Advanced Glutathione-Responsive Bi-Cyanine Dyes: Technical Breakthroughs and Commercial Scalability
The landscape of molecular imaging is undergoing a significant transformation driven by the need for higher specificity and lower background noise in tumor detection. Patent CN110283474B introduces a groundbreaking class of glutathione-responsive disulfide bond bi-cyanine dyes that address the critical limitations of conventional fluorescent probes. Unlike traditional cyanine dyes which often suffer from non-specific accumulation and poor contrast in complex biological environments, this novel technology leverages a smart activation mechanism triggered by the unique biochemical signature of cancer cells. The core innovation lies in the strategic incorporation of a disulfide linkage that connects two dye molecules, inducing a quenched state through pi-pi stacking interactions until the probe encounters the elevated glutathione concentrations found exclusively within malignant tissues. This patent not only details the chemical architecture but also validates the utility of these compounds for both high-resolution fluorescence imaging and potential photodynamic therapy applications. For pharmaceutical developers and procurement specialists, this represents a pivotal shift towards smarter, activatable diagnostic agents that promise to enhance surgical navigation and pathological analysis with unprecedented clarity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional molecular imaging agents, such as the FDA-approved indocyanine green (ICG), have long been utilized for their biocompatibility and near-infrared absorption properties. However, these first-generation probes fundamentally lack intrinsic tumor-targeting capabilities, relying heavily on passive accumulation which often results in suboptimal signal-to-noise ratios. To achieve specificity, researchers have historically attempted to conjugate these dyes with targeting ligands like folate or RGD peptides, but this strategy invariably complicates the synthetic pathway and introduces significant purification challenges. The resulting conjugates are often expensive to produce due to multi-step synthesis requirements and difficult separation processes, creating a bottleneck for widespread clinical adoption. Furthermore, the constant fluorescence emission of these conventional dyes leads to high background signals in healthy tissues, making it difficult for surgeons to distinguish tumor margins accurately during minimally invasive procedures. This lack of contrast can compromise the efficacy of tumor resection and limit the diagnostic value of the imaging modality in early-stage cancer detection.
The Novel Approach
The technology disclosed in the patent offers a sophisticated solution by utilizing an aggregation-caused quenching (ACQ) mechanism that is reversibly controlled by the intracellular redox environment. By linking two cyanine dye units via a disulfide bond, the molecule adopts a conformation that suppresses fluorescence emission through strong intramolecular and intermolecular pi-pi interactions while circulating in the bloodstream. This ensures that the background signal remains negligible in normal cells where glutathione concentrations are low, effectively eliminating the noise that plagues traditional imaging agents. Upon reaching the tumor microenvironment, the high concentration of glutathione triggers the cleavage of the disulfide bond, causing the dimer to dissociate into monomeric units that restore intense fluorescence emission. This switch-like behavior provides a dramatic increase in imaging resolution, allowing for precise visualization of tumor boundaries without the need for complex external targeting moieties. 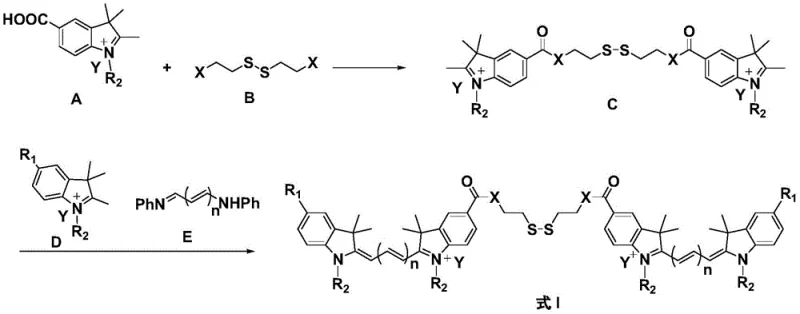
Mechanistic Insights into Disulfide-Mediated Fluorescence Activation
The chemical elegance of this system is rooted in the precise engineering of the electronic properties of the cyanine backbone coupled with the redox sensitivity of the disulfide linker. As illustrated in the general structural formulas, the dye molecules are designed with specific substituents that facilitate strong stacking interactions in the dimeric state, effectively lowering the quantum yield to less than 30% of the monomeric form. This quenching effect is critical for minimizing off-target signals and ensuring that the probe remains dark until it interacts with the specific biochemical trigger. The cleavage mechanism is highly selective for glutathione, which is present at millimolar concentrations in tumor cells compared to micromolar levels in normal plasma, providing a robust biological threshold for activation. Once the bond is broken, the steric hindrance is reduced, and the planar conjugated system of the individual dye molecules is restored, leading to a rapid and significant recovery of fluorescence intensity. 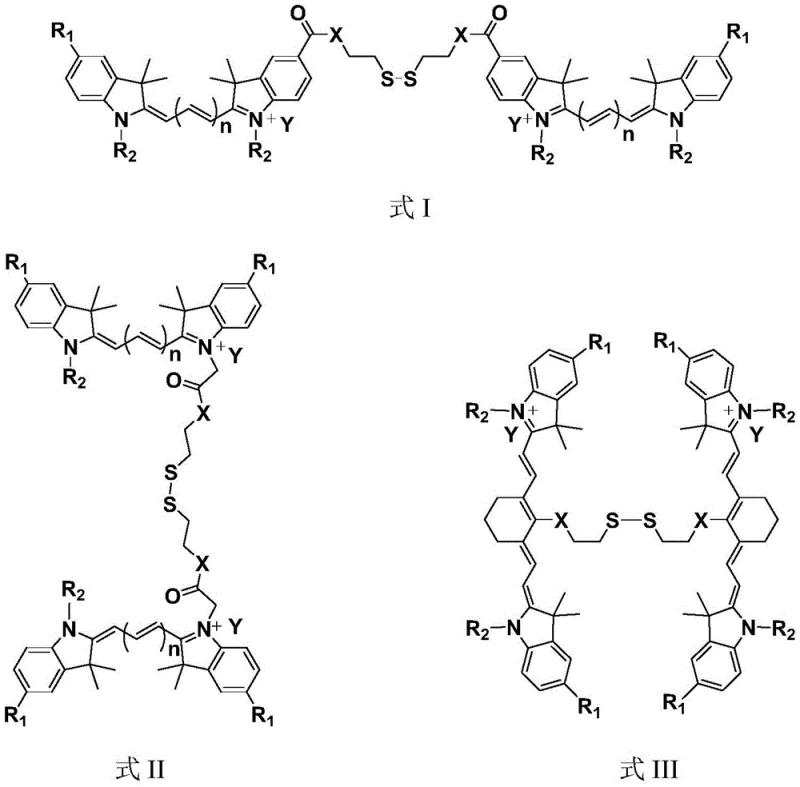
From a quality control and impurity profile perspective, the synthesis route is designed to minimize the formation of side products that could interfere with imaging performance or introduce toxicity. The patent specifies the use of standard condensing agents and solvent systems that allow for efficient purification via column chromatography or recrystallization techniques. This ensures that the final product meets stringent purity specifications required for in vivo applications, reducing the risk of immunogenic reactions or non-specific binding. The ability to tune the emission wavelength by adjusting the polymethine chain length (n=1 to 3) allows for the customization of the probe for different imaging depths and instrument configurations. Such flexibility is essential for R&D teams looking to integrate these probes into existing imaging workflows or develop new theranostic platforms that combine diagnosis and therapy in a single molecular entity.
How to Synthesize Glutathione-Responsive Bi-Cyanine Dye Efficiently
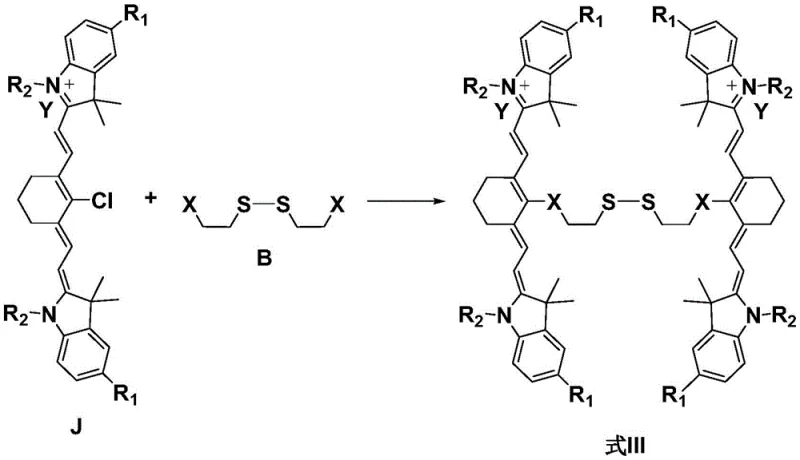
The synthesis of these advanced fluorescent probes follows a modular approach that allows for the systematic assembly of the disulfide core and the cyanine fluorophores. The process begins with the activation of carboxyl-functionalized indole derivatives, which are then coupled with disulfide-containing linkers to form the central scaffold. This intermediate is subsequently condensed with heterocyclic salts to extend the conjugated system, creating the final bi-cyanine structure. The patent outlines specific reaction conditions, including temperature ranges and solvent ratios, that optimize yield and minimize degradation of the sensitive disulfide bond. Detailed standardized synthesis steps are provided below to guide process chemists in replicating this high-value intermediate.
- React carboxyl indole compounds with disulfide bond compounds containing active groups using condensing agents like HATU or TSTU in polar solvents.
- Condense the resulting bis-indole intermediate with hemicyanine precursors in an acetic acid-anhydride mixture to form the conjugated system.
- Purify the final crude product via column chromatography or recrystallization to achieve high-purity fluorescent probes suitable for biological applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits in terms of cost efficiency and operational reliability. The streamlined nature of the reaction sequence reduces the number of unit operations required, directly translating to lower labor costs and reduced consumption of utilities such as heating and cooling. By eliminating the need for expensive targeting ligands and complex bioconjugation steps, the overall cost of goods sold is significantly reduced, making high-performance imaging agents more accessible for routine clinical use. The reliance on commercially available starting materials, such as substituted indoles and common coupling reagents, mitigates the risk of supply chain disruptions associated with exotic or proprietary raw materials. This accessibility ensures a stable supply pipeline, allowing manufacturers to maintain consistent production schedules even in volatile market conditions.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent avoids the use of precious metal catalysts or harsh reaction conditions that typically drive up processing costs. By utilizing mild temperatures and standard organic solvents, the process reduces energy consumption and simplifies waste management protocols. The high yields reported in the examples indicate efficient atom economy, meaning less raw material is wasted during production. Furthermore, the simplified purification process reduces the consumption of chromatography media and solvents, contributing to substantial cost savings in downstream processing. These factors collectively enhance the economic viability of producing these dyes at a commercial scale.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various indole derivatives and disulfide linkers, are widely produced by multiple chemical suppliers globally. This diversity in the supplier base reduces dependency on single-source vendors and minimizes the risk of shortages that could delay production timelines. The robustness of the chemical reactions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with batch-to-batch fluctuations. This reliability is crucial for maintaining long-term contracts with pharmaceutical partners who require guaranteed delivery of critical imaging intermediates.
- Scalability and Environmental Compliance: The patent explicitly states that the method is suitable for industrial production, highlighting its potential for seamless scale-up from laboratory to manufacturing plant. The reaction conditions are mild enough to be handled in standard glass-lined or stainless steel reactors without requiring specialized high-pressure equipment. Additionally, the absence of heavy metals and toxic reagents simplifies environmental compliance and waste disposal, aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glutathione-responsive dye technology. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages and application scope for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this chemistry into your existing product portfolio or research pipeline.
Q: How does the disulfide bond improve tumor imaging resolution compared to traditional cyanine dyes?
A: The disulfide bond induces aggregation-caused quenching in normal tissues, keeping background fluorescence low. Upon encountering high glutathione levels in tumor cells, the bond cleaves, restoring strong fluorescence specifically at the target site.
Q: Are the raw materials for this synthesis readily available for large-scale production?
A: Yes, the synthesis utilizes common indole derivatives and standard condensing agents. The patent highlights that raw materials are easy to prepare, supporting scalable industrial manufacturing without exotic reagents.
Q: What are the stability characteristics of these bi-cyanine dyes in physiological conditions?
A: The dyes exhibit good stability and low toxicity. They remain stable in circulation until they reach the reducing environment of tumor cells, ensuring reliable performance during diagnostic and therapeutic procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glutathione-Responsive Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of activatable fluorescent probes in the next generation of precision medicine. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. We are committed to delivering these complex intermediates with stringent purity specifications and rigorous QC labs to guarantee performance consistency. Our facility is equipped to handle the specific purification requirements of sensitive cyanine dyes, ensuring that every batch meets the high standards required for in vivo diagnostic applications.
We invite you to collaborate with us to optimize your supply chain for these advanced imaging agents. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your regulatory filings and development timelines. Let us help you accelerate the deployment of high-resolution tumor imaging solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
