Advanced Cage Arene CA-1: Revolutionizing Benzene Separation and Commercial Scale-Up
Advanced Cage Arene CA-1: Revolutionizing Benzene Separation and Commercial Scale-Up
The petrochemical industry has long grappled with the formidable challenge of separating benzene from cyclohexane, a critical purification step given their nearly identical physical properties. Patent CN114181056A introduces a groundbreaking solution through the synthesis and application of a novel cage arene, designated as CA-1 (C105H96O12). This sophisticated macrocyclic compound offers a paradigm shift from energy-intensive distillation to efficient adsorption-based separation. By leveraging specific host-guest interactions, CA-1 enables the programmed release of high-purity hydrocarbons, addressing the limitations of traditional azeotropic distillation. As a leading reliable cage arene supplier, we recognize this technology as a pivotal advancement for reducing operational expenditures in aromatic processing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of benzene and cyclohexane has been plagued by thermodynamic constraints that render standard physical separation methods economically burdensome. The boiling points of these two compounds differ by a mere 0.6K, and they tend to form azeotropes, which makes conventional fractional distillation exceptionally difficult and energy-prohibitive. Existing industrial solutions, such as extractive distillation or azeotropic distillation, require massive energy inputs and complex equipment setups to break these azeotropes. Furthermore, processes involving mechanical vapor recompression, while improving efficiency slightly, still incur substantial operating costs due to the continuous demand for thermal energy. These legacy methods not only strain facility budgets but also increase the carbon footprint of chemical manufacturing, conflicting with modern sustainability goals.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a non-porous adaptive crystalline material that operates on the principles of molecular recognition rather than volatility differences. The cage arene CA-1 functions as a solid-state adsorbent that selectively captures benzene and cyclohexane based on subtle differences in their electronic properties and molecular geometry. This method eliminates the need for high-energy rectification towers, replacing them with a low-temperature adsorption and thermal desorption cycle. The process is operationally simple, requiring only controlled heating to trigger the programmed release of the captured species. This transition from thermal separation to adsorptive separation represents a significant leap forward in cost reduction in specialty chemical manufacturing, offering a cleaner and more manageable workflow for plant operators.
Mechanistic Insights into AlCl3-Catalyzed Coupling and Acid-Mediated Cyclization
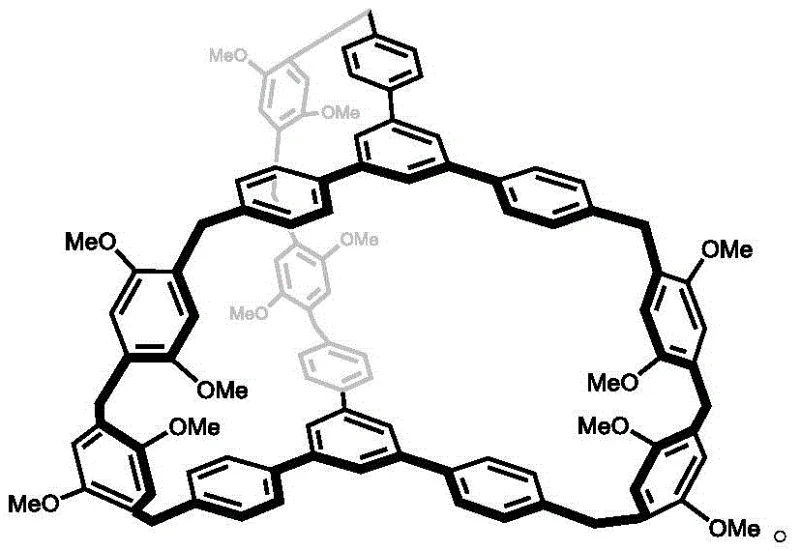
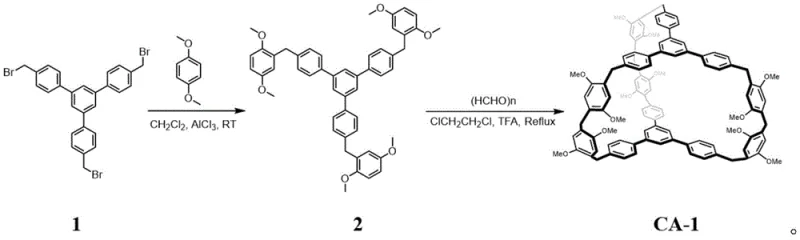
The synthesis of CA-1 is a testament to precise organic construction, utilizing a modular two-step strategy that ensures high structural fidelity. The first stage involves a Friedel-Crafts alkylation where Compound 1 reacts with p-dimethoxybenzene in the presence of aluminum trichloride (AlCl3) in dichloromethane at room temperature. This Lewis acid-catalyzed reaction efficiently constructs the linear precursor, Compound 2, by forming robust carbon-carbon bonds between the aromatic rings. The second, and perhaps more critical step, is the macrocyclization. Here, Compound 2 undergoes condensation with paraformaldehyde in 1,2-dichloroethane, catalyzed by trifluoroacetic acid (TFA) under reflux conditions. This acid-mediated cyclization closes the large ring structure, creating the internal cavity essential for host-guest chemistry. The rigorous control of reaction conditions, particularly the anhydrous environment and specific acid strength, is vital to preventing polymerization and ensuring the formation of the discrete cage structure.
Beyond synthesis, the mechanism of separation relies on distinct CH-pi and electrostatic interactions between the electron-rich cavity of CA-1 and the guest molecules. Benzene, being electron-rich, forms a host-guest complex with a stoichiometric ratio of roughly 1:3, but this interaction is relatively weak. Conversely, cyclohexane forms a more stable complex, theoretically at a 1:1 ratio, due to different interaction dynamics within the cage. This differential stability is the key to the "programmed release" capability. By heating the loaded material to 70°C, the weaker benzene complex dissociates, releasing pure benzene. Subsequent heating to over 130°C breaks the stronger cyclohexane bonds, releasing cyclohexane and regenerating the adsorbent. This precise thermal control ensures high-purity output without cross-contamination, a critical factor for downstream pharmaceutical or polymer applications.
How to Synthesize Cage Arene CA-1 Efficiently
The preparation of this advanced adsorbent requires strict adherence to the patented protocol to ensure the formation of the correct crystalline phase capable of adaptive inclusion. The process begins with the careful handling of moisture-sensitive reagents like aluminum trichloride to prevent catalyst deactivation during the initial coupling phase. Following the isolation of the intermediate, the macrocyclization step demands precise temperature control during reflux to drive the equilibrium toward the cyclic product rather than oligomers. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the standardized protocol below.
- Dissolve compound 1 and p-dimethoxybenzene in dichloromethane, add aluminum trichloride, and react at room temperature to obtain compound 2.
- Dissolve compound 2 and paraformaldehyde in 1,2-dichloroethane, add trifluoroacetic acid, and reflux to obtain Cage Arene CA-1.
- Activate the resulting solid material by recrystallization and heating above 130°C to remove solvent molecules prior to adsorption applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of CA-1 technology offers compelling economic and logistical benefits that extend beyond simple material substitution. The primary advantage lies in the drastic simplification of the separation infrastructure. By moving away from complex distillation columns and compressors, facilities can significantly reduce capital expenditure (CAPEX) on heavy machinery and the associated maintenance overheads. The operational simplicity of the adsorption process means that fewer specialized operators are needed, and the risk of process upsets common in high-pressure distillation is minimized. This reliability translates directly into more consistent production schedules and reduced downtime, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The elimination of energy-intensive distillation steps results in substantial utility savings. Since the separation relies on moderate heating for desorption rather than continuous boiling and condensation, the thermal load on the plant is drastically lowered. Additionally, the reagents used in the synthesis of CA-1, such as aluminum trichloride and trifluoroacetic acid, are commodity chemicals with established supply chains, avoiding the cost volatility associated with exotic noble metal catalysts. The ability to recycle the adsorbent material multiple times without loss of performance further amortizes the material cost over a longer lifecycle, driving down the cost per kilogram of separated product.
- Enhanced Supply Chain Reliability: The robustness of the CA-1 material contributes to a more stable supply of high-purity intermediates. Traditional separation methods are often bottlenecks; if a distillation column fouls or fails, the entire line stops. In contrast, the adsorption beds using CA-1 can be managed in parallel, allowing for continuous operation even during regeneration cycles. The raw materials for synthesizing the cage arene are readily available in the global chemical market, reducing the risk of supply disruptions. This availability ensures that manufacturers can scale production to meet demand fluctuations without being constrained by scarce or single-source reagents.
- Scalability and Environmental Compliance: From an environmental perspective, this technology aligns perfectly with green chemistry principles. The process generates less waste compared to extractive distillation, which often requires large volumes of solvent that must be recovered or disposed of. The solid-state nature of the adsorbent minimizes liquid effluent streams, simplifying wastewater treatment requirements. Furthermore, the scalability of the synthesis is proven, moving from gram-scale laboratory examples to potential tonnage production. The ability to operate at atmospheric pressure and moderate temperatures also reduces safety risks, lowering insurance premiums and regulatory compliance burdens associated with high-pressure chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Cage Arene CA-1 in industrial separation processes. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: Why is conventional distillation inefficient for separating benzene and cyclohexane?
A: Conventional distillation is highly energy-intensive because benzene and cyclohexane form an azeotrope and have boiling points differing by only 0.6K, making separation via standard rectification extremely difficult and costly.
Q: How does Cage Arene CA-1 achieve selective separation?
A: CA-1 utilizes host-guest chemistry where benzene forms a weaker complex (1:3 ratio) released at 70°C, while cyclohexane forms a stronger complex released only above 130°C, allowing for programmed thermal release.
Q: Is the Cage Arene CA-1 adsorbent recyclable?
A: Yes, the material demonstrates high thermal stability up to 130°C and maintains its crystal structure and selectivity after multiple adsorption-desorption cycles without significant degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cage Arene CA-1 Supplier
At NINGBO INNO PHARMCHEM, we understand that translating a promising patent into a commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless. We are committed to delivering high-purity Cage Arene CA-1 that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or bulk supply for your separation units, our infrastructure is designed to support your growth.
We invite you to collaborate with us to optimize your separation workflows. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific facility constraints. By partnering with us, you gain access to specific COA data and route feasibility assessments that will validate the economic viability of switching to this advanced adsorption technology. Contact us today to discuss how we can support your supply chain with reliable, high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
