Advanced Organocatalytic Synthesis of Chiral Octahydroacridine Skeletons for Pharmaceutical Applications
Advanced Organocatalytic Synthesis of Chiral Octahydroacridine Skeletons for Pharmaceutical Applications
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN108774170B introduces a groundbreaking methodology for constructing an octahydroacridine skeleton featuring three continuous chiral centers, a structural motif prevalent in gastric acid secretion inhibitors and antimalarial agents. This innovation leverages a silane-modified 4-hydroxyproline catalyst to drive an asymmetric aza-Diels-Alder reaction, achieving exceptional enantioselectivity and yield under remarkably mild conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in accessing complex chiral building blocks without the burden of expensive transition metal catalysts or harsh reaction environments.
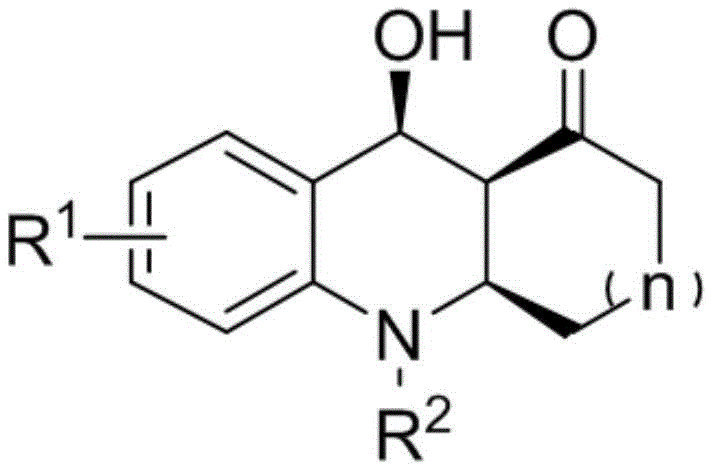
The core value of this invention lies in its ability to generate a highly functionalized scaffold where three newly formed bonds on the same ring are oriented consistently, facilitating further derivatization into more complex drug candidates. Unlike traditional approaches that often struggle with stereocontrol, this organocatalytic system ensures the production of stable, optically active molecules with high purity. The versatility of the substrate scope allows for the incorporation of various halogen and alkyl substituents, making it an adaptable platform for the cost reduction in API manufacturing where diverse analog libraries are required for structure-activity relationship studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octahydroacridine skeletons has relied heavily on methods such as acid-catalyzed condensation of isophorone and aniline, Beckmann rearrangement of oxime sulfonates, or catalytic hydrogenation of acridine derivatives. While these pathways can construct the basic carbon framework, they fundamentally suffer from a critical deficiency: they predominantly yield racemic mixtures lacking optical activity. To obtain the desired enantiomer, manufacturers are forced to employ kinetic resolution or chiral separation techniques, which inherently cap the maximum theoretical yield at 50% and introduce significant processing costs and waste. Furthermore, some existing asymmetric methods, such as those involving asymmetric hydrogen transfer, often entail complex multi-step sequences, low overall yields, and limited substrate universality, creating bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108774170B utilizes a biomimetic organocatalytic strategy that bypasses the need for racemic synthesis and subsequent resolution. By employing a silane-modified 4-hydroxyproline derivative, the reaction proceeds through a highly organized transition state that dictates the stereochemistry of three contiguous chiral centers in a single operation. This direct asymmetric synthesis not only simplifies the process flow by eliminating resolution steps but also dramatically improves atom economy and overall throughput. The reaction operates at 0°C in a benign solvent system of pyridine and toluene, avoiding the extreme temperatures or pressures often associated with traditional heterocycle synthesis, thereby enhancing operational safety and equipment longevity in a production setting.
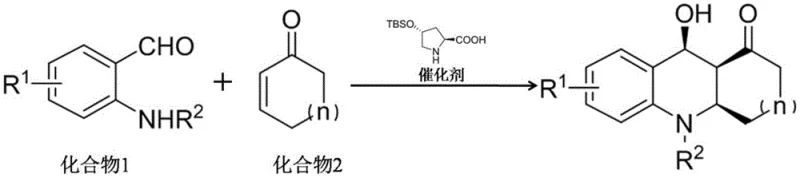
Mechanistic Insights into Silane-Modified Proline Catalyzed Aza-Diels-Alder Reaction
The success of this transformation is rooted in the sophisticated interplay between the organocatalyst and the substrates, which orchestrates the formation of the tetrahydroquinoline core with precise stereocontrol. The mechanism initiates with the activation of the cyclic enone (Compound 2) by the secondary amine of the catalyst, forming a reactive iminium ion intermediate (Int-I). Simultaneously, the substituted anthranilaldehyde (Compound 1) undergoes deprotonation and isomerization to generate an aza-diene species. The catalyst plays a dual role here, not only activating the electrophile but also stabilizing the nucleophilic aza-diene through hydrogen bonding interactions, leading to a highly ordered transition state (TS). This supramolecular assembly ensures that the subsequent [4+2] cycloaddition occurs with high facial selectivity, locking in the configuration of the new stereocenters before the catalyst is released to regenerate the cycle.
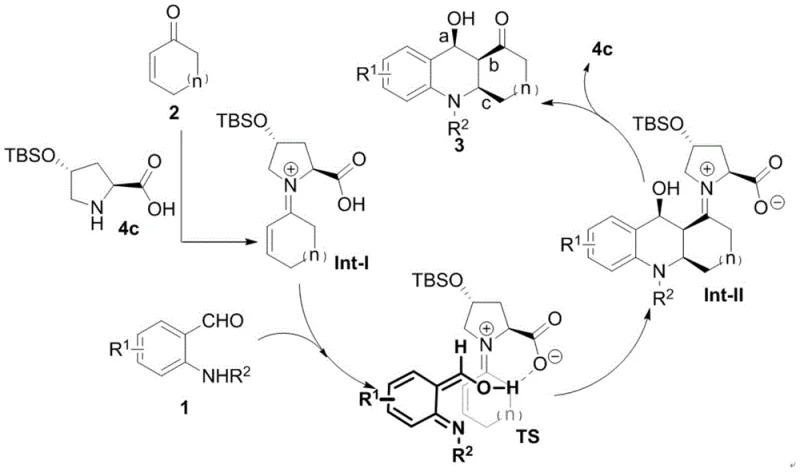
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates that often requires costly scavenging steps to meet regulatory limits. Furthermore, the mild reaction conditions minimize side reactions such as polymerization or decomposition of sensitive functional groups, resulting in a cleaner crude reaction profile. The high enantioselectivity observed, reaching up to 99% ee in optimized examples, indicates that the competing background reaction is effectively suppressed, ensuring that the final product stream is dominated by the desired enantiomer. This level of purity reduces the burden on downstream purification units, directly contributing to process efficiency and cost effectiveness.
How to Synthesize Octahydroacridine Derivatives Efficiently
The practical implementation of this synthesis is straightforward and robust, designed to be easily translated from laboratory discovery to pilot plant operations. The process begins by dissolving the appropriate N-substituted anthranilaldehyde in a specific ratio of pyridine and toluene, followed by the addition of the chiral organocatalyst. Once the mixture is homogenized, the cyclic enone is introduced, and the reaction vessel is immediately placed in a constant-temperature ice bath to maintain the critical 0°C setpoint. After the reaction reaches completion, typically monitored by TLC within 10 to 48 hours depending on the substrate, the product is isolated as a white solid following standard chromatographic purification.
- Dissolve the substituted anthranilaldehyde (Compound 1) in a pyridine and toluene mixed solvent system.
- Add the silane-modified 4-hydroxyproline catalyst and the cyclic enone (Compound 2) to the reaction mixture.
- Maintain the reaction at 0°C in an ice bath for 10-48 hours, then purify the resulting white solid via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers compelling strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; the starting anthranilaldehydes and cyclic enones are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Moreover, the elimination of precious metal catalysts removes a significant cost driver and volatility factor associated with commodities like palladium or rhodium. The mild operating conditions also imply lower energy consumption for heating or cooling compared to high-temperature reflux or cryogenic processes, contributing to a smaller carbon footprint and reduced utility costs in large-scale manufacturing.
- Cost Reduction in Manufacturing: The most significant economic impact comes from the high atom economy and the avoidance of resolution steps. Traditional racemic synthesis followed by chiral separation inherently wastes at least half of the material produced, whereas this asymmetric method directs the synthesis towards the desired isomer with yields reaching up to 99%. Additionally, the removal of transition metals means there is no need for expensive metal scavengers or specialized filtration equipment to meet ppm-level residue specifications. The simplified workup procedure, often requiring only standard column chromatography or crystallization, reduces solvent usage and labor hours, leading to substantial cost savings in the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply continuity by minimizing the risk of batch failures due to sensitive parameters. Since the process does not require strict anhydrous or anaerobic environments, it is less susceptible to variations in raw material moisture content or minor leaks in reactor seals, which are common causes of downtime in fine chemical production. The broad substrate tolerance means that if a specific substituted aldehyde faces supply constraints, the process can often accommodate alternative analogs with minimal re-optimization, providing flexibility in sourcing strategies. This resilience ensures a steady flow of high-purity intermediates to downstream API manufacturing sites.
- Scalability and Environmental Compliance: Scaling this process is facilitated by the use of common organic solvents like toluene and pyridine, which are well-understood in terms of handling and waste treatment. The absence of heavy metals simplifies wastewater treatment protocols, as there is no need for complex heavy metal precipitation or recovery systems, aligning with increasingly stringent environmental regulations. The exothermic nature of the cycloaddition is manageable at 0°C, allowing for safe scale-up in standard jacketed reactors without the need for specialized cryogenic infrastructure. This ease of scale-up supports the rapid transition from kilogram-scale clinical supply to multi-ton commercial production, ensuring that market demand can be met without prolonged lead times.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this octahydroacridine synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this organocatalytic method over traditional acid-catalyzed condensation?
A: Traditional methods often produce racemic mixtures requiring difficult resolution. This novel organocatalytic approach directly yields optically active products with up to 99% ee and three continuous chiral centers, significantly simplifying downstream processing.
Q: Is the reaction condition harsh or sensitive to air and moisture?
A: No, the process utilizes mild reaction conditions (0°C) in a standard organic solvent mixture. It does not require harsh anhydrous or anaerobic environments, making it highly suitable for scalable industrial manufacturing.
Q: What is the substrate scope for R1 and R2 groups?
A: The method demonstrates strong universality, accommodating various substituents such as H, F, Cl, Br, I, Me, and OMe at the R1 position, and diverse sulfonyl groups like Ts, PhSO2, and Ms at the R2 position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octahydroacridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the silane-modified proline catalytic system described in CN108774170B and is fully prepared to support your project from gram-scale optimization to full commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yield observed in the lab are maintained at the plant level. Our rigorous QC labs and stringent purity specifications guarantee that every batch of octahydroacridine skeleton meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your project needs, obtain specific COA data for our reference standards, and receive comprehensive route feasibility assessments that will accelerate your time to market. Let us handle the complexities of chiral synthesis so you can focus on delivering life-saving medicines to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
