Scalable Chemical Synthesis of Bioactive Panax Notoginseng Polysaccharide Fragments for Pharmaceutical Applications
The pharmaceutical industry continuously seeks reliable sources for bioactive oligosaccharides, particularly those derived from traditional medicinal plants like Panax notoginseng. Patent CN1369500A presents a groundbreaking chemical synthesis method for the β-D-galactopyranosyl-(1→3)-[α-L-arabinofuranosyl-(1→6)]-D-galactopyranose active unit, which serves as the core repeating structure of natural notoginseng polysaccharides. This specific trisaccharide fragment is renowned for its potential medicinal value, including hemostatic and anti-inflammatory properties, making it a high-value target for drug development. Unlike traditional extraction methods which often yield heterogeneous mixtures, this patented chemical approach ensures structural homogeneity and precise control over stereochemistry. By leveraging specific protecting group strategies, the process overcomes the inherent challenges of carbohydrate synthesis, providing a robust pathway for producing high-purity pharmaceutical intermediates. This report analyzes the technical merits and commercial viability of this synthesis route for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining specific oligosaccharide fragments from natural sources like Panax notoginseng involves complex extraction and purification processes that are fraught with inefficiencies. Natural polysaccharides often exist as heterogeneous mixtures with varying chain lengths and branching patterns, making it extremely difficult to isolate a single, well-defined trisaccharide structure in high purity. Furthermore, the biological variability of the plant source can lead to inconsistent batch quality, which is unacceptable for rigorous pharmaceutical applications requiring strict regulatory compliance. Chemical synthesis via conventional routes often struggles with regioselectivity, particularly when differentiating between the multiple hydroxyl groups on the galactose ring. Without precise control, side reactions and isomer formation can drastically reduce overall yields and complicate downstream purification, leading to significant waste of expensive starting materials and reagents.
The Novel Approach
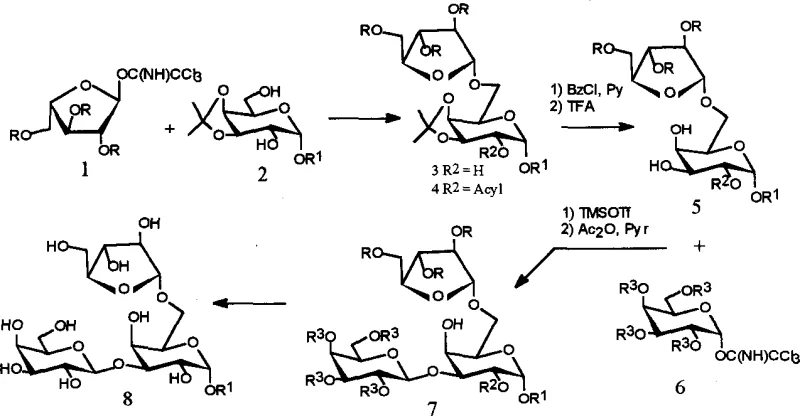
The method disclosed in patent CN1369500A introduces a sophisticated strategy utilizing 3,4-O-isopropylidene galactopyranoside as a key building block to address these selectivity issues. By temporarily masking the 3 and 4 positions of the galactose ring, the synthesis directs glycosylation specifically to the desired 6-position, enabling the efficient construction of the 1→6 linkage. Subsequent manipulation of the protecting groups allows for the precise formation of the 1→3 linkage, resulting in the correct 3,6-branched architecture found in nature. This modular approach not only simplifies the synthetic route but also significantly enhances the overall yield and purity of the final trisaccharide product. The use of trichloroacetimidate donors activated by Lewis acids ensures high stereoselectivity, minimizing the formation of unwanted anomers and reducing the burden on purification processes.
Mechanistic Insights into TMSOTf-Catalyzed Glycosylation
The core of this synthesis relies on the precise activation of trichloroacetimidate donors using trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a catalyst. This Lewis acid promotes the formation of an oxocarbenium ion intermediate, which is then attacked by the hydroxyl group of the glycosyl acceptor. The reaction conditions, typically maintained between -42°C and 0°C in anhydrous dichloromethane, are critical for controlling the reactivity and preventing side reactions such as hydrolysis of the sensitive imidate donor. The choice of solvent and temperature ensures that the kinetic product is favored, leading to the desired alpha or beta linkage with high fidelity. This mechanistic control is essential for building complex oligosaccharides where a single incorrect linkage can render the entire molecule biologically inactive.
Regioselectivity is further enforced by the strategic use of protecting groups, specifically the isopropylidene acetal which blocks the 3,4-positions of the galactose acceptor. This steric and electronic modulation ensures that the incoming sugar donor couples exclusively at the primary 6-hydroxyl group during the initial step. Following this, selective deprotection using trifluoroacetic acid reveals the secondary hydroxyl at the 3-position for the subsequent glycosylation event. This stepwise unmasking strategy is a hallmark of modern carbohydrate chemistry, allowing for the assembly of branched structures that would otherwise be inaccessible. The final deprotection using sodium methoxide or ammonia in methanol cleanly removes all acyl groups to yield the native trisaccharide structure without degrading the glycosidic bonds.
How to Synthesize Panax Notoginseng Trisaccharide Efficiently
The synthesis of this bioactive trisaccharide involves a sequence of highly specific chemical transformations that require careful monitoring of reaction parameters. The process begins with the preparation of the glycosyl acceptor, followed by sequential coupling reactions using activated donors. Each step necessitates strict anhydrous conditions and precise temperature control to maintain the integrity of the intermediates. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures, are outlined in the structured guide below to ensure reproducibility and safety in a laboratory or pilot plant setting.
- Prepare the glycosyl acceptor using 3,4-O-isopropylidene protected D-galactopyranoside to ensure regioselectivity during coupling.
- Perform the first glycosylation using fully benzoylated α-L-arabinofuranosyl trichloroacetimidate as the donor with TMSOTf catalyst.
- Execute selective deprotection and subsequent 1→3 linkage formation to complete the trisaccharide core structure.
Commercial Advantages for Procurement and Supply Chain Teams
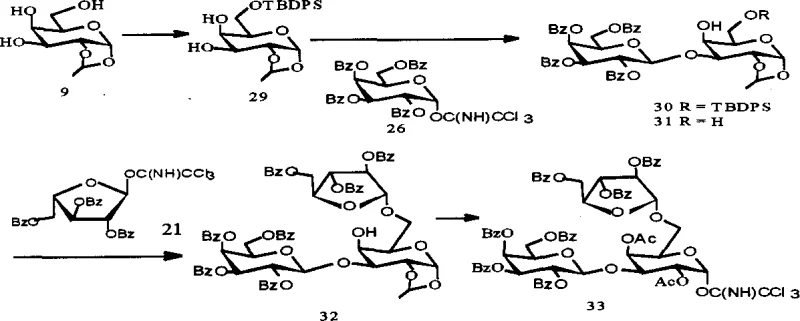
From a procurement perspective, this synthetic route offers substantial advantages over extraction-based sourcing, primarily due to the independence from agricultural variables and seasonal fluctuations. The reliance on commercially available chemical reagents such as benzoyl chloride, pyridine, and standard solvents ensures a stable and predictable supply chain. The high yields reported in specific steps, such as the 98.8% yield in the formation of intermediate 30, indicate a highly efficient process that minimizes raw material consumption. This efficiency translates directly into cost stability and reduced waste disposal requirements, aligning with modern green chemistry initiatives. Furthermore, the scalability of the reaction conditions, which do not require exotic catalysts or extreme pressures, facilitates a smoother transition from laboratory scale to commercial production.
- Cost Reduction in Manufacturing: The elimination of complex extraction and purification steps associated with natural polysaccharides significantly lowers the operational expenditure. By utilizing high-yielding chemical transformations, the process reduces the amount of starting material required per unit of final product, thereby optimizing the cost of goods sold. The use of robust protecting group strategies minimizes the need for repetitive purification, saving both time and solvent costs. Additionally, the ability to synthesize the exact active structure avoids the costs associated with standardizing variable natural extracts. These factors collectively contribute to a more economically viable manufacturing process for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Chemical synthesis provides a consistent and reliable source of the trisaccharide fragment, unaffected by crop failures or geopolitical issues impacting herbal supply chains. The reagents used are standard industrial chemicals with multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the synthetic route allows for production scheduling that is independent of harvest seasons, ensuring continuous availability for downstream drug manufacturing. This reliability is crucial for pharmaceutical companies that require guaranteed supply continuity to meet regulatory commitments and market demand without interruption.
- Scalability and Environmental Compliance: The process utilizes common organic solvents like dichloromethane and methanol, which are well-understood in terms of handling and waste management protocols. The high atom economy of the glycosylation steps reduces the volume of chemical waste generated per kilogram of product. Scalability is supported by the use of standard reaction vessels and temperature ranges that are easily replicated in large-scale reactors. The method's compatibility with standard industrial purification techniques, such as column chromatography and crystallization, ensures that scale-up does not introduce unforeseen technical barriers, facilitating a compliant and efficient production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this specific oligosaccharide structure. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating this intermediate into their drug development pipelines. The information covers aspects of reaction conditions, structural verification, and potential applications in the pharmaceutical sector.
Q: What is the key advantage of using 3,4-O-isopropylidene protection in this synthesis?
A: The 3,4-O-isopropylidene group provides critical chemoselectivity, allowing for the specific formation of 3,6-branched galactooligosaccharide derivatives without complex protecting group manipulations.
Q: What catalysts are recommended for the glycosylation steps?
A: The patent specifies trimethylsilyl trifluoromethanesulfonate (TMSOTf) or boron trifluoride ether solution as effective promoters for the trichloroacetimidate coupling reactions.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the method utilizes standard solvents like dichloromethane and pyridine and achieves high yields in key steps, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Panax Notoginseng Trisaccharide Supplier
NINGBO INNO PHARMCHEM stands ready to support your development needs with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of carbohydrate chemistry and can adapt the patented route to meet your specific purity and volume requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure every batch meets the highest international standards. Our commitment to quality and consistency makes us an ideal partner for long-term supply agreements in the competitive pharmaceutical market.
We invite you to contact our technical procurement team to discuss your specific project needs and request a Customized Cost-Saving Analysis. Our experts are available to provide specific COA data and route feasibility assessments tailored to your manufacturing constraints. By collaborating with us, you can accelerate your timeline to market while ensuring a secure and cost-effective supply of this critical bioactive intermediate. Let us help you optimize your supply chain and achieve your commercial goals with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
