Advanced Catalytic Transfer Hydrogenation for Commercial Dihydroberberine Production
Advanced Catalytic Transfer Hydrogenation for Commercial Dihydroberberine Production
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for the synthesis of bioactive alkaloids, particularly those with significant metabolic potential like dihydroberberine. Patent CN108997332B introduces a transformative preparation method that addresses critical bottlenecks in the existing manufacturing landscape. This technology leverages a catalytic transfer hydrogenation strategy to convert berberine salts directly into dihydroberberine with exceptional efficiency. By shifting away from hazardous stoichiometric reductants, this innovation not only enhances operational safety but also drastically simplifies the downstream purification process. As a leading entity in fine chemical manufacturing, we recognize this patent as a pivotal advancement for producing high-purity pharmaceutical intermediates. The structural integrity of the target molecule, dihydroberberine, is preserved through mild reaction conditions, ensuring minimal formation of over-reduced impurities that often plague traditional reduction protocols.
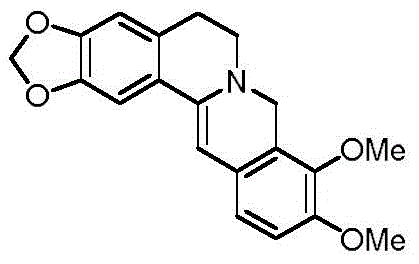
This breakthrough offers a compelling value proposition for R&D directors and procurement managers alike, promising a reliable pharmaceutical intermediate supplier pathway that balances cost, quality, and safety. The ability to utilize common hydrogen donors and heterogeneous catalysts positions this method as a superior alternative for the commercial scale-up of complex alkaloids, ensuring a consistent supply chain for downstream drug development programs targeting metabolic disorders and oncology applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydroberberine has been fraught with significant technical and safety challenges that hinder industrial adoption. The conventional approach typically involves the reduction of berberine using metal hydrides under alkaline conditions. While these methods can offer moderate yields, they rely on reagents such as lithium aluminum hydride or metal borohydrides, which are inherently hazardous to transport, store, and handle on a large scale. Furthermore, the post-processing of these reactions is notoriously difficult; removing residual metal species often requires complex complexation steps or extensive washing, which increases waste generation and processing time. Alternative routes reported in literature, such as those utilizing precious metal rhodium catalysts, introduce prohibitive costs due to the expense of the catalyst and the lack of commercially available starting materials, rendering them economically unviable for mass production.
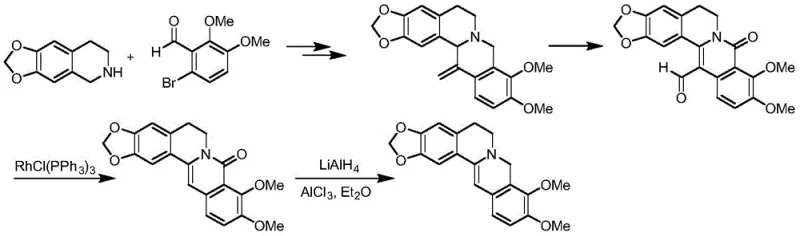
Additionally, strategies employing the Pictet-Spengler reaction to construct the isoquinoline core have demonstrated moderate success but suffer from critical scalability issues. These routes often require an excess of aldehyde, leading to lower atom economy and increased impurity profiles. A major drawback is the partial hydrolysis of acetal groups during the reduction ammoniation process, which significantly reduces yield upon amplification. Literature indicates that the separation yield of intermediate dihydroberberine in these legacy processes is often as low as 55%, creating a substantial bottleneck for supply chain continuity. The reliance on column chromatography for purification in many of these older methods further exacerbates cost and time inefficiencies, making them unsuitable for modern GMP manufacturing environments.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in patent CN108997332B utilizes a catalytic transfer hydrogenation mechanism that fundamentally reshapes the production landscape. This method employs readily available berberine salts, such as hydrochloride, nitrate, or acetate, as the starting material, reacting them with safe hydrogen donors like ammonium formate, formic acid, or isopropanol in the presence of a heterogeneous catalyst. The reaction proceeds under mild alkaline conditions, utilizing bases such as sodium bicarbonate or potassium carbonate to facilitate the transformation. This strategy eliminates the need for dangerous metal hydrides and expensive precious metals like rhodium, replacing them with cost-effective catalysts such as palladium on carbon or Raney nickel. The result is a process that is not only safer but also delivers superior selectivity and yield, often exceeding 95% molar yield with product purity reaching 98% without the need for chromatographic purification.
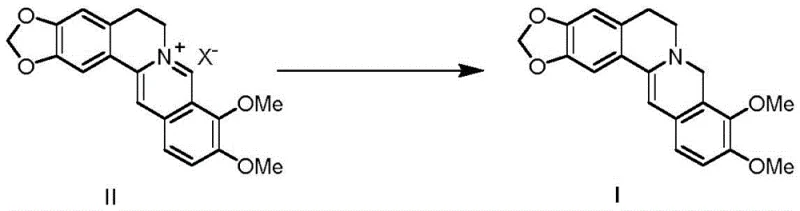
The versatility of this novel approach allows for flexibility in solvent selection, including methanol, ethanol, tetrahydrofuran, and even water, providing manufacturers with the ability to optimize for green chemistry principles. By avoiding the formation of difficult-to-remove metal complexes and minimizing side reactions like acetal hydrolysis, this method ensures a cleaner reaction profile. The simplicity of the workup procedure, which involves mere filtration of the catalyst followed by concentration and slurrying, represents a paradigm shift in process efficiency. This makes the technology ideally suited for cost reduction in API manufacturing, offering a streamlined path from raw material to high-value intermediate that aligns perfectly with the demands of modern pharmaceutical supply chains.
Mechanistic Insights into Catalytic Transfer Hydrogenation
The core of this technological advancement lies in the efficient mechanism of catalytic transfer hydrogenation, which facilitates the selective reduction of the iminium ion within the berberine skeleton. In this process, the heterogeneous catalyst, such as palladium black or palladium on carbon, acts as a surface mediator for hydrogen transfer. When a hydrogen donor like ammonium formate is employed, it decomposes on the catalyst surface to generate active hydrogen species in situ. These species are then transferred directly to the electron-deficient iminium carbon of the berberine cation, effecting a 1,2-reduction to form the saturated amine bond characteristic of dihydroberberine. The presence of a base is crucial in this mechanism, as it helps to neutralize the acid generated during the decomposition of the hydrogen donor and maintains the reaction environment conducive to the stability of the intermediate species. This surface-mediated process ensures that the reduction is highly chemoselective, targeting the specific double bond without affecting other sensitive functional groups on the aromatic rings, such as the methoxy substituents.
Impurity control is another critical aspect where this mechanism excels compared to stoichiometric reductions. In traditional metal hydride reductions, the high reactivity of the reductant can lead to over-reduction or attack on other electrophilic sites, generating a complex mixture of byproducts that are difficult to separate. In the catalytic transfer hydrogenation pathway, the controlled release of hydrogen from the donor molecule limits the availability of reactive species, thereby preventing over-reduction. Furthermore, the use of berberine salts with stable counterions like chloride or nitrate ensures that the starting material remains stable throughout the reaction, minimizing degradation pathways. The high selectivity observed, with liquid phase purity consistently around 98%, indicates that the catalyst effectively discriminates between the target iminium bond and other potential reduction sites. This mechanistic precision translates directly to commercial value, as it reduces the burden on downstream purification units and ensures a consistent quality profile for the final product.
How to Synthesize Dihydroberberine Efficiently
The implementation of this synthesis route is designed for straightforward execution in a standard chemical manufacturing facility. The process begins with the dissolution of the berberine salt in a polar protic or aprotic solvent, followed by the sequential addition of the catalyst, hydrogen donor, and base. The reaction is typically conducted at mild temperatures ranging from 40°C to 80°C, which minimizes energy consumption and thermal stress on the equipment. Monitoring the reaction progress via TLC ensures precise endpoint determination, preventing unnecessary prolongation of the batch time. Once the reaction is complete, the heterogeneous catalyst is easily removed by simple filtration, allowing for immediate recovery and potential recycling of the precious metal. The filtrate is then concentrated, and the crude product is isolated through a slurry or crystallization step using water or a mixed solvent system. This operational simplicity underscores the industrial viability of the method, making it an attractive option for manufacturers looking to optimize their production lines.
- Dissolve berberine salt (e.g., hydrochloride, nitrate, or acetate) in a suitable solvent such as methanol, isopropanol, or tetrahydrofuran under inert atmosphere.
- Add a heterogeneous catalyst like 10% palladium on carbon or Raney nickel, followed by a hydrogen donor such as ammonium formate or isopropanol, and a base like sodium bicarbonate.
- Stir the mixture at mild temperatures (40-80°C) until reaction completion, then filter off the catalyst and isolate the product via crystallization or slurry without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic transfer hydrogenation technology offers profound strategic advantages that extend beyond mere technical feasibility. The shift from hazardous, specialized reagents to commodity chemicals significantly de-risks the supply chain. By utilizing berberine salts, which are abundant and inexpensive natural product derivatives, manufacturers can secure a stable raw material base that is less susceptible to market volatility compared to synthetic precursors required in other routes. The elimination of complex purification steps like column chromatography not only reduces the consumption of silica gel and solvents but also shortens the overall cycle time per batch. This efficiency gain allows for higher throughput in existing facilities, effectively increasing capacity without the need for capital-intensive infrastructure upgrades. Consequently, this leads to substantial cost savings in the overall manufacturing budget, enhancing the competitiveness of the final pharmaceutical product.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive and dangerous reagents with cost-effective alternatives. The use of heterogeneous catalysts like palladium on carbon or Raney nickel, which can often be recovered and reused, drastically lowers the cost of goods sold compared to homogeneous precious metal catalysts or stoichiometric metal hydrides. Additionally, the avoidance of complex workup procedures reduces labor costs and waste disposal fees associated with hazardous metal residues. The high yield and purity achieved directly translate to better material utilization, minimizing the loss of valuable starting materials. These factors combined create a leaner, more cost-efficient production model that maximizes profit margins while maintaining high quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available raw materials and reagents. Berberine hydrochloride and ammonium formate are commodity chemicals with robust global supply networks, reducing the risk of shortages that can plague specialized synthetic intermediates. The mild reaction conditions and tolerance for various solvents provide flexibility in sourcing, allowing manufacturers to adapt to regional availability without compromising process performance. Furthermore, the simplified process flow reduces the number of unit operations, thereby decreasing the probability of operational delays or equipment failures. This reliability ensures consistent delivery schedules to downstream clients, fostering stronger long-term partnerships and trust in the supply network.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with green chemistry principles and increasingly stringent global regulations. The absence of toxic metal hydrides and the reduction of heavy metal waste simplify the environmental impact assessment and permitting processes for new manufacturing lines. The ability to perform the reaction in greener solvents like ethanol or water further enhances the sustainability profile of the operation. Scalability is inherent in the design, as the heterogeneous nature of the catalyst and the exothermic management of the hydrogen transfer are well-understood engineering challenges that can be easily managed in large-scale reactors. This ensures that the transition from pilot scale to commercial production is smooth and compliant with environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydroberberine synthesis technology. These insights are derived directly from the patent data and practical manufacturing considerations, aiming to provide clarity for stakeholders evaluating this process for integration into their supply chains. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the primary safety advantages of this dihydroberberine synthesis method?
A: Unlike conventional methods utilizing hazardous metal hydrides like lithium aluminum hydride, this patented process employs safe hydrogen donors such as ammonium formate or isopropanol, significantly reducing transport and storage risks while eliminating difficult post-processing steps associated with metal waste removal.
Q: How does this route improve purification efficiency compared to prior art?
A: The method achieves high selectivity and purity (up to 98%) allowing for isolation via simple filtration and crystallization. This avoids the need for complex and costly column chromatography often required in Pictet-Spengler routes or metal hydride reductions, thereby streamlining the manufacturing workflow.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and readily available raw materials like berberine hydrochloride and common heterogeneous catalysts. The mild reaction conditions and simple workup procedure make it highly scalable and economically viable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroberberine Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our team of expert chemists and engineers is adept at optimizing catalytic processes to meet stringent purity specifications, leveraging rigorous QC labs to guarantee that every batch of dihydroberberine meets the highest international standards. We understand the critical importance of consistency in pharmaceutical intermediates and have established robust protocols to monitor and control every variable in the synthesis, from raw material intake to final packaging. Our commitment to quality and safety makes us an ideal partner for companies seeking to secure a stable supply of this vital metabolic therapeutic precursor.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this catalytic method. We encourage potential partners to reach out for specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to engineer a supply chain solution that drives efficiency, reduces costs, and accelerates your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
