst system such as Pd(PPh3)4 or Pd2dba3/S-phos.
- Purify the crude product via silica gel column chromatography using methylene dichloride and petroleum ether to obtain the final oyster-white solid semiconductor material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from laboratory synthesis to commercial manufacturing is often fraught with challenges regarding cost, scalability, and raw material availability. The synthetic route described in this patent offers several distinct logistical and economic advantages that mitigate these risks. Firstly, the starting materials, including substituted triphenylamines and pyrene boronates, are commercially available commodity chemicals or can be synthesized via established industrial processes, ensuring a stable supply chain foundation. The high yields reported in the experimental examples, particularly the 91% yield for the core intermediate, suggest a process with minimal material loss, which directly translates to lower raw material consumption per kilogram of final product. This efficiency is a key driver for cost reduction in electronic chemical manufacturing, allowing for competitive pricing in the volatile display materials market.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for exotic or prohibitively expensive reagents, relying instead on standard palladium catalysts and common inorganic bases like sodium carbonate or potassium carbonate. The ability to use relatively inexpensive solvents such as toluene or tetrahydrofuran further drives down operational expenditures. Moreover, the high thermal stability of the final product, indicated by a 5% weight loss temperature of 469°C, implies that the material can withstand rigorous purification processes like sublimation without degradation, ensuring high purity without significant yield loss during finishing. This robustness reduces the overall cost of quality control and waste disposal associated with failed batches.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis, where the core intermediate can be stockpiled and then coupled with various boronic acids if needed, provides flexibility in production planning. The use of robust catalyst systems like Pd2dba3/S-phos, which are less sensitive to oxygen and moisture compared to traditional tetrakis catalysts, reduces the risk of batch failures due to environmental fluctuations in the manufacturing plant. This reliability ensures consistent delivery schedules for downstream OLED panel manufacturers, who operate on tight production timelines. The scalability of the reflux conditions (10 to 30 hours) fits well within standard batch reactor cycles, facilitating a smooth transition from pilot plant to multi-ton production scales.
- Scalability and Environmental Compliance: The purification methods described, involving standard extraction and silica gel column chromatography, are well-understood unit operations that can be easily adapted for large-scale processing. While column chromatography is often a bottleneck at scale, the high crude purity suggested by the yields indicates that simpler recrystallization techniques might be viable for bulk production, significantly reducing solvent usage and waste generation. The absence of heavy metal contaminants in the final structure, combined with the ability to remove palladium residues through standard scavenging techniques, ensures compliance with stringent environmental regulations governing electronic waste. This environmental compatibility is increasingly becoming a prerequisite for suppliers entering the supply chains of major global electronics conglomerates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this bipolar blue OLED host material. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and synthetic feasibility. Understanding these details is essential for technical teams evaluating this material for integration into their existing device architectures.
Q: What are the key structural advantages of this organic semiconductor material?
A: The material integrates triphenylamine for hole transport, carboline for electron transport, and pyrene for high fluorescence quantum yield, creating a bipolar host with balanced carrier mobility and high triplet energy suitable for blue phosphorescent devices.
Q: How does the thermal stability of this material compare to conventional hosts?
A: Thermogravimetric analysis indicates a 5% weight loss temperature of 469°C, demonstrating exceptional thermal stability that prevents degradation during the vacuum deposition process and extends device operational lifetime.
Q: Which palladium catalyst systems are most effective for the final coupling step?
A: The patent demonstrates that while tetrakis(triphenylphosphine)palladium(0) is effective, the use of Pd2dba3 combined with S-phos ligands in THF with potassium carbonate can achieve yields up to 78%, offering a robust alternative for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-bis-pyrenes-9,9-bis-(4-(two-p-methylphenyl) aminophenyl)-1,8-carboline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced OLED materials hinges on the seamless translation of patented chemistry into reliable industrial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale laboratory synthesis to metric-ton manufacturing is executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and electronic properties of every batch. Our commitment to quality assurance guarantees that the bipolar host materials we supply meet the exacting standards required for high-resolution, long-lifetime electroluminescent devices.
We invite potential partners to engage with our technical procurement team to discuss how this specific synthetic route can be optimized for your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our manufacturing efficiencies can lower your total cost of ownership. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a secure and efficient supply of high-performance organic semiconductors for your next-generation display applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
