Scalable Chiral Induction Strategy for Netadoline Dimesylate Manufacturing and Commercial Supply
The pharmaceutical landscape for ROCK kinase and norepinephrine transporter inhibitors is undergoing a significant transformation driven by the need for more efficient chiral synthesis methodologies. Patent CN113121436A introduces a groundbreaking preparation method for Netadoline Dimesylate that fundamentally shifts the paradigm from expensive chiral separation to cost-effective chiral induction. This technical breakthrough addresses the critical bottleneck of obtaining high-optical-purity intermediates without relying on preparative chiral chromatography, a process often deemed prohibitive for large-scale commercial manufacturing. By utilizing a specifically designed chiral ligand, the invention enables the direct construction of the stereocenter during the bond-forming steps, thereby streamlining the entire production workflow. This approach not only enhances the overall yield but also significantly reduces the environmental footprint associated with excessive solvent usage in separation processes. For global procurement teams, this represents a pivotal opportunity to secure a more stable and economically viable supply chain for this high-value therapeutic intermediate.
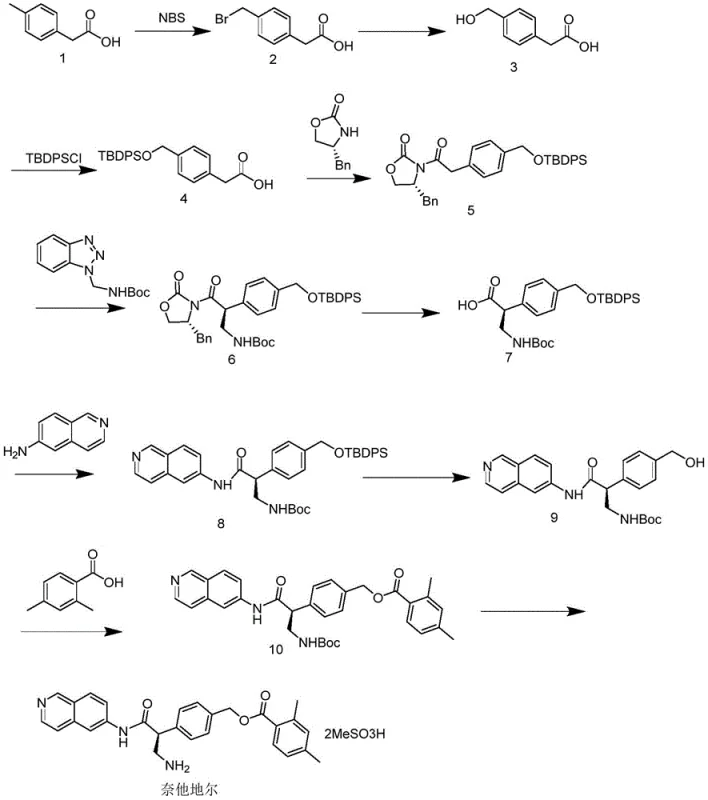
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral pharmaceutical intermediates like Netadoline has heavily relied on resolution techniques, particularly chiral column chromatography, to isolate the desired enantiomer from a racemic mixture. This conventional approach presents severe limitations when transitioning from laboratory benchtop to industrial scale, primarily due to the exorbitant costs associated with chiral stationary phases and the massive volumes of mobile phase solvents required. The throughput of chiral columns is inherently low, creating a significant bottleneck that restricts production capacity and leads to extended lead times for bulk orders. Furthermore, the recovery of solvents and the disposal of waste streams from these separation processes add layers of complexity to environmental compliance and operational expenditure. The low overall yield inherent in resolution methods, where theoretically half of the material is discarded or requires recycling, further exacerbates the cost burden, making the final API intermediate less competitive in the global market.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages asymmetric synthesis using a chiral oxazolidinone auxiliary to induce stereochemistry directly during the carbon-carbon bond formation. This method eliminates the need for post-reaction separation of enantiomers, as the chirality is established with high fidelity at the molecular level before the final structure is assembled. The use of a robust chiral ligand, synthesized from readily available precursors, allows for precise control over the stereochemical outcome, consistently achieving optical purities exceeding 99% as demonstrated in the experimental examples. By integrating the chiral information into the synthesis backbone, the process simplifies the purification steps to standard crystallization and extraction, which are far more scalable and cost-efficient than chromatography. This strategic shift not only improves the material throughput but also enhances the reproducibility of the manufacturing process, ensuring batch-to-batch consistency that is critical for regulatory approval and commercial success.
Mechanistic Insights into Chiral Induction via Oxazolidinone Auxiliary
The core of this technological advancement lies in the sophisticated application of chiral induction mechanisms utilizing a specific oxazolidinone derivative, identified as Formula 5 in the patent documentation. The synthesis begins with the protection of 4-(hydroxymethyl)phenylacetic acid using tert-butyldiphenylchlorosilane (TBDPSCl), creating a robust intermediate that withstands subsequent harsh reaction conditions. This protected acid is then activated to an acyl chloride and coupled with the chiral auxiliary under cryogenic conditions using n-butyllithium to form the imide. The steric bulk of the auxiliary group effectively shields one face of the enolate intermediate formed upon deprotonation with lithium hexamethyldisilazide, forcing the incoming electrophile to attack from the less hindered face. This facial selectivity is the key determinant of the high enantiomeric excess observed in the final product, showcasing the power of rational catalyst and auxiliary design in modern organic synthesis.
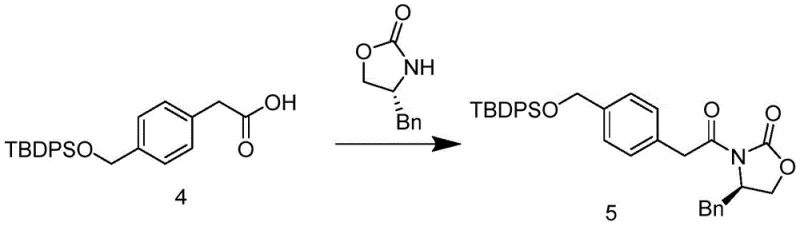
Following the establishment of the chiral center, the process involves a series of carefully orchestrated transformations to install the remaining functional groups while preserving the stereochemical integrity. The alkylation step with the triazole-containing intermediate is conducted at controlled low temperatures to prevent racemization, followed by the removal of the chiral auxiliary under mild oxidative conditions using hydrogen peroxide and lithium hydroxide. This cleavage step is crucial as it releases the chiral acid intermediate without compromising the newly formed stereocenter, allowing the auxiliary to be potentially recovered and recycled. Subsequent amidation with 6-aminoisoquinoline and deprotection of the silyl ether group proceed with high efficiency, culminating in the formation of the Netadoline base. The final salt formation with methanesulfonic acid ensures the stability and crystallinity of the product, meeting the rigorous physical-chemical specifications required for pharmaceutical formulation and long-term storage.
How to Synthesize Netadoline Dimesylate Efficiently
The synthesis of Netadoline Dimesylate via this chiral induction route requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize yield and optical purity. The process begins with the preparation of the chiral ligand, followed by the sequential assembly of the molecular framework through protection, alkylation, and functional group interconversion. Each step is designed to minimize side reactions and facilitate easy purification, ensuring that the intermediate quality remains high throughout the sequence. For research and development teams looking to replicate or scale this process, adherence to the specific molar ratios and cooling protocols described in the patent is essential to achieve the reported 99% optical purity. The detailed standardized synthesis steps see the guide below.
- Protect 4-(hydroxymethyl)phenylacetic acid with TBDPSCl to form the silyl ether intermediate, followed by activation with oxalyl chloride.
- React the activated acid with a chiral oxazolidinone auxiliary using n-butyllithium at cryogenic temperatures to establish the chiral center.
- Perform alkylation with the triazole intermediate, followed by auxiliary removal, amidation with 6-aminoisoquinoline, and final salt formation with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this chiral induction methodology offers substantial advantages in terms of cost structure and supply chain resilience. By eliminating the dependency on chiral chromatography, manufacturers can drastically reduce the variable costs associated with solvent procurement, waste disposal, and column replacement, leading to a more competitive pricing model for the final intermediate. The simplified workflow also reduces the number of unit operations required, which translates to shorter manufacturing cycles and the ability to respond more agilely to fluctuating market demands. For supply chain heads, the robustness of this chemical route means fewer risks of batch failures due to column overloading or resolution inefficiencies, ensuring a continuous and reliable flow of materials to downstream API production facilities. This stability is paramount for maintaining uninterrupted drug supply chains in the highly regulated pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of preparative chiral HPLC removes one of the most expensive steps in fine chemical manufacturing, significantly lowering the cost of goods sold. The use of standard reagents and recyclable auxiliaries further optimizes the material cost profile, allowing for substantial savings that can be passed down the supply chain. This economic efficiency makes the production of Netadoline Dimesylate viable for broader therapeutic applications where cost sensitivity is a key factor in market access.
- Enhanced Supply Chain Reliability: The reliance on scalable unit operations like crystallization and filtration rather than low-throughput chromatography enhances the overall reliability of the supply chain. This process robustness minimizes the risk of production delays caused by equipment bottlenecks or consumable shortages, ensuring that delivery schedules are met consistently. Suppliers utilizing this technology can offer more secure long-term contracts, providing peace of mind to pharmaceutical partners planning multi-year clinical or commercial campaigns.
- Scalability and Environmental Compliance: The route is inherently designed for scale-up, utilizing common organic solvents and reagents that are readily available in bulk quantities for industrial production. The reduction in solvent waste and the potential for auxiliary recovery align with green chemistry principles, simplifying environmental compliance and reducing the carbon footprint of the manufacturing process. This sustainability advantage is increasingly important for multinational corporations aiming to meet their corporate social responsibility goals while sourcing critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Netadoline Dimesylate using the patented chiral induction method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing development pipelines.
Q: How does this patent improve upon traditional chiral separation methods?
A: Traditional methods rely on chiral column chromatography, which is costly and difficult to scale due to high solvent consumption and low throughput. This patent utilizes chiral induction with a reusable auxiliary, significantly reducing processing time and material costs while maintaining high optical purity.
Q: What is the expected optical purity of the final Netadoline Dimesylate?
A: According to the experimental data in Patent CN113121436A, the described chiral induction route achieves an optical purity of 99%, ensuring the compound meets stringent pharmaceutical specifications for ROCK kinase inhibition.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route avoids preparative HPLC, using standard crystallization and extraction techniques instead. The use of robust protecting groups like TBDPS and standard reagents like oxalyl chloride makes the process highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Netadoline Dimesylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial supply chains for our global partners. Our CDMO expertise allows us to adapt complex chiral induction routes like the one described in CN113121436A from bench scale to full commercial production with precision and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with the highest standards of quality and consistency. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of Netadoline Dimesylate meets the exacting requirements of the pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through advanced process chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our technical capabilities can support your long-term strategic goals in the development of ROCK kinase inhibitors.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
