Advanced Manufacturing of Chiral Diazabicyclo Nonane for High-Performance Antibiotic Intermediates
Advanced Manufacturing of Chiral Diazabicyclo Nonane for High-Performance Antibiotic Intermediates
The pharmaceutical industry continuously demands more efficient and safer pathways for constructing complex chiral scaffolds essential for next-generation antibiotics. Patent CN101657448B introduces a groundbreaking methodology for the preparation of (S,S)-2,8-diazabicyclo[4.3.0]nonane and its enantiomers, which serve as critical building blocks for quinolone and naphthyridine derivatives such as Moxifloxacin. This novel process addresses long-standing challenges in stereochemical control and operational safety that have plagued conventional synthesis routes. By leveraging a unique sulfonamide masking strategy, the invention enables a streamlined four-step sequence that avoids hazardous reagents while delivering exceptional optical purity. For R&D directors and procurement specialists, understanding this technological shift is vital for securing a robust supply of high-purity pharmaceutical intermediates. The following analysis details how this innovation transforms the manufacturing landscape for these valuable compounds.
![Chemical structures of (S,S) and (R,R) enantiomers of 2,8-diazabicyclo[4.3.0]nonane](/insights/img/s-s-2-8-diazabicyclo-nonane-synthesis-supplier-20260313062037-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,8-diazabicyclo[4.3.0]nonane derivatives has relied on routes that present significant safety and efficiency bottlenecks for commercial scale-up. One prominent prior art method, described in EP 0350733B1, utilizes a benzyl protection strategy that necessitates the use of lithium aluminum hydride (LAH) for the removal of dicarbonyl groups. LAH is notoriously pyrophoric and requires stringent anhydrous conditions, posing severe safety risks in large-scale reactors and complicating waste disposal protocols. Furthermore, another existing pathway disclosed in US 5770597 employs sodium hydride (NaH) in dimethylformamide (DMF) slurries. This combination is particularly dangerous due to the evolution of hydrogen gas throughout the reaction and the documented potential for explosive accidents within NaH and DMF mixture systems. These conventional approaches not only endanger facility personnel but also introduce substantial cost variability due to the need for specialized safety infrastructure and expensive solvent recovery systems.
The Novel Approach
In stark contrast, the process disclosed in CN101657448B revolutionizes the synthesis by substituting hazardous reagents with economically viable and safer alternatives. The inventors have successfully replaced sodium hydride with sodium ethoxide and substituted the problematic DMF solvent with ethanol, a widely available and environmentally benign alcohol. This strategic substitution eliminates the risk of hydrogen evolution explosions and simplifies the reaction environment significantly. Additionally, the new method utilizes a sulfonamide group as a masked nitrogen source, which avoids the need for harsh LAH reductions entirely. The resulting pathway is not only safer but also more economical, as ethanol is far less costly than DMF and the sulfonamide intermediates exhibit superior crystallinity, facilitating easier purification. This shift represents a paradigm change in how these complex bicyclic amines are manufactured, offering a sustainable route for reliable pharmaceutical intermediate supplier networks.
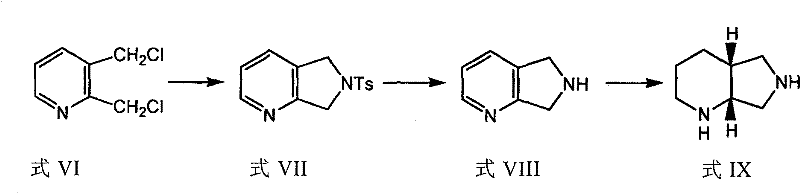
Mechanistic Insights into Sulfonamide-Masked Cyclization
The core innovation of this technology lies in the chemical behavior of the sulfonamide group, which acts as a masked ammonia molecule to facilitate the construction of the bicyclic framework. Sulfonamides possess a unique electronic property where the electron-withdrawing sulfonyl group significantly enhances the acidity of the germinal hydrogen atoms attached to the nitrogen. This increased acidity allows for smooth alkylation under basic conditions using mild bases like sodium ethoxide, rather than requiring aggressive deprotonation agents. Following alkylation with 2,3-dihalogenomethylpyridine, the sulfonamide intermediate undergoes cyclization to form the pyrrolopyridine core. The stability of the N-S bond during subsequent transformations is crucial, as it withstands the conditions required for ring saturation while remaining labile enough for final cleavage. This delicate balance ensures that the nitrogen atom is protected during the harsh hydrogenation steps but can be efficiently liberated in the final stage to yield the free amine.
Impurity control is inherently built into this mechanistic design through the physical properties of the sulfonamide intermediates. Unlike corresponding amides or carbamates, the sulfonamide derivatives generated in this process exhibit high crystallinity, which is a critical attribute for pharmaceutical manufacturing. This crystalline nature allows for the effective removal of non-polar impurities and side products through simple recrystallization steps, typically using ethanol or alcohol-water mixtures. Moreover, the chiral resolution step leverages this crystallinity by forming diastereomeric salts with enantiomerically pure organic acids such as mandelic acid or dibenzoyltartaric acid. The differential solubility of these salts enables the selective precipitation of the desired (S,S)-enantiomer with high optical purity. This mechanism ensures that the final product meets stringent purity specifications without requiring complex chromatographic separations, thereby enhancing the overall robustness of the commercial scale-up of complex pharmaceutical intermediates.
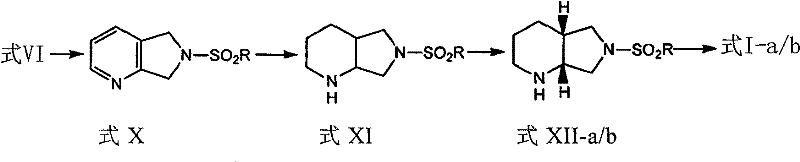
How to Synthesize (S,S)-2,8-diazabicyclo[4.3.0]nonane Efficiently
The synthesis of this high-value chiral intermediate follows a logical four-step sequence designed for maximum operational efficiency and yield optimization. The process begins with the condensation of 2,3-dihalogenomethylpyridine with a substituted sulfonamide in the presence of sodium ethoxide to form the key pyrrolopyridine scaffold. This is followed by a catalytic hydrogenation step to saturate the pyridine ring, creating the octahydro structure necessary for the bicyclic framework. The third critical step involves the chiral resolution of the racemic mixture using optically pure acids to isolate the specific enantiomer required for antibiotic activity. Finally, the protecting sulfonamide group is cleaved under acidic conditions to reveal the free amine. Detailed standardized synthesis steps see the guide below.
- Condense 2,3-dihalogenomethylpyridine with substituted sulfonamide using sodium ethoxide in ethanol to form the pyrrolopyridine intermediate.
- Perform catalytic hydrogenation on the pyridine ring using Pd/C or PtO2 to generate the octahydro-pyrrolopyridine structure.
- Resolve the racemic mixture using enantiomerically pure organic acids like mandelic acid to isolate the desired S,S-enantiomer salt.
- Cleave the N-S bond using hydrobromic acid and propionic acid to yield the final free base or stable dihydrobromide salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like sodium hydride and lithium aluminum hydride directly translates to reduced operational risks and lower insurance costs for manufacturing facilities. Furthermore, the substitution of expensive and toxic solvents like DMF with ethanol significantly lowers raw material costs and simplifies solvent recovery and waste treatment processes. The high crystallinity of the intermediates ensures consistent quality and reduces the likelihood of batch failures, which is critical for maintaining supply chain continuity. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of costly and hazardous reagents with economical alternatives like sodium ethoxide and ethanol drives down the overall cost of goods sold. By avoiding the need for specialized handling equipment required for pyrophoric materials, capital expenditure is also minimized. The high yields reported in the patent examples indicate efficient material utilization, reducing waste and maximizing output per batch. Additionally, the ability to recover and reuse chiral resolving agents further enhances the economic viability of the process. These qualitative improvements ensure substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The use of widely available and stable reagents ensures that production is not vulnerable to supply disruptions associated with specialized or controlled chemicals. Ethanol and sodium ethoxide are commodity chemicals with robust global supply networks, guaranteeing consistent availability. The robustness of the reaction conditions, which tolerate standard industrial equipment, reduces the risk of unplanned downtime due to equipment corrosion or failure. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream antibiotic production schedules are met without delay. The process stability supports a dependable partnership with any reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory to commercial production scales. The use of ethanol as a primary solvent aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. The crystalline nature of the intermediates minimizes the need for energy-intensive purification methods like distillation or chromatography. Furthermore, the avoidance of heavy metal catalysts in the resolution step simplifies waste stream management and ensures compliance with stringent environmental regulations. This facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining high environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent data to provide accurate guidance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your supply chain. The responses highlight the safety, purity, and scalability advantages of the new method.
Q: Why is the sulfonamide masking strategy superior to benzyl protection for this intermediate?
A: The sulfonamide group enhances the acidity of germinal hydrogens, facilitating easier alkylation under milder conditions compared to benzyl protection which requires harsh LAH reduction for removal.
Q: How does this process improve safety compared to traditional NaH/DMF methods?
A: This method replaces hazardous sodium hydride and dimethylformamide with sodium ethoxide and ethanol, significantly reducing explosion risks and simplifying waste treatment protocols.
Q: What optical purity can be achieved using the mandelic acid resolution method?
A: The process consistently achieves an enantiomeric excess (ee) greater than 99% through crystallization of the diastereomeric salt followed by basification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S,S)-2,8-diazabicyclo[4.3.0]nonane Supplier
The technological potential of this sulfonamide-based synthesis route is immense, offering a pathway to produce high-quality antibiotic intermediates with superior safety and efficiency profiles. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative process can be seamlessly implemented at an industrial level. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch meets the exacting standards required by global pharmaceutical companies. We understand the critical nature of chiral intermediates in drug development and are equipped to handle the complexities of this specific chemistry.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a reliable supply of high-value intermediates backed by deep technical expertise and a commitment to quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
