Scalable Synthesis of ((1R,8S,9S,Z)-Bicyclo[6.1.0]non-4-en-9-yl)methanol for Bioorthogonal Applications
Scalable Synthesis of ((1R,8S,9S,Z)-Bicyclo[6.1.0]non-4-en-9-yl)methanol for Bioorthogonal Applications
The rapidly evolving field of bioorthogonal chemistry has created an unprecedented demand for highly specialized strained alkyne probes, particularly those capable of undergoing strain-promoted azide-alkyne cycloaddition (SPAAC) without cytotoxic copper catalysts. At the heart of this technology lies the critical intermediate ((1R,8S,9S,Z)-bicyclo[6.1.0]non-4-en-9-yl)methanol, a precursor essential for generating Bicyclo[6.1.0]nonyne (BCN) derivatives used in protein labeling and in vivo imaging. Patent CN114085130B discloses a groundbreaking synthetic methodology that addresses the longstanding bottlenecks associated with producing this complex scaffold. By leveraging a unique combination of rhodium-catalyzed cyclopropanation and stereoselective thermal decarboxylation, this innovation delivers a route that is not only chemically elegant but also commercially viable for industrial-scale production. This report analyzes the technical superiority of this approach, highlighting its potential to redefine supply chain standards for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ((1R,8S,9S,Z)-bicyclo[6.1.0]non-4-en-9-yl)methanol has been plagued by poor diastereoselectivity and cumbersome purification requirements. Traditional protocols typically involve the reaction of cyclooctadiene with ethyl diazoacetate in the presence of rhodium catalysts. As illustrated in the conventional pathway below, this reaction invariably produces a complex mixture of exo and endo ester isomers.
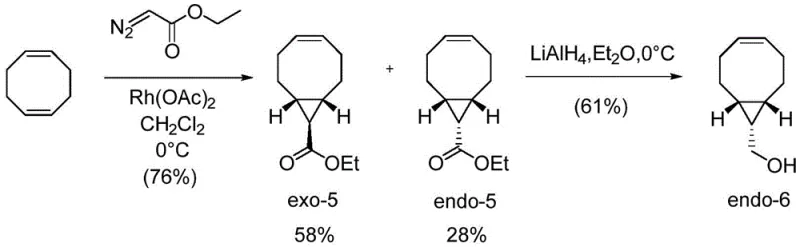
In standard implementations, the ratio of the undesired exo-configured compound to the desired endo-configured compound is often unfavorable, typically hovering around 58% exo to 28% endo, or in optimized variations, 41% exo to 24% endo. This lack of selectivity creates a massive downstream burden. To isolate the requisite endo-isomer, manufacturers are forced to employ silica gel column chromatography using precise solvent gradients (e.g., ethyl acetate to heptane). From a process chemistry perspective, column chromatography is the enemy of scalability; it is solvent-intensive, time-consuming, and difficult to automate for multi-kilogram batches. Consequently, the conventional route results in low overall throughput, high waste generation, and prohibitive costs that hinder the widespread adoption of BCN-based bioorthogonal tools in commercial drug discovery pipelines.
The Novel Approach
The methodology outlined in patent CN114085130B represents a paradigm shift by fundamentally altering the substrate and reaction sequence to enforce selectivity. Instead of relying on the separation of isomers post-reaction, this novel approach utilizes dimethyl diazomalonate as the carbene source. The initial cyclopropanation of cyclooctadiene (SM1) with dimethyl diazomalonate (SM2), catalyzed by dirhodium tetraacetate, proceeds with high efficiency to form the dicarboxylate intermediate (Compound 1).
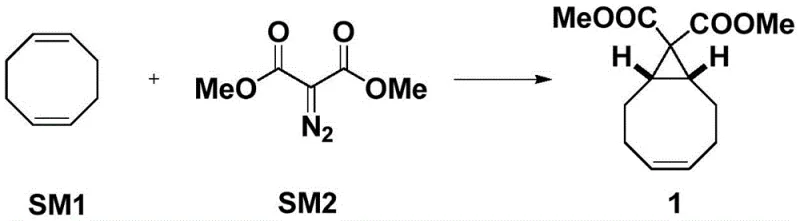
Crucially, this new route bypasses the immediate need for difficult isomer separation. The subsequent steps are designed to leverage the symmetry and reactivity of the dicarboxylate group to drive the formation of the single desired stereoisomer. By shifting the selectivity burden from a physical separation (chromatography) to a chemical transformation (decarboxylation), the process dramatically simplifies the workflow. The result is a streamlined synthesis that avoids the pitfalls of the traditional ethyl diazoacetate route, offering a direct path to the target alcohol with significantly reduced operational complexity and enhanced suitability for a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Stereoselective Decarboxylation
The cornerstone of this invention's success lies in the second step: the thermal decarboxylation of the dicarboxylate intermediate. This transformation is not merely a functional group removal but a sophisticated stereochemical control element. When Compound 1 is subjected to hydrolysis and subsequent heating in an aqueous sodium hydroxide solution at 160-165°C, it undergoes decarboxylation to yield a mixture of mono-acids. However, the distribution is heavily skewed towards the desired endo-configuration (Compound 3).
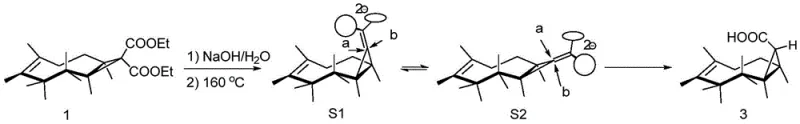
The mechanistic rationale, as detailed in the patent, involves the formation of enol-type intermediates (S1 and S2) during the loss of carbon dioxide. The stereochemical outcome is dictated by the direction of proton attack on these intermediates. Attack from the 'a' direction is sterically hindered by the proximal cyclopropane ring and the bulk of the molecular framework, leading to only trace amounts of the exo-configured byproduct (Compound 2). Conversely, proton attack from the 'b' direction encounters significantly less steric resistance, thereby funneling the reaction predominantly towards the formation of Compound 3 (the endo-isomer). This intrinsic bias allows the process to achieve a selectivity where the desired endo-compound constitutes the major product (55% yield vs 5% for exo), effectively self-purifying the stream before the final reduction step. Such mechanistic elegance ensures that the final product meets stringent purity specifications without the need for extensive chromatographic intervention.
How to Synthesize ((1R,8S,9S,Z)-Bicyclo[6.1.0]non-4-en-9-yl)methanol Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the inherent selectivity of the pathway. The process begins with the careful addition of the diazo compound to the cyclooctadiene solution to manage exothermicity, followed by a prolonged stirring period at room temperature to ensure complete conversion to the dicarboxylate. The subsequent decarboxylation demands high-temperature conditions in a sealed or reflux system to drive the equilibrium forward, followed by a strategic workup involving ether precipitation to isolate the solid acid intermediate. Finally, the reduction is performed under anhydrous conditions to prevent reagent decomposition. For laboratory and pilot-scale teams looking to replicate this high-efficiency route, the detailed standardized synthesis steps are provided in the guide below.
- Perform rhodium-catalyzed cyclopropanation of cyclooctadiene with dimethyl diazomalonate to obtain the dicarboxylate intermediate with high selectivity.
- Execute thermal decarboxylation using aqueous sodium hydroxide at 160-165°C to selectively form the endo-configured mono-acid via steric control.
- Reduce the resulting carboxylic acid using lithium aluminum hydride in tetrahydrofuran to yield the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from conventional chromatography-dependent routes to this crystallization-based protocol offers transformative economic benefits. The primary value driver is the complete elimination of silica gel column chromatography, which is notoriously expensive in terms of both consumable materials (silica, solvents) and labor hours. By replacing this batch-wise separation with filtration and crystallization, the manufacturing process becomes continuous-friendly and significantly more robust.
- Cost Reduction in Manufacturing: The removal of column chromatography directly translates to substantial cost savings in bioorthogonal chemistry manufacturing. Solvent consumption is drastically reduced because large volumes of eluents are no longer required for purification. Furthermore, the recovery of solvents is more efficient in a crystallization/filtration workflow compared to the complex mixtures generated by chromatography. The use of commodity reagents like dimethyl diazomalonate and sodium hydroxide, rather than specialized separation media, further lowers the Bill of Materials (BOM) cost, making the final BCN alcohol intermediate more price-competitive for high-volume applications.
- Enhanced Supply Chain Reliability: Reducing lead time for high-purity pharmaceutical intermediates is critical in the fast-paced drug discovery sector. Conventional methods are prone to bottlenecks; a single overloaded chromatography column can delay an entire production campaign. This novel method mitigates that risk by utilizing unit operations (filtration, distillation) that are easily parallelized and scaled. The robustness of the decarboxylation step ensures consistent batch-to-batch quality, reducing the rate of failed batches and ensuring a steady flow of material to downstream conjugation teams. This reliability is essential for maintaining the continuity of supply for critical research reagents.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior. Chromatography generates significant hazardous waste streams containing mixed organic solvents and silica dust. By minimizing solvent usage and eliminating silica waste, the process aligns better with green chemistry principles and reduces the burden on waste treatment facilities. The commercial scale-up of complex strained alkynes is thus facilitated by a cleaner, safer process profile that meets rigorous environmental compliance standards without sacrificing yield or purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of ((1R,8S,9S,Z)-bicyclo[6.1.0]non-4-en-9-yl)methanol. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring that stakeholders have accurate information for decision-making.
Q: How does this new method improve selectivity compared to conventional routes?
A: Conventional methods using ethyl diazoacetate typically yield a difficult-to-separate mixture of exo and endo isomers (approx. 2:1 ratio). This novel protocol utilizes dimethyl diazomalonate followed by a stereoselective decarboxylation step, which inherently favors the formation of the desired endo-configuration through steric hindrance mechanisms, significantly simplifying purification.
Q: Is column chromatography required for purification in this process?
A: No. A major advantage of this patented technology (CN114085130B) is the elimination of silica gel column chromatography. The intermediates and final product can be isolated through crystallization, filtration, and concentration, which is critical for reducing costs and facilitating large-scale manufacturing.
Q: What are the typical yields for the key steps in this synthesis?
A: The process demonstrates robust efficiency: the initial cyclization yields approximately 72%, the decarboxylation step provides the desired endo-isomer in about 55% yield (with minimal exo-impurity), and the final reduction step achieves a high yield of 93%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ((1R,8S,9S,Z)-Bicyclo[6.1.0]non-4-en-9-yl)methanol Supplier
As the demand for bioorthogonal tools continues to surge in the pharmaceutical and biotechnology sectors, securing a stable supply of high-quality precursors is paramount. NINGBO INNO PHARMCHEM stands ready to support your R&D and commercial needs with our advanced manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early discovery phase or late-stage clinical manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of ((1R,8S,9S,Z)-bicyclo[6.1.0]non-4-en-9-yl)methanol meets the exacting standards required for sensitive biological applications.
We invite you to leverage our technical expertise to optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patented technology can enhance your project's efficiency and reduce your overall cost of goods sold.
