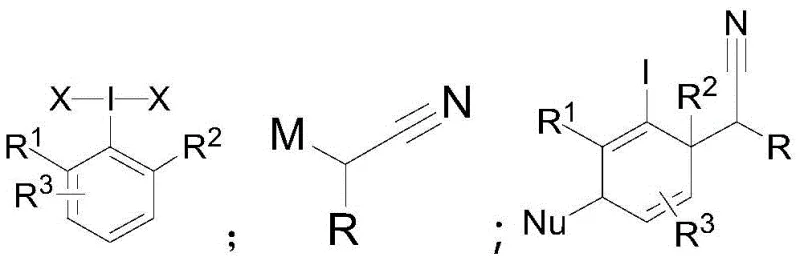
Advanced Synthesis of Polysubstituted Alicyclic Compounds for Commercial Scale-up
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient and selective methods to construct complex molecular architectures. Patent CN110526832B introduces a groundbreaking methodology for the preparation of polysubstituted alicyclic compounds, leveraging the unique reactivity of aryl hypervalent iodine compounds. This technology represents a significant leap forward in dearomatization chemistry, transforming readily available aromatic precursors into highly functionalized cyclohexadiene derivatives. For research and development teams focused on drug discovery, the ability to rapidly access these dense structural motifs is invaluable. The process described involves the activation of aryl hypervalent iodine species followed by a rearrangement with alpha-tin or silicon-substituted nitriles at cryogenic temperatures ranging from -70°C to -100°C. This specific temperature control is crucial for stabilizing the highly electrophilic dearomatization intermediates before they are trapped by various nucleophiles. The result is a versatile platform for generating diverse chemical libraries that serve as critical building blocks in the synthesis of bioactive molecules and advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the dearomatization of benzene rings has been a formidable challenge in synthetic organic chemistry, often requiring harsh conditions that limit substrate scope and functional group compatibility. Classical approaches such as Birch reduction necessitate the use of liquid ammonia and alkali metals at extremely low temperatures, posing significant safety risks and operational complexities on a large scale. Furthermore, phenol oxidation methods often suffer from over-oxidation issues or require stoichiometric amounts of toxic heavy metal oxidants, creating substantial waste disposal burdens for manufacturing facilities. These conventional pathways typically allow for the introduction of only a single functional group during the dearomatization event, necessitating multiple additional steps to achieve the desired substitution pattern. Such multi-step sequences inevitably lead to reduced overall yields and increased production costs, which are critical pain points for procurement managers aiming to optimize supply chain efficiency. The lack of stereoselectivity in many traditional methods also complicates the isolation of pure isomers, often requiring expensive chromatographic separations that are not feasible for ton-scale production.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes hypervalent iodine chemistry to achieve dual functionalization of the arene ring in a single operational sequence. By employing activated aryl iodine species, the method generates a highly reactive intermediate that undergoes a rapid [3,3]-sigma rearrangement with alpha-metalated nitriles. This cascade process effectively breaks the aromaticity of the starting material while simultaneously installing both an iodine atom and a nitrile-containing side chain. The reaction conditions are remarkably mild, typically conducted in dichloromethane at -78°C, which is easily achievable with standard industrial cooling systems. This shift from extreme reducing conditions to controlled electrophilic activation dramatically expands the range of compatible functional groups, allowing for the preservation of sensitive moieties that would otherwise be degraded. The inherent modularity of this system means that by simply varying the nucleophile added in the final step, manufacturers can access a wide array of distinct polysubstituted products from a common intermediate, thereby streamlining inventory management and reducing the need for multiple dedicated synthesis lines.
Mechanistic Insights into Hypervalent Iodine Mediated Dearomatization
The core of this technological advancement lies in the precise generation and capture of a transient electrophilic species. Upon activation by a Lewis acid such as trimethylsilyl trifluoromethanesulfonate (TMSOTf), the aryl hypervalent iodine compound transforms into a potent electrophile, often an iodonium species with enhanced leaving group ability. This activated complex is then attacked by the alpha-tin substituted nitrile, initiating a concerted rearrangement process. The synergy between the triflate anion and the tin center facilitates the cleavage of the carbon-tin bond, driving the formation of the rearrangement precursor. This step is critical as it sets the stage for the subsequent sigma-bond migration that disrupts the aromatic pi-system. The resulting intermediate possesses a cyclohexadienyl cation character, making it exceptionally susceptible to nucleophilic attack. Understanding this mechanistic pathway is essential for R&D directors who need to predict impurity profiles and optimize reaction parameters for robustness.
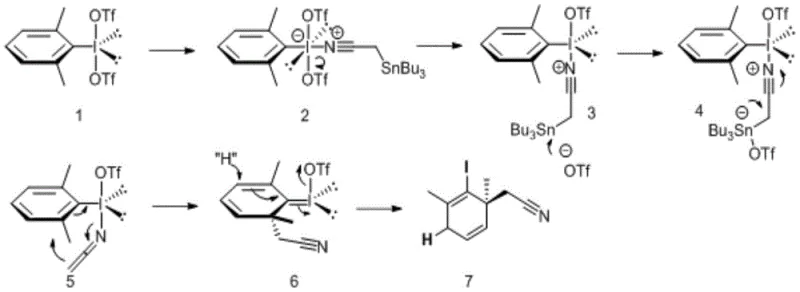
Controlling the stereochemical outcome and minimizing side reactions requires a deep understanding of the intermediate's lifetime and reactivity. The patent highlights that the dearomatized species is highly unstable and must be captured in situ at low temperatures to prevent decomposition or polymerization. The choice of nucleophile, whether it be a hydride source like triethylsilane or a carbon nucleophile like an allyl silane, dictates the final substitution pattern at the ring position. The high regioselectivity observed in these reactions is attributed to the steric and electronic environment created by the initial iodine placement and the substituents on the aromatic ring. For quality control teams, this predictability is a major advantage, as it ensures consistent product specifications across different batches. Furthermore, the presence of the iodine atom in the final product provides a valuable handle for downstream diversification via palladium-catalyzed cross-coupling reactions, adding another layer of value to the synthetic utility of these intermediates.
How to Synthesize Polysubstituted Alicyclic Compounds Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent addition rates to manage the exothermic nature of the activation step. The process begins with the dissolution of the aryl hypervalent iodine precursor in dry dichloromethane under an inert atmosphere to prevent moisture interference. Activation is achieved by adding the silyl triflate reagent at -78°C, followed by a brief stirring period to ensure complete conversion to the active species. Subsequently, the alpha-tin nitrile component is introduced, triggering the rearrangement cascade that forms the key dearomatized intermediate. Finally, the chosen nucleophile is added to quench the reaction and lock in the desired structural features. Detailed standardized synthesis steps see the guide below.
- Activate the aryl hypervalent iodine compound with TMSOTf in DCM at -78°C for 10 minutes.
- Add the alpha-tin substituted nitrile compound at -78°C and stir for 10 minutes to form the rearrangement precursor.
- Introduce the nucleophile (e.g., triethylsilane) at -78°C and stir for 12 hours to capture the dearomatized intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling benefits that directly address the cost and reliability concerns of modern chemical manufacturing. The reliance on hypervalent iodine reagents, which are increasingly available from bulk chemical suppliers, reduces dependency on scarce or geopolitically sensitive materials. The mild reaction conditions translate to lower energy consumption compared to processes requiring cryogenic liquids like ammonia or high-temperature reflux, contributing to a smaller carbon footprint and reduced utility costs. Moreover, the high selectivity of the reaction minimizes the formation of byproducts, which simplifies the purification workflow and reduces the volume of solvent and silica gel required for chromatography. These factors combined lead to a more streamlined production process that enhances overall throughput and reliability of supply for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the primary dearomatization step significantly lowers the raw material cost per kilogram of product. Additionally, the high atom economy of the rearrangement process ensures that a larger proportion of the starting mass ends up in the final product, reducing waste disposal fees. The ability to perform multiple functionalizations in a single pot operation cuts down on labor hours and equipment usage time, further driving down the cost of goods sold. By avoiding the need for specialized high-pressure or ultra-low temperature equipment, capital expenditure for new production lines is also substantially minimized.
- Enhanced Supply Chain Reliability: The starting materials for this process, including substituted phenols and iodine sources, are commodity chemicals with stable global supply chains. This reduces the risk of production stoppages due to raw material shortages, a critical factor for supply chain heads managing long-term contracts. The robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their production base to mitigate regional risks. Furthermore, the stability of the intermediates allows for potential telescoping of steps, which reduces the need for intermediate storage and handling, thereby speeding up the overall lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent is well-established in the industry, with mature recovery and recycling protocols that ensure compliance with environmental regulations. The absence of heavy metal waste streams simplifies the effluent treatment process, making it easier to meet stringent discharge standards. The reaction's scalability has been demonstrated through various examples in the patent, showing consistent yields from milligram to gram scales, which suggests a smooth path to commercial scale-up of complex polymer additives or drug substances. This environmental and operational efficiency positions the technology as a sustainable choice for green chemistry initiatives within the organization.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this dearomatization technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances helps in evaluating the feasibility of adopting this method for specific project requirements.
Q: What are the advantages of this hypervalent iodine method over traditional Birch reduction?
A: This method operates under significantly milder conditions (-78°C vs liquid ammonia temperatures) and avoids the safety hazards associated with dissolving metal reductions, while offering superior functional group tolerance.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available reagents and standard low-temperature techniques, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates without requiring exotic equipment.
Q: What is the stereochemical outcome of the dearomatization reaction?
A: The reaction proceeds through a highly organized [3,3]-sigma rearrangement transition state, often yielding specific diastereomers with high selectivity, which simplifies downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Alicyclic Compounds Supplier
As the demand for complex chiral building blocks continues to rise in the pharmaceutical sector, having a partner with deep technical expertise is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and stringent purity specifications guarantee that every batch of polysubstituted alicyclic compounds meets the highest industry standards, facilitating smoother regulatory filings for your end products. We understand the critical nature of timeline adherence in drug development and are committed to providing reliable support throughout the lifecycle of your project.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this methodology might impact your overall budget and production timeline. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the performance of these intermediates in your downstream processes. Let us collaborate to drive efficiency and innovation in your chemical supply chain.
