Advanced Copper-Catalyzed Synthesis of Beta-Functionalized Multi-Substituted Allyl Alcohols for Commercial Scale
The chemical landscape for constructing complex molecular architectures is constantly evolving, driven by the need for more efficient and selective synthetic methodologies. Patent CN100528820C introduces a significant breakthrough in the synthesis of beta-functionalized multi-substituted allyl alcohols, which are pivotal intermediates in the production of high-value pharmaceuticals and agrochemicals. This technology leverages a copper-catalyzed carbometallation reaction between Grignard reagents and allenols, enabling the formation of new carbon-carbon and carbon-metal bonds with exceptional precision. Unlike traditional methods that often struggle with regioselectivity and substrate scope, this approach allows for the simultaneous introduction of multiple substituents, thereby expanding the chemical space available to process chemists. The operational simplicity and the use of readily available reagents make this patent a cornerstone for developing robust manufacturing processes for complex organic molecules. By addressing the limitations of previous techniques, this method offers a reliable pathway for producing high-purity intermediates that meet the stringent quality standards required by the global fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-functionalized allyl alcohols has relied heavily on the carbometallation of alkynols, a process that is inherently restricted in its scope and selectivity. Conventional literature indicates that when alkynols are used as substrates, the reaction predominantly yields primary alcohols, severely limiting the structural diversity of the final products. Furthermore, attempts to utilize secondary or tertiary alkynols often result in poor regioselectivity or the formation of undesired alpha-functionalized isomers, complicating the purification process and reducing overall yield. Previous reports on allenol carbometallation have typically been confined to the simplest allenol substrates or required highly active allyl metal reagents that are difficult to handle on a large scale. Some existing methods, such as those described in earlier patents, are not compatible with secondary Grignard reagents and show only general selectivity towards aryl Grignard reagents, creating significant bottlenecks for synthesizing diverse molecular libraries. These limitations necessitate multi-step synthetic routes or extensive purification efforts, which drive up costs and extend lead times for commercial production. Consequently, the industry has long sought a more versatile and selective method that can accommodate a broader range of substrates without compromising on efficiency or product quality.
The Novel Approach
The methodology outlined in patent CN100528820C represents a paradigm shift by utilizing substituted allenols as the primary substrates for carbometallation reactions. This novel approach effectively overcomes the regioselectivity issues associated with alkynols, allowing for the high-yield synthesis of both secondary and tertiary allyl alcohols. The reaction is catalyzed by cuprous chloride, a cost-effective and widely available transition metal salt, which facilitates the formation of a key organocopper intermediate that dictates the stereochemical outcome of the transformation. By expanding the substrate scope to include secondary and tertiary allenols, this method provides chemists with the flexibility to introduce a wide array of functional groups at the beta-position of the allylic system. The process is characterized by its operational simplicity, requiring standard laboratory equipment and conditions that are easily transferable to industrial reactors. Moreover, the products generated through this route are reported to be easy to separate and purify, significantly reducing the downstream processing burden. This combination of high selectivity, broad substrate tolerance, and ease of purification makes the novel approach an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into CuCl-Catalyzed Carbometallation
The core of this synthetic innovation lies in the copper-catalyzed carbometallation mechanism, which proceeds through a well-defined organometallic pathway to ensure high regio- and stereoselectivity. Under the influence of the cuprous chloride catalyst, the Grignard reagent undergoes a transmetallation process to generate an active organocopper species in situ. This species then adds across the allene double bond of the allenol substrate in a highly controlled manner, forming a new carbon-carbon bond and a stable carbon-metal intermediate, often referred to as intermediate 2 in the patent documentation. The presence of the hydroxyl group in the allenol substrate plays a crucial role in directing the addition, likely through coordination with the copper center, which locks the conformation and prevents unwanted isomerization. This mechanistic feature is critical for achieving the observed beta-functionalization, as it prevents the formation of alpha-isomers that are common in uncatalyzed or poorly catalyzed reactions. The resulting organometallic intermediate is sufficiently stable to withstand the reaction conditions yet reactive enough to be trapped by various electrophiles in the subsequent step.
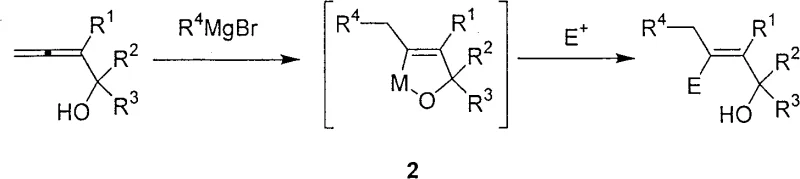
Following the formation of the organocopper intermediate, the reaction proceeds to the quenching phase where various electrophiles are introduced to functionalize the beta-position. The patent specifies that electrophiles such as iodine or allyl bromide can be used to cap the reaction, resulting in the final beta-functionalized multi-substituted allyl alcohol products. The high stereoselectivity of the process is maintained throughout this step, ensuring that the chiral information from the starting material, if present, is preserved or selectively transformed. For instance, when chiral allenols are employed, the reaction can produce optically active products with high enantiomeric excess, which is a vital attribute for the synthesis of active pharmaceutical ingredients. The mechanism also accounts for the compatibility with a wide range of Grignard reagents, including primary, secondary, alkyl, aryl, and substituted aryl variants, demonstrating the robustness of the catalytic cycle. Understanding this mechanistic pathway is essential for process optimization, as it allows chemists to fine-tune reaction parameters such as temperature and stoichiometry to maximize yield and purity while minimizing byproduct formation.
How to Synthesize Beta-Functionalized Multi-Substituted Allyl Alcohols Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to ensure the formation of the desired intermediate and subsequent product. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere to prevent oxidation of the sensitive organometallic species. Cuprous chloride, the allenol substrate, and anhydrous ether are combined in a reaction vessel and cooled to cryogenic temperatures, typically around -78°C, to control the exothermic nature of the Grignard addition. The Grignard reagent is then added dropwise to the cooled mixture, after which the reaction is allowed to warm slowly to room temperature and stirred for an extended period to ensure complete conversion. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency protocol.
- Prepare the reaction mixture with cuprous chloride, allenol, and ether under nitrogen protection, cooling to -78°C.
- Add the Grignard reagent solution dropwise, allow to warm to room temperature, and react for 10-18 hours to form the intermediate.
- Quench with electrophiles like iodine or allyl bromide, followed by standard workup and purification to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly address the pain points of procurement and supply chain management in the fine chemical sector. The use of readily available and inexpensive reagents such as cuprous chloride and common Grignard reagents significantly lowers the raw material costs compared to methods requiring exotic catalysts or specialized organometallic compounds. The high regio- and stereoselectivity of the reaction minimizes the formation of byproducts, which reduces the need for complex and costly purification steps such as extensive chromatography or recrystallization. This efficiency translates into shorter production cycles and higher throughput, allowing suppliers to meet tight delivery schedules and maintain consistent inventory levels for their clients. Furthermore, the scalability of the process ensures that production can be ramped up from laboratory scale to multi-ton commercial quantities without significant re-engineering of the manufacturing line. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of simple ether solvents drastically reduce the overall cost of goods sold for these intermediates. By avoiding the need for specialized reagents that require complex synthesis or importation, manufacturers can achieve significant savings on raw material procurement. The high yield and selectivity of the reaction mean that less starting material is wasted, further optimizing the material balance and reducing waste disposal costs. Additionally, the simplified workup procedure reduces the consumption of utilities such as energy and water, contributing to lower operational expenditures. These cost advantages can be passed down the supply chain, offering competitive pricing for end-users without compromising on product quality or purity specifications.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like cuprous chloride and standard Grignard reagents ensures a stable and secure supply of raw materials, mitigating the risk of shortages that can disrupt production. Since the reagents are widely produced and available from multiple suppliers, procurement teams can diversify their sourcing strategies to avoid single points of failure. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the likelihood of batch failures and ensuring consistent output. This reliability is crucial for maintaining long-term supply agreements with pharmaceutical companies that require guaranteed availability of key intermediates for their drug manufacturing pipelines. Consequently, partners adopting this technology can offer greater supply security and flexibility to their customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations that are easily implemented in existing chemical manufacturing facilities. The use of ether as a solvent, while requiring careful handling, is well-understood in the industry, and recovery systems can be easily integrated to minimize environmental impact. The high selectivity of the reaction reduces the generation of hazardous waste streams, simplifying compliance with increasingly stringent environmental regulations. Moreover, the ability to synthesize complex molecules in fewer steps reduces the overall carbon footprint of the manufacturing process. This alignment with green chemistry principles not only meets regulatory requirements but also enhances the corporate sustainability profile of the manufacturer, which is increasingly important for global supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation to provide clarity on its practical application. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of integrating this technology into their existing workflows. The responses highlight the versatility, efficiency, and scalability of the method, reinforcing its value proposition for the production of high-purity chemical intermediates.
Q: What are the advantages of using allenols over alkynols in this synthesis?
A: Using allenols allows for the synthesis of secondary and tertiary alcohols with high regioselectivity, overcoming the limitations of alkynol substrates which often yield only primary alcohols or suffer from poor selectivity.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the method uses readily available reagents like cuprous chloride and Grignard reagents, and the products are easy to separate and purify, making it highly scalable for industrial production.
Q: What kind of stereoselectivity can be expected?
A: The reaction demonstrates high stereoselectivity, capable of producing optically active beta-functionalized allyl alcohols with retained enantiomeric excess when chiral substrates are used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Functionalized Allyl Alcohol Supplier
The technical potential of this copper-catalyzed carbometallation route is immense, offering a pathway to complex molecular structures that are otherwise difficult to access. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of beta-functionalized allyl alcohol meets the highest industry standards. We understand the critical nature of these intermediates in the drug development lifecycle and are committed to providing a seamless transition from process development to full-scale manufacturing. Our team of chemists is ready to optimize this route for your specific target molecules, ensuring maximum efficiency and yield.
We invite you to initiate a dialogue with our technical procurement team to explore how this synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic benefits of adopting this technology for your specific projects. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of this method against your current processes. Our goal is to partner with you to deliver high-quality intermediates that accelerate your time to market while reducing overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
