Optimizing 6-Hydroxy-3,4-Dihydro-2(1H)-Quinolinone Production for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks more efficient pathways for producing critical heterocyclic intermediates, and the technology disclosed in patent CN108383781B represents a significant leap forward in the synthesis of 6-hydroxy-3,4-dihydro-2(1H)-quinolinone. This compound serves as a pivotal building block for Cilostazol and various other cardiovascular and therapeutic agents, making its production efficiency a matter of strategic importance for global supply chains. The patented method introduces a novel palladium-catalyzed approach that fundamentally restructures the synthetic logic, moving away from cumbersome multi-step sequences towards a more direct and atom-economical process. By leveraging the unique reactivity of p-alkoxyaniline derivatives under specific thermal and pressure conditions, this technology achieves a remarkable simplification of the manufacturing workflow. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating potential cost reductions and supply chain resilience in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 6-hydroxy-3,4-dihydro-2(1H)-quinolinone has relied on circuitous routes that impose significant burdens on both operational efficiency and environmental compliance. Traditional methodologies often commence with aniline, subjecting it to a rigorous sequence of acylation, cyclization, nitration, reduction, diazotization, and finally hydrolysis to install the requisite hydroxyl group. This six-step trajectory not only accumulates material losses at every stage but also necessitates the handling of hazardous reagents such as strong acids and reducing agents, leading to substantial waste generation. Furthermore, alternative routes utilizing p-hydroxy phenylamine with aluminum chloride catalysts have struggled with difficult cyclization kinetics and sluggish reaction rates, resulting in inconsistent yields and purification challenges. These legacy processes create bottlenecks in manufacturing capacity and inflate the cost of goods sold due to the extensive labor and utility requirements associated with each additional unit operation.
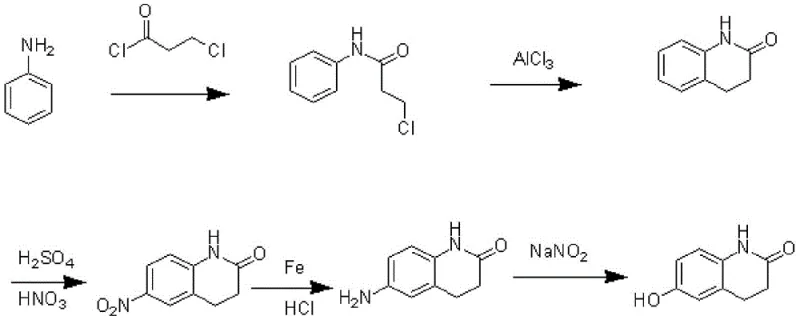
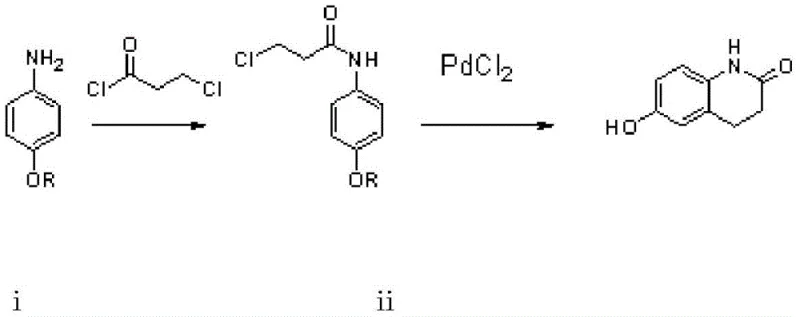
The Novel Approach
In stark contrast to these legacy methods, the innovative strategy outlined in the patent utilizes a streamlined two-stage reaction sequence that dramatically enhances process throughput and reliability. The core of this advancement lies in the use of p-alkoxyaniline as a starting material, which undergoes acylation to form an amide intermediate that is subsequently subjected to a palladium chloride-catalyzed transformation. This single catalytic step accomplishes both cyclization and dealkylation simultaneously, effectively collapsing what were previously multiple discrete operations into one potent chemical event. The reaction proceeds smoothly in tetrahydrofuran at temperatures between 100°C and 110°C under moderate pressure, conditions that are readily achievable in standard industrial reactors without requiring specialized cryogenic or ultra-high-pressure equipment. This consolidation of steps not only accelerates the production timeline but also inherently improves the overall mass balance, as evidenced by total yields reaching up to 85% in optimized examples, a figure that vastly outperforms the cumulative yields of traditional multi-step syntheses.
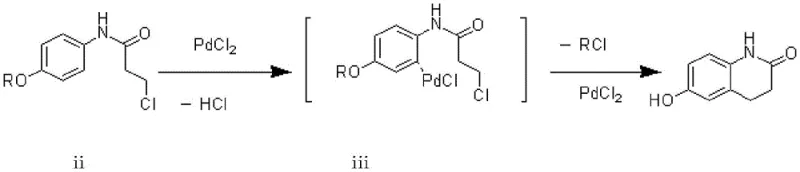
Mechanistic Insights into PdCl2-Catalyzed Cyclization and Dealkylation
From a mechanistic perspective, the efficacy of this synthesis relies on the precise coordination chemistry facilitated by the palladium chloride catalyst, which orchestrates the formation of the quinolinone core with high regioselectivity. The process begins with the interaction between the N-(4-alkoxyphenyl)-3-chloropropenamide intermediate and the palladium species, forming a transient organometallic complex that activates the aromatic ring towards intramolecular nucleophilic attack. This activation lowers the energy barrier for cyclization, allowing the reaction to proceed rapidly at elevated temperatures while simultaneously promoting the cleavage of the alkoxy group. The generation of HCl as a byproduct during the initial complexation further aids in the dealkylation process, creating a synergistic effect where the catalyst and the acidic environment work in concert to deliver the final hydroxyl-substituted product. Understanding this dual-function mechanism is crucial for process chemists, as it highlights the importance of maintaining strict control over reaction parameters such as temperature and pressure to ensure the catalyst remains active and selective throughout the transformation.
Furthermore, the impurity profile of the resulting product is significantly cleaner compared to routes involving nitration and reduction, primarily because the catalytic cycle avoids the formation of nitro-reduction byproducts and azo-coupling impurities that are notoriously difficult to remove. The specificity of the palladium-mediated C-N bond formation ensures that side reactions are minimized, leading to a crude product that requires less intensive purification efforts. Subsequent recrystallization from alcohols such as ethanol or methanol effectively removes any residual palladium species and unreacted starting materials, yielding a white solid with a sharp melting point range of 235.9-236.4°C. This high level of purity is paramount for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metals must be adhered to, thereby reducing the regulatory risk associated with the manufacturing process.
How to Synthesize 6-Hydroxy-3,4-Dihydro-2(1H)-Quinolinone Efficiently
Implementing this synthesis at a commercial scale requires careful attention to the stoichiometry and solvent selection defined in the patent examples to maximize yield and safety. The process initiates with the acylation of p-alkoxyaniline using 3-chloropropionyl chloride in a solvent like toluene or ethyl acetate, where the molar ratio of base to amine is carefully controlled to neutralize the generated acid without promoting hydrolysis. Following the isolation of the amide intermediate, the critical cyclization step is performed in tetrahydrofuran with palladium chloride, necessitating a sealed vessel capable of withstanding 3-5 kg of pressure to maintain solvent integrity at 110°C. Detailed standardized synthetic steps see the guide below.
- Acylation of p-alkoxyaniline with 3-chloropropionyl chloride in organic solvent to form N-(4-alkoxyphenyl)-3-chloropropenamide.
- Palladium chloride catalyzed cyclization and dealkylation at 100-110°C under pressure to form the crude quinolinone core.
- Recrystallization of the crude product using alcohols such as ethanol or methanol to achieve high purity white solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line through operational efficiencies. By eliminating the need for hazardous nitration and reduction steps, the facility requirements for production are simplified, reducing the capital expenditure associated with specialized corrosion-resistant equipment and waste treatment infrastructure. The reduction in the number of unit operations also translates to a shorter manufacturing cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations without compromising on quality standards. Additionally, the use of readily available starting materials like p-alkoxyaniline ensures a stable supply base, mitigating the risks associated with sourcing exotic or highly regulated precursors that often plague complex pharmaceutical syntheses.
- Cost Reduction in Manufacturing: The consolidation of multiple synthetic steps into a streamlined catalytic process significantly lowers the variable costs associated with labor, utilities, and solvent consumption. By avoiding the use of expensive reducing agents and the extensive workup procedures required to remove inorganic salts from nitration reactions, the overall cost of goods is substantially reduced. Furthermore, the high yield achieved in the cyclization step minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into saleable product, which directly enhances profit margins in a competitive market environment.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain by reducing the likelihood of batch failures caused by complex multi-step sensitivities. With fewer intermediate isolations and purifications, there are fewer opportunities for material loss or contamination, leading to more predictable production schedules and consistent delivery timelines. This reliability is critical for downstream API manufacturers who depend on a steady flow of high-quality intermediates to maintain their own production continuity and meet regulatory filing commitments without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common organic solvents and moderate reaction conditions that translate seamlessly from laboratory benchtop to multi-ton production scales. From an environmental perspective, the avoidance of nitro-compounds and heavy metal reducing agents simplifies effluent treatment, lowering the environmental footprint and compliance costs associated with waste disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly becoming a key factor in supplier selection criteria for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical manufacturing considerations. These insights are intended to clarify the operational advantages and technical feasibility for stakeholders evaluating this route for potential integration into their supply networks. Comprehensive answers are provided below to assist in decision-making processes.
Q: What are the primary advantages of the Pd-catalyzed route over traditional nitration methods?
A: The Pd-catalyzed route eliminates multiple steps including nitration, reduction, and diazotization, significantly reducing environmental pollution and improving overall yield from multi-step low yields to over 85% in optimized examples.
Q: Is this synthesis method scalable for commercial API production?
A: Yes, the method utilizes common organic solvents like toluene and THF and operates at moderate temperatures (100-110°C), making it highly suitable for scale-up in standard stainless steel reactors without requiring cryogenic conditions.
Q: How is impurity control managed in this novel synthetic pathway?
A: Impurity control is achieved through the specificity of the palladium catalyst which directs the cyclization precisely, followed by a robust recrystallization step that removes residual catalyst and side products effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Hydroxy-3,4-Dihydro-2(1H)-Quinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain, and we are uniquely positioned to leverage technologies like CN108383781B to serve our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant with international standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-hydroxy-3,4-dihydro-2(1H)-quinolinone meets the exacting requirements necessary for downstream API synthesis, providing our clients with peace of mind regarding quality and consistency.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply strategy to drive efficiency and cost-effectiveness. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and logistical constraints. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being a partner that delivers both technical excellence and commercial value in the competitive landscape of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
